Supplementary Materials
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Tones in Zhangzhou
Tones in Zhangzhou: Pitch and Beyond Tones in Zhangzhou: Pitch and Beyond By Yishan Huang Tones in Zhangzhou: Pitch and Beyond By Yishan Huang This book first published 2020 Cambridge Scholars Publishing Lady Stephenson Library, Newcastle upon Tyne, NE6 2PA, UK British Library Cataloguing in Publication Data A catalogue record for this book is available from the British Library Copyright © 2020 by Yishan Huang All rights for this book reserved. No part of this book may be reproduced, stored in a retrieval system, or transmitted, in any form or by any means, electronic, mechanical, photocopying, recording or otherwise, without the prior permission of the copyright owner. ISBN (10): 1-5275-4421-4 ISBN (13): 978-1-5275-4421-5 TABLE OF CONTENTS Acknowledgments .................................................................................... vii Abstract ................................................................................................... viii Chapter One ................................................................................................ 1 Introduction 1.1. Motivations .................................................................................... 1 1.2. Goals .............................................................................................. 7 1.3. Tonetics .......................................................................................... 8 1.4. Southern Min Tone Sandhi .......................................................... 13 Chapter Two ............................................................................................ -

Protection and Transmission of Chinese Nanyin by Prof
Protection and Transmission of Chinese Nanyin by Prof. Wang, Yaohua Fujian Normal University, China Intangible cultural heritage is the memory of human historical culture, the root of human culture, the ‘energic origin’ of the spirit of human culture and the footstone for the construction of modern human civilization. Ever since China joined the Convention for the Safeguarding of the Intangible Cultural Heritage in 2004, it has done a lot not only on cognition but also on action to contribute to the protection and transmission of intangible cultural heritage. Please allow me to expatiate these on the case of Chinese nanyin(南音, southern music). I. The precious multi-values of nanyin decide the necessity of protection and transmission for Chinese nanyin. Nanyin, also known as “nanqu” (南曲), “nanyue” (南乐), “nanguan” (南管), “xianguan” (弦管), is one of the oldest music genres with strong local characteristics. As major musical genre, it prevails in the south of Fujian – both in the cities and countryside of Quanzhou, Xiamen, Zhangzhou – and is also quite popular in Taiwan, Hongkong, Macao and the countries of Southeast Asia inhabited by Chinese immigrants from South Fujian. The music of nanyin is also found in various Fujian local operas such as Liyuan Opera (梨园戏), Gaojia Opera (高甲戏), line-leading puppet show (提线木偶戏), Dacheng Opera (打城戏) and the like, forming an essential part of their vocal melodies and instrumental music. As the intangible cultural heritage, nanyin has such values as follows. I.I. Academic value and historical value Nanyin enjoys a reputation as “a living fossil of the ancient music”, as we can trace its relevance to and inheritance of Chinese ancient music in terms of their musical phenomena and features of musical form. -

MEDIA STATEMENT Fujian Beckons Singapore Investors As Chinese
MEDIA STATEMENT REVISED - Embargoed until 25 October 2009, 1800hrs Fujian beckons Singapore investors as Chinese Central Government makes plans for its economic transformation Organised by IE Singapore, the business mission is held in conjunction with SM Goh’s visit from 26 – 30 October 2009. MR No.: 063/09 Singapore, Sunday, 25 October 2009 1. In conjunction with Senior Minister Goh Chok Tong’s visit to Fujian province from 26 – 30 October 2009, International Enterprise (IE) Singapore is organising a mission to Xiamen and Fuzhou to help Singapore-based companies seek business opportunities in the province. Besides IE Singapore officials, the 16- strong delegation comprises representatives from 13 companies of various industries, including transport & logistics, tourism, real estate, masterplanning & consultancy, food processing and more. 2. Fujian province’s GDP grew at a stable 8.5% in the first half of this year. Its investment climate is now even more attractive with the recent announcement by the Central Government to elevate the Economic Zone of the West Coast of Taiwan Straits1 (EZWCTS)(海峡西岸经济区)as a national-level strategy. The EZWCTS is being developed to transform Fujian into an economic hub that not only connects the economic powerhouses of the Pearl River Delta and the Yangtze River Delta, but also serves as a springboard to bring overseas investments into Western and Central China. A key focus of the EZWCTS is the development of Pingtan (平潭岛), a group of islands located just 68 nautical 1 The EZWCTS consists of 21 prefecture cities, the focus of which is on the nine Fujian cities of Xiamen, Fuzhou, Quanzhou, Putian, Zhangzhou, Ningde, Sanming, Longyan and Nanping. -

