Primary Immunodeficiency Disorders
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

IDF Patient & Family Handbook
Immune Deficiency Foundation Patient & Family Handbook for Primary Immunodeficiency Diseases This book contains general medical information which cannot be applied safely to any individual case. Medical knowledge and practice can change rapidly. Therefore, this book should not be used as a substitute for professional medical advice. FIFTH EDITION COPYRIGHT 1987, 1993, 2001, 2007, 2013 IMMUNE DEFICIENCY FOUNDATION Copyright 2013 by Immune Deficiency Foundation, USA. REPRINT 2015 Readers may redistribute this article to other individuals for non-commercial use, provided that the text, html codes, and this notice remain intact and unaltered in any way. The Immune Deficiency Foundation Patient & Family Handbook may not be resold, reprinted or redistributed for compensation of any kind without prior written permission from the Immune Deficiency Foundation. If you have any questions about permission, please contact: Immune Deficiency Foundation, 110 West Road, Suite 300, Towson, MD 21204, USA; or by telephone at 800-296-4433. Immune Deficiency Foundation Patient & Family Handbook for Primary Immunodeficency Diseases 5th Edition This publication has been made possible through a generous grant from Baxalta Incorporated Immune Deficiency Foundation 110 West Road, Suite 300 Towson, MD 21204 800-296-4433 www.primaryimmune.org [email protected] EDITORS R. Michael Blaese, MD, Executive Editor Francisco A. Bonilla, MD, PhD Immune Deficiency Foundation Boston Children’s Hospital Towson, MD Boston, MA E. Richard Stiehm, MD M. Elizabeth Younger, CPNP, PhD University of California Los Angeles Johns Hopkins Los Angeles, CA Baltimore, MD CONTRIBUTORS Mark Ballow, MD Joseph Bellanti, MD R. Michael Blaese, MD William Blouin, MSN, ARNP, CPNP State University of New York Georgetown University Hospital Immune