Food Waste Composting and Microbial Community Structure Profiling
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
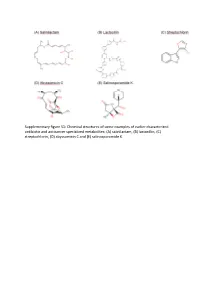
Chemical Structures of Some Examples of Earlier Characterized Antibiotic and Anticancer Specialized
Supplementary figure S1: Chemical structures of some examples of earlier characterized antibiotic and anticancer specialized metabolites: (A) salinilactam, (B) lactocillin, (C) streptochlorin, (D) abyssomicin C and (E) salinosporamide K. Figure S2. Heat map representing hierarchical classification of the SMGCs detected in all the metagenomes in the dataset. Table S1: The sampling locations of each of the sites in the dataset. Sample Sample Bio-project Site depth accession accession Samples Latitude Longitude Site description (m) number in SRA number in SRA AT0050m01B1-4C1 SRS598124 PRJNA193416 Atlantis II water column 50, 200, Water column AT0200m01C1-4D1 SRS598125 21°36'19.0" 38°12'09.0 700 and above the brine N "E (ATII 50, ATII 200, 1500 pool water layers AT0700m01C1-3D1 SRS598128 ATII 700, ATII 1500) AT1500m01B1-3C1 SRS598129 ATBRUCL SRS1029632 PRJNA193416 Atlantis II brine 21°36'19.0" 38°12'09.0 1996– Brine pool water ATBRLCL1-3 SRS1029579 (ATII UCL, ATII INF, N "E 2025 layers ATII LCL) ATBRINP SRS481323 PRJNA219363 ATIID-1a SRS1120041 PRJNA299097 ATIID-1b SRS1120130 ATIID-2 SRS1120133 2168 + Sea sediments Atlantis II - sediments 21°36'19.0" 38°12'09.0 ~3.5 core underlying ATII ATIID-3 SRS1120134 (ATII SDM) N "E length brine pool ATIID-4 SRS1120135 ATIID-5 SRS1120142 ATIID-6 SRS1120143 Discovery Deep brine DDBRINP SRS481325 PRJNA219363 21°17'11.0" 38°17'14.0 2026– Brine pool water N "E 2042 layers (DD INF, DD BR) DDBRINE DD-1 SRS1120158 PRJNA299097 DD-2 SRS1120203 DD-3 SRS1120205 Discovery Deep 2180 + Sea sediments sediments 21°17'11.0" -

Nitrobacter As an Indicator of Toxicity in Wastewater
State Water Survey Division WATER QUALITY SECTION AT Illinois Department of PEORIA, ILLINOIS Energy and Natural Resources SWS Contract Report 326 NITROBACTER AS AN INDICATOR OF TOXICITY IN WASTEWATER by Wuncheng Wang and Paula Reed Prepared for and funded by the Illinois Department of Energy and Natural Resources September 1983 CONTENTS PAGE Abstract 1 Introduction 1 Scope of study 3 Acknowledgments 3 Literature review 3 Microbial nitrification 3 Influence of toxicants on nitrification 5 Materials and methods 10 Culture 10 Methods 11 Results 12 Preliminary tests 13 Metal toxicity 13 Organic compounds toxicity 16 Time effect 22 Discussion 22 References 27 NITROBACTER AS AN INDICATOR OF TOXICITY IN WASTEWATER by Wuncheng Wang and Paula Reed ABSTRACT This report presents the results of a study of the use of Nitrobacter as an indicator of toxicity. Nitrobacter are strictly aerobic, autotrophic, and slow growing bacteria. Because they convert nitrite to nitrate, the effects that toxins have on them can be detected easily by monitoring changes in their nitrite consumption rate. The bacterial cultures were obtained from two sources — the Peoria and Princeton (Illinois) wastewater treatment plants — and tests were con• ducted to determine the effects on the cultures of inorganic ions and organic compounds. The inorganic ions included cadmium, copper, lead, and nickel. The organic compounds were phenol, chlorophenol (three derivatives), dichlo- rophenol (two derivatives), and trichlorophenol. The bioassay procedure is relatively simple and the results are repro• ducible . The effects of these chemical compounds on Nitrobacter were not dramatic. For example, of the compounds tested, 2,4,6-trichlorophenol was the most toxic to Nitrobacter. -