(AHP)-Based Assessment of the Value of Non-World Heritage Tulou
Tourism Management Perspectives 26 (2018) 67–77 Contents lists available at ScienceDirect Tourism Management Perspectives journal homepage: www.elsevier.com/locate/tmp Analytic Hierarchy Process (AHP)-based assessment of the value of non- T World Heritage Tulou: A case study of Pinghe County, Fujian Province ⁎ Hang Maa, Shanting Lib, Chung-Shing Chanc, a Harbin Institute of Technology, Shenzhen Graduate School, Shenzhen 518050, China b Shanghai W&R Group, Shanghai 200052, China c Department of Geography and Resource Management, The Chinese University of Hong Kong, Sha Tin, N.T, Hong Kong ARTICLE INFO ABSTRACT Keywords: China's Fujian Tulou (earthen buildings constructed dating to the 12th century) represent a valuable source of Analytic Hierarchy Process (AHP) human cultural heritage. As the Tulou have not been classified as World Heritage Sites by UNESCO, they lack Conservation and reuse financial support, receive minimal attention and face structural deterioration. The purpose of this study is to Cultural heritage explore a methodological approach to assess the value of non-World Heritage Tulou (NWHT) and provide Evaluation system grounds for the reuse of Tulou accordingly. First, building-type, planar layout and other characteristics of Pinghe Tulou NWHTs in Pinghe are reviewed. Next, an Analytic Hierarchy Process (AHP) is applied to the value evaluation of Pinghe Tulou. Then, policy recommendations for reuse and redevelopment are put forward. The findings suggest that focusing on the reuse of Tulou alone is not justifiable. Rather, funding, public participation and the con- tinuity of community life are important factors relating to the reuse of NWHTs. 1. Introduction Educational, Scientific and Cultural Organization (UNESCO) in 2008 (and are thus referred to here as ‘World Heritage Tulous’ (Fig. -

Deciphering the Spatial Structures of City Networks in the Economic Zone of the West Side of the Taiwan Strait Through the Lens of Functional and Innovation Networks
sustainability Article Deciphering the Spatial Structures of City Networks in the Economic Zone of the West Side of the Taiwan Strait through the Lens of Functional and Innovation Networks Yan Ma * and Feng Xue School of Architecture and Urban-Rural Planning, Fuzhou University, Fuzhou 350108, Fujian, China; [email protected] * Correspondence: [email protected] Received: 17 April 2019; Accepted: 21 May 2019; Published: 24 May 2019 Abstract: Globalization and the spread of information have made city networks more complex. The existing research on city network structures has usually focused on discussions of regional integration. With the development of interconnections among cities, however, the characterization of city network structures on a regional scale is limited in the ability to capture a network’s complexity. To improve this characterization, this study focused on network structures at both regional and local scales. Through the lens of function and innovation, we characterized the city network structure of the Economic Zone of the West Side of the Taiwan Strait through a social network analysis and a Fast Unfolding Community Detection algorithm. We found a significant imbalance in the innovation cooperation among cities in the region. When considering people flow, a multilevel spatial network structure had taken shape. Among cities with strong centrality, Xiamen, Fuzhou, and Whenzhou had a significant spillover effect, which meant the region was depolarizing. Quanzhou and Ganzhou had a significant siphon effect, which was unsustainable. Generally, urbanization in small and midsize cities was common. These findings provide support for government policy making. Keywords: city network; spatial organization; people flows; innovation network 1. -