Genomics 98 (2011) 370–375
Genomics 98 (2011) 370–375 Contents lists available at ScienceDirect Genomics journal homepage: www.elsevier.com/locate/ygeno Whole-genome comparison clarifies close phylogenetic relationships between the phyla Dictyoglomi and Thermotogae Hiromi Nishida a,⁎, Teruhiko Beppu b, Kenji Ueda b a Agricultural Bioinformatics Research Unit, Graduate School of Agricultural and Life Sciences, University of Tokyo, 1-1-1 Yayoi, Bunkyo-ku, Tokyo 113-8657, Japan b Life Science Research Center, College of Bioresource Sciences, Nihon University, Fujisawa, Japan article info abstract Article history: The anaerobic thermophilic bacterial genus Dictyoglomus is characterized by the ability to produce useful Received 2 June 2011 enzymes such as amylase, mannanase, and xylanase. Despite the significance, the phylogenetic position of Accepted 1 August 2011 Dictyoglomus has not yet been clarified, since it exhibits ambiguous phylogenetic positions in a single gene Available online 7 August 2011 sequence comparison-based analysis. The number of substitutions at the diverging point of Dictyoglomus is insufficient to show the relationships in a single gene comparison-based analysis. Hence, we studied its Keywords: evolutionary trait based on whole-genome comparison. Both gene content and orthologous protein sequence Whole-genome comparison Dictyoglomus comparisons indicated that Dictyoglomus is most closely related to the phylum Thermotogae and it forms a Bacterial systematics monophyletic group with Coprothermobacter proteolyticus (a constituent of the phylum Firmicutes) and Coprothermobacter proteolyticus Thermotogae. Our findings indicate that C. proteolyticus does not belong to the phylum Firmicutes and that the Thermotogae phylum Dictyoglomi is not closely related to either the phylum Firmicutes or Synergistetes but to the phylum Thermotogae. © 2011 Elsevier Inc. -

Diversity of Understudied Archaeal and Bacterial Populations of Yellowstone National Park: from Genes to Genomes Daniel Colman
University of New Mexico UNM Digital Repository Biology ETDs Electronic Theses and Dissertations 7-1-2015 Diversity of understudied archaeal and bacterial populations of Yellowstone National Park: from genes to genomes Daniel Colman Follow this and additional works at: https://digitalrepository.unm.edu/biol_etds Recommended Citation Colman, Daniel. "Diversity of understudied archaeal and bacterial populations of Yellowstone National Park: from genes to genomes." (2015). https://digitalrepository.unm.edu/biol_etds/18 This Dissertation is brought to you for free and open access by the Electronic Theses and Dissertations at UNM Digital Repository. It has been accepted for inclusion in Biology ETDs by an authorized administrator of UNM Digital Repository. For more information, please contact [email protected]. Daniel Robert Colman Candidate Biology Department This dissertation is approved, and it is acceptable in quality and form for publication: Approved by the Dissertation Committee: Cristina Takacs-Vesbach , Chairperson Robert Sinsabaugh Laura Crossey Diana Northup i Diversity of understudied archaeal and bacterial populations from Yellowstone National Park: from genes to genomes by Daniel Robert Colman B.S. Biology, University of New Mexico, 2009 DISSERTATION Submitted in Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy Biology The University of New Mexico Albuquerque, New Mexico July 2015 ii DEDICATION I would like to dedicate this dissertation to my late grandfather, Kenneth Leo Colman, associate professor of Animal Science in the Wool laboratory at Montana State University, who even very near the end of his earthly tenure, thought it pertinent to quiz my knowledge of oxidized nitrogen compounds. He was a man of great curiosity about the natural world, and to whom I owe an acknowledgement for his legacy of intellectual (and actual) wanderlust. -

Developing a Genetic Manipulation System for the Antarctic Archaeon, Halorubrum Lacusprofundi: Investigating Acetamidase Gene Function
www.nature.com/scientificreports OPEN Developing a genetic manipulation system for the Antarctic archaeon, Halorubrum lacusprofundi: Received: 27 May 2016 Accepted: 16 September 2016 investigating acetamidase gene Published: 06 October 2016 function Y. Liao1, T. J. Williams1, J. C. Walsh2,3, M. Ji1, A. Poljak4, P. M. G. Curmi2, I. G. Duggin3 & R. Cavicchioli1 No systems have been reported for genetic manipulation of cold-adapted Archaea. Halorubrum lacusprofundi is an important member of Deep Lake, Antarctica (~10% of the population), and is amendable to laboratory cultivation. Here we report the development of a shuttle-vector and targeted gene-knockout system for this species. To investigate the function of acetamidase/formamidase genes, a class of genes not experimentally studied in Archaea, the acetamidase gene, amd3, was disrupted. The wild-type grew on acetamide as a sole source of carbon and nitrogen, but the mutant did not. Acetamidase/formamidase genes were found to form three distinct clades within a broad distribution of Archaea and Bacteria. Genes were present within lineages characterized by aerobic growth in low nutrient environments (e.g. haloarchaea, Starkeya) but absent from lineages containing anaerobes or facultative anaerobes (e.g. methanogens, Epsilonproteobacteria) or parasites of animals and plants (e.g. Chlamydiae). While acetamide is not a well characterized natural substrate, the build-up of plastic pollutants in the environment provides a potential source of introduced acetamide. In view of the extent and pattern of distribution of acetamidase/formamidase sequences within Archaea and Bacteria, we speculate that acetamide from plastics may promote the selection of amd/fmd genes in an increasing number of environmental microorganisms. -

Thermaerobacter Marianensis Type Strain (7P75a)
Lawrence Berkeley National Laboratory Recent Work Title Complete genome sequence of Thermaerobacter marianensis type strain (7p75a). Permalink https://escholarship.org/uc/item/2km2g1rj Journal Standards in genomic sciences, 3(3) ISSN 1944-3277 Authors Han, Cliff Gu, Wei Zhang, Xiaojing et al. Publication Date 2010-12-15 DOI 10.4056/sigs.1373474 Peer reviewed eScholarship.org Powered by the California Digital Library University of California Standards in Genomic Sciences (2010) 3:337-345 DOI:10.4056/sigs.1373474 Complete genome sequence of Thermaerobacter T marianensis type strain (7p75a ) Cliff Han1,2, Wei Gu1,2, Xiaojing Zhang1,2, Alla Lapidus1, Matt Nolan1, Alex Copeland1, Susan Lucas1, Tijana Glavina Del Rio1, Hope Tice1, Jan-Fang Cheng1, Roxane Tapia1,2, Lynne Goodwin1,2, Sam Pitluck1, Ioanna Pagani1, Natalia Ivanova1, Konstantinos Mavromatis1, Natalia Mikhailova1, Amrita Pati1, Amy Chen3, Krishna Palaniappan3, Miriam Land1,4, Loren Hauser1,4, Yun-Juan Chang1,4, Cynthia D. Jeffries1,4, Susanne Schneider5, Manfred Rohde6, Markus Göker5, Rüdiger Pukall5, Tanja Woyke1, James Bristow1, Jonathan A. Eisen1,7, Victor Markowitz3, Philip Hugenholtz1, Nikos C. Kyrpides1, Hans-Peter Klenk5*, and John C. Detter1,2 1 DOE Joint Genome Institute, Walnut Creek, California, USA 2 Los Alamos National Laboratory, Bioscience Division, Los Alamos, New Mexico, USA 3 Biological Data Management and Technology Center, Lawrence Berkeley National Laboratory, Berkeley, California, USA 4 Oak Ridge National Laboratory, Oak Ridge, Tennessee, USA 5 DSMZ - German Collection of Microorganisms and Cell Cultures GmbH, Braunschweig, Germany 6 HZI – Helmholtz Centre for Infection Research, Braunschweig, Germany 7 University of California Davis Genome Center, Davis, California, USA *Corresponding author: Hans-Peter Klenk Keywords: strictly aerobic, none-motile, Gram-variable, thermophilic, chemoheterotrophic, deep-sea, family Incertae Sedis XVII, Clostridiales, GEBA Thermaerobacter marianensis Takai et al. -