Effective Inhibition of Mrna Accumulation and Protein Expression of H5N1 Avian Influenza Virus NS1 Gene in Vitro by Small Interfering Rnas
Folia Microbiol (2013) 58:335–342 DOI 10.1007/s12223-012-0212-8 Effective inhibition of mRNA accumulation and protein expression of H5N1 avian influenza virus NS1 gene in vitro by small interfering RNAs Hanwei Jiao & Li Du & Yongchang Hao & Ying Cheng & Jing Luo & Wenhua Kuang & Donglin Zhang & Ming Lei & Xiaoxiao Jia & Xiaoru Zhang & Chao Qi & Hongxuan He & Fengyang Wang Received: 18 June 2012 /Accepted: 7 November 2012 /Published online: 29 November 2012 # Institute of Microbiology, Academy of Sciences of the Czech Republic, v.v.i. 2012 Abstract Avian influenza has emerged as a devastating protein expressing HEK293 cell lines were established, four disease and may cross species barrier and adapt to a new small interfering RNAs (siRNAs) targeting NS1 gene were host, causing enormous economic loss and great public designed, synthesized, and used to transfect the stable cell health threats, and non-structural protein 1 (NS1) is a mul- lines. Flow cytometry, real-time quantitative polymerase tifunctional non-structural protein of avian influenza virus chain reaction, and Western blot were performed to assess (AIV) that counters cellular antiviral activities and is a the expression level of NS1. The results suggested that virulence factor. RNA interference (RNAi) provides a pow- sequence-dependent specific siRNAs effectively inhibited erful promising approach to inhibit viral infection specifi- mRNA accumulation and protein expression of AIV NS1 cally. To explore the possibility of using RNAi as a strategy in vitro. These findings provide useful information for the against AIV infection, after the fusion protein expression development of RNAi-based prophylaxis and therapy for plasmids pNS1-enhanced green fluorescent protein (EGFP), AIV infection. -

Guorui Properties Limited 國瑞置業有限公司
Hong Kong Exchanges and Clearing Limited and The Stock Exchange of Hong Kong Limited take no responsibility for the contents of this announcement, make no representation as to its accuracy or completeness and expressly disclaim any liability whatsoever for any loss howsoever arising from or in reliance upon the whole or any part of the contents of this announcement. GUORUI PROPERTIES LIMITED 國瑞置業有限公司 (Incorporated in the Cayman Islands with limited liability under the name of “Glory Land Company Limited (國瑞置業有限公司)” and carrying on business in Hong Kong as “Guorui Properties Limited”) (Stock Code: 2329) INTERIM RESULTS ANNOUNCEMENT FOR THE SIX MONTHS ENDED JUNE 30, 2019 FINANCIAL HIGHLIGHTS – Achieved contracted sales for the six months ended June 30, 2019 (the “Reporting Period”) was RMB12,828.3 million with corresponding gross floor area (“GFA”) of approximately 604,636 sq.m., representing a period-on-period increase of 87.5% and 7.3%, respectively; – Revenue for the Reporting Period was RMB3,774.9 million, of which the revenue from property development was RMB3,411.7 million; – Gross profit for the Reporting Period was RMB1,074.7 million, of which the gross profit from property development was RM804.6 million; – Net profit for the Reporting Period was RMB569.8 million, of which RMB428.6 million was attributable to the owners of the Company; – Basic earnings per share for the Reporting Period were RMB9.6 cents; – Land reserves reached a total GFA of 16,084,092 sq.m. and the average cost of land reserves was RMB2,877.8 per sq.m. for the Reporting Period; – Contracted average selling price (“ASP”) for the Reporting Period was RMB21,216.7 per sq.m. -