Sporulation Evolution and Specialization in Bacillus
bioRxiv preprint doi: https://doi.org/10.1101/473793; this version posted March 11, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. Research article From root to tips: sporulation evolution and specialization in Bacillus subtilis and the intestinal pathogen Clostridioides difficile Paula Ramos-Silva1*, Mónica Serrano2, Adriano O. Henriques2 1Instituto Gulbenkian de Ciência, Oeiras, Portugal 2Instituto de Tecnologia Química e Biológica, Universidade Nova de Lisboa, Oeiras, Portugal *Corresponding author: Present address: Naturalis Biodiversity Center, Marine Biodiversity, Leiden, The Netherlands Phone: 0031 717519283 Email: [email protected] (Paula Ramos-Silva) Running title: Sporulation from root to tips Keywords: sporulation, bacterial genome evolution, horizontal gene transfer, taxon- specific genes, Bacillus subtilis, Clostridioides difficile 1 bioRxiv preprint doi: https://doi.org/10.1101/473793; this version posted March 11, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. Abstract Bacteria of the Firmicutes phylum are able to enter a developmental pathway that culminates with the formation of a highly resistant, dormant spore. Spores allow environmental persistence, dissemination and for pathogens, are infection vehicles. In both the model Bacillus subtilis, an aerobic species, and in the intestinal pathogen Clostridioides difficile, an obligate anaerobe, sporulation mobilizes hundreds of genes. -

Recycling of Vitamin B12 and NAD+ Within the Pdu Microcompartment of Salmonella Enterica Shouqiang Cheng Iowa State University
Iowa State University Capstones, Theses and Graduate Theses and Dissertations Dissertations 2010 Recycling of vitamin B12 and NAD+ within the Pdu microcompartment of Salmonella enterica Shouqiang Cheng Iowa State University Follow this and additional works at: https://lib.dr.iastate.edu/etd Part of the Biochemistry, Biophysics, and Structural Biology Commons Recommended Citation Cheng, Shouqiang, "Recycling of vitamin B12 and NAD+ within the Pdu microcompartment of Salmonella enterica" (2010). Graduate Theses and Dissertations. 11713. https://lib.dr.iastate.edu/etd/11713 This Dissertation is brought to you for free and open access by the Iowa State University Capstones, Theses and Dissertations at Iowa State University Digital Repository. It has been accepted for inclusion in Graduate Theses and Dissertations by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. + Recycling of vitamin B12 and NAD within the Pdu microcompartment of Salmonella enterica by Shouqiang Cheng A dissertation submitted to the graduate faculty in partial fulfillment of the requirements for the degree of DOCTOR OF PHILOSOPHY Major: Biochemistry Program of Study Committee: Thomas A. Bobik, Major Professor Alan DiSpirito Basil Nikolau Reuben Peters Gregory J. Phillips Iowa State University Ames, Iowa 2010 Copyright © Shouqiang Cheng, 2010. All rights reserved. ii Table of contents Abstract............................................................................................................................. -

Organic Matter Decomposition in Simulated Aquaculture Ponds Group Fish Culture and Fisheries Daily Supervisor(S) Dr
O rganic matter decomposition in simulated aquaculture ponds Beatriz Torres Beristain Promotor: Prof. Dr. J.A .J. V erreth H oogleraar in de V isteelt en V isserij W ageningen U niversiteit C o-promotor: Dr. M .C .J. V erdegem U niversitair docent bij the Leerstoelgroep V isteelt en V isserij W ageningen U niversiteit Samenstelling promotiecommissie: Prof. Dr. Y . A vnimelech Technion, Israel Institute of Technology Prof. Dr. Ir. H .J. Gijzen U N ESC O -IH E, Delf, N etherlands Prof. Dr. Ir. M . W .A . V erstegen W ageningen U niversiteit Prof. Dr. Ir. A .A . K oelmans W ageningen U niversiteit Dit onderzoek is uitgevoerd binnen de onderzoekschool W IA S O rganic matter decomposition in simulated aquaculture ponds Beatriz Torres Beristain Proefschrift Ter verkrijging van de graad van doctor O p gezag van de rector magnificus van W ageningen U niversiteit, Prof. Dr. Ir. L. Speelman, In het openbaar te verdedigen O p dinsdag 15 A pril 2005 des namiddags te half tw ee in de A ula Torres Beristain, B. O rganic matter decomposition in simulated aquaculture ponds PhD thesis, Fish C ulture and Fisheries Group, W ageningen Institute of A nimal Sciences. W ageningen U niversity, P.O . Box 338, 6700 A H W ageningen, The N etherlands. - W ith R ef. œW ith summary in Spanish, Dutch and English ISBN : 90-8504-170-8 A Domingo, Y olanda y A lejandro Table of contents C hapter 1 General introduction. 1 C hapter 2 R eview microbial ecology and role in aquaculture ponds. -

Mai Muun Muntant Un an to the Man Uniti
MAIMUUN MUNTANTUS009855303B2 UN AN TO THEMAN UNITI (12 ) United States Patent ( 10 ) Patent No. : US 9 , 855 ,303 B2 McKenzie et al. (45 ) Date of Patent: * Jan . 2 , 2018 ( 54 ) COMPOSITIONS AND METHODS (58 ) Field of Classification Search ??? . A61K 35 / 742 (71 ) Applicant : SERES THERAPEUTICS , INC . , See application file for complete search history . Cambridge, MA (US ) (72 ) Inventors : Gregory McKenzie , Arlington , MA ( 56 ) References Cited (US ) ; Mary - Jane Lombardo McKenzie , Arlington , MA (US ) ; David U . S . PATENT DOCUMENTS N . Cook , Brooklyn , NY (US ) ; Marin 3 ,009 , 864 A 11 / 1961 Gordon - Aldterton et al. Vulic , Boston , MA (US ) ; Geoffrey von 3 ,228 , 838 A 1 / 1966 Rinfret Maltzahn , Boston , MA (US ) ; Brian 3 ,608 ,030 A 9 / 1971 Tint Goodman , Boston , MA (US ) ; John 4 ,077 , 227 A 3 / 1978 Larson Grant Aunins , Doylestown , PA (US ) ; 4 , 205 , 132 A 5 / 1980 Sandine Matthew R . Henn , Somerville , MA 4 ,655 ,047 A 4 / 1987 Temple (US ) ; David Arthur Berry , Brookline , 4 ,689 , 226 A 8 / 1987 Nurmi MA (US ) ; Jonathan Winkler , Boston , 4 ,839 , 281 A 6 / 1989 Gorbach et al . 5 , 196 , 205 A 3 / 1993 Borody MA (US ) 5 , 425 , 951 A 6 / 1995 Goodrich 5 ,436 ,002 A 7 / 1995 Payne ( 73 ) Assignee : Seres Therapeutics , Inc ., Cambridge , 5 ,443 ,826 A 8 / 1995 Borody MA (US ) 5 , 599 , 795 A 2 / 1997 McCann 5 ,648 ,206 A 7 / 1997 Goodrich ( * ) Notice : Subject to any disclaimer , the term of this 5 , 951 , 977 A 9 / 1999 Nisbet et al. patent is extended or adjusted under 35 5 , 965 , 128 A 10 / 1999 Doyle et al . -