Haikou Is a Tropical Coastal City, Situated on the Northern Coast of Hainan, by the Mouth of the Nandu River
Haikou is a tropical coastal city, situated on the northern coast of Hainan, by the mouth of the Nandu River. The northern part of the city is Haidian Island, which is separated from the main part of Haikou by the Haidian River, a branch of the Nandu. Haikou was originally a port city. Today, it’s the capital of Hainan province and the most populous city in the province. The city covers 2,280 square kilometers and is home to around 2 million inhabitants. Haikou is on the northern edge of the torrid zone, and is part of the inter-tropical Convergence Zone. April to October is the active period for tropical storms and typhoons, most of which occur between August and September. May to October is the rainy season with the heaviest rainfall occurring in September. Despite its location, the city has a humid subtropical climate, falling just short of a tropical climate, with strong monsoonal influences. Nevertheless, the area has hot summers and warm winters, usually with high humidity. Extremes temperatures have ranged from 2.8 to 39.6 °C (37 to 103 °F) From June to October torrential rains may occur, with 7.0 days annually receiving at least 50 mm (1.97 in) of rain; this period accounts for nearly 70 percent of the annual rainfall of 1,650 mm (65 in). With monthly percentage of possible sunshine ranging from 31% in February to 61% in July, the city receives 2,070 hours of bright sunshine annually. Haikou is a transport hub with flights to many cities and boats to nearby mainland cities such as Beihai and Zhanjiang. -
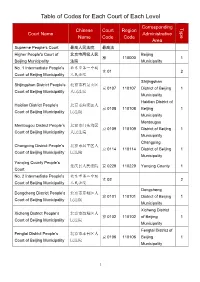
Table of Codes for Each Court of Each Level
Table of Codes for Each Court of Each Level Corresponding Type Chinese Court Region Court Name Administrative Name Code Code Area Supreme People’s Court 最高人民法院 最高法 Higher People's Court of 北京市高级人民 Beijing 京 110000 1 Beijing Municipality 法院 Municipality No. 1 Intermediate People's 北京市第一中级 京 01 2 Court of Beijing Municipality 人民法院 Shijingshan Shijingshan District People’s 北京市石景山区 京 0107 110107 District of Beijing 1 Court of Beijing Municipality 人民法院 Municipality Haidian District of Haidian District People’s 北京市海淀区人 京 0108 110108 Beijing 1 Court of Beijing Municipality 民法院 Municipality Mentougou Mentougou District People’s 北京市门头沟区 京 0109 110109 District of Beijing 1 Court of Beijing Municipality 人民法院 Municipality Changping Changping District People’s 北京市昌平区人 京 0114 110114 District of Beijing 1 Court of Beijing Municipality 民法院 Municipality Yanqing County People’s 延庆县人民法院 京 0229 110229 Yanqing County 1 Court No. 2 Intermediate People's 北京市第二中级 京 02 2 Court of Beijing Municipality 人民法院 Dongcheng Dongcheng District People’s 北京市东城区人 京 0101 110101 District of Beijing 1 Court of Beijing Municipality 民法院 Municipality Xicheng District Xicheng District People’s 北京市西城区人 京 0102 110102 of Beijing 1 Court of Beijing Municipality 民法院 Municipality Fengtai District of Fengtai District People’s 北京市丰台区人 京 0106 110106 Beijing 1 Court of Beijing Municipality 民法院 Municipality 1 Fangshan District Fangshan District People’s 北京市房山区人 京 0111 110111 of Beijing 1 Court of Beijing Municipality 民法院 Municipality Daxing District of Daxing District People’s 北京市大兴区人 京 0115 -

Info Bulletin.Pmd
The East Asian Seas Congress 2006 The year 2003 created new opportunities for the sustainable development of the seas of East Asia, as it heralded the birth of the first-ever East Asian Seas Congress. The five-day event, held from 8-12 December in Putrajaya, Malaysia, brought together various stakeholders from diverse disciplines to work hand-in-hand to uphold the welfare of the East Asian Seas and ensure the sustainability of their resources. It will be remembered as the occasion where the Sustainable Development Strategy for the Seas of East Asia (SDS-SEA) was endorsed by environment- and ocean- related ministers and senior officials from PEMSEA’s 12 participating countries, with the signing of the Putrajaya Declaration. This Congress, a follow-up to the Putrajaya event, promises to drive the wheels of sustainable development in continuous motion, thereby bringing East Asia closer and closer to ultimately achieving a healthy and bountiful future for its seas and its people. Designed to address pressing coastal and marine environmental issues in practical formats and against a multidisciplinary setting, the various Congress components provide participants the opportunity to take active roles in sustainable development. Features The EAS Congress 2006 features the: • Ministerial Forum on the Implementation of the SDS-SEA* 14–15 December; • International Conference on Coastal and Ocean Governance: One Ocean, One People, One Vision 12–14 December; • Meeting of the EAS Partnership Council* 16 December; and • Youth Forum, Exhibits, Poster Sessions, Field Trip and other Side Events. *By invitation only. Host/Local Organizing Committee The 2006 Congress is being hosted by the government of PR China and co-organized by the State Oceanic Administration (SOA). -

Chen Hawii 0085A 10047.Pdf
PROTO-ONG-BE A DISSERTATION SUBMITTED TO THE GRADUATE DIVISION OF THE UNIVERSITY OF HAWAIʻI AT MĀNOA IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY IN LINGUISTICS DECEMBER 2018 By Yen-ling Chen Dissertation Committee: Lyle Campbell, Chairperson Weera Ostapirat Rory Turnbull Bradley McDonnell Shana Brown Keywords: Ong-Be, Reconstruction, Lingao, Hainan, Kra-Dai Copyright © 2018 by Yen-ling Chen ii 知之為知之,不知為不知,是知也。 “Real knowledge is to know the extent of one’s ignorance.” iii Acknowlegements First of all, I would like to acknowledge Dr. Lyle Campbell, the chair of my dissertation and the historical linguist and typologist in my department for his substantive comments. I am always amazed by his ability to ask mind-stimulating questions, and I thank him for allowing me to be part of the Endangered Languages Catalogue (ELCat) team. I feel thankful to Dr. Shana Brown for bringing historical studies on minorities in China to my attention, and for her support as the university representative on my committee. Special thanks go to Dr. Rory Turnbull for his constructive comments and for encouraging a diversity of point of views in his class, and to Dr. Bradley McDonnell for his helpful suggestions. I sincerely thank Dr. Weera Ostapirat for his time and patience in dealing with me and responding to all my questions, and for pointing me to the directions that I should be looking at. My reconstruction would not be as readable as it is today without his insightful feedback. I would like to express my gratitude to Dr. Alexis Michaud. -

Evergrande Real Estate Group Limited 恒 大 地 產 集 團 有 限 公 司 (Incorporated in the Cayman Islands with Limited Liability) (Stock Code: 3333)
Hong Kong Exchanges and Clearing Limited and The Stock Exchange of Hong Kong Limited take no responsibility for the contents of this announcement, make no representation as to its accuracy or completeness and expressly disclaim any liability whatsoever for any loss howsoever arising from or in reliance upon the whole or any part of the contents of this announcement. Evergrande Real Estate Group Limited 恒 大 地 產 集 團 有 限 公 司 (Incorporated in the Cayman Islands with limited liability) (Stock Code: 3333) DISCLOSEABLE TRANSACTION The Board announces that on 2 December 2015, Shengyu (BVI) Limited, a wholly-owned subsidiary of the Company, as the purchaser, entered into three agreements respectively with the Vendor, pursuant to which the Purchaser agreed to acquire the interests in the relevant shares in and loans to the target companies held by the Vendor. These target companies hold the interests in the Haikou Project, the Huiyang Project and two projects located in Wuhan. Since the Vendor is the seller in these three agreements, such agreements will be aggregated in accordance with Rule 14.22 of the Listing Rules. As the applicable percentage ratios for the Acquisition exceed 5% but are less than 25%, the Acquisition constitutes a discloseable transaction for the Company under Chapter 14 of the Listing Rules and is subject to the reporting and announcement requirements under Chapter 14 of the Listing Rules. 1. INTRODUCTION The Board announces that on 2 December 2015, Shengyu (BVI) Limited, a wholly-owned subsidiary of the Company, as the purchaser, entered into three agreements respectively with the Vendor, pursuant to which the Purchaser agreed to acquire the interests in the relevant shares in and loans to the target companies held by the Vendor.