Nutrient Control Design Manual: State of Technology Review Report,” Were
United States Office of Research and EPA/600/R‐09/012 Environmental Protection Development January 2009 Agency Washington, DC 20460 Nutrient Control Design Manual State of Technology Review Report EPA/600/R‐09/012 January 2009 Nutrient Control Design Manual State of Technology Review Report by The Cadmus Group, Inc 57 Water Street Watertown, MA 02472 Scientific, Technical, Research, Engineering, and Modeling Support (STREAMS) Task Order 68 Contract No. EP‐C‐05‐058 George T. Moore, Task Order Manager United States Environmental Protection Agency Office of Research and Development / National Risk Management Research Laboratory 26 West Martin Luther King Drive, Mail Code 445 Cincinnati, Ohio, 45268 Notice This document was prepared by The Cadmus Group, Inc. (Cadmus) under EPA Contract No. EP‐C‐ 05‐058, Task Order 68. The Cadmus Team was lead by Patricia Hertzler and Laura Dufresne with Senior Advisors Clifford Randall, Emeritus Professor of Civil and Environmental Engineering at Virginia Tech and Director of the Occoquan Watershed Monitoring Program; James Barnard, Global Practice and Technology Leader at Black & Veatch; David Stensel, Professor of Civil and Environmental Engineering at the University of Washington; and Jeanette Brown, Executive Director of the Stamford Water Pollution Control Authority and Adjunct Professor of Environmental Engineering at Manhattan College. Disclaimer The views expressed in this document are those of the individual authors and do not necessarily, reflect the views and policies of the U.S. Environmental Protection Agency (EPA). Mention of trade names or commercial products does not constitute endorsement or recommendation for use. This document has been reviewed in accordance with EPA’s peer and administrative review policies and approved for publication. -

Phylogeny of Nitrogenase Structural and Assembly Components Reveals New Insights Into the Origin and Distribution of Nitrogen Fixation Across Bacteria and Archaea
microorganisms Article Phylogeny of Nitrogenase Structural and Assembly Components Reveals New Insights into the Origin and Distribution of Nitrogen Fixation across Bacteria and Archaea Amrit Koirala 1 and Volker S. Brözel 1,2,* 1 Department of Biology and Microbiology, South Dakota State University, Brookings, SD 57006, USA; [email protected] 2 Department of Biochemistry, Genetics and Microbiology, University of Pretoria, Pretoria 0004, South Africa * Correspondence: [email protected]; Tel.: +1-605-688-6144 Abstract: The phylogeny of nitrogenase has only been analyzed using the structural proteins NifHDK. As nifHDKENB has been established as the minimum number of genes necessary for in silico predic- tion of diazotrophy, we present an updated phylogeny of diazotrophs using both structural (NifHDK) and cofactor assembly proteins (NifENB). Annotated Nif sequences were obtained from InterPro from 963 culture-derived genomes. Nif sequences were aligned individually and concatenated to form one NifHDKENB sequence. Phylogenies obtained using PhyML, FastTree, RapidNJ, and ASTRAL from individuals and concatenated protein sequences were compared and analyzed. All six genes were found across the Actinobacteria, Aquificae, Bacteroidetes, Chlorobi, Chloroflexi, Cyanobacteria, Deferribacteres, Firmicutes, Fusobacteria, Nitrospira, Proteobacteria, PVC group, and Spirochaetes, as well as the Euryarchaeota. The phylogenies of individual Nif proteins were very similar to the overall NifHDKENB phylogeny, indicating the assembly proteins have evolved together. Our higher resolution database upheld the three cluster phylogeny, but revealed undocu- Citation: Koirala, A.; Brözel, V.S. mented horizontal gene transfers across phyla. Only 48% of the 325 genera containing all six nif genes Phylogeny of Nitrogenase Structural and Assembly Components Reveals are currently supported by biochemical evidence of diazotrophy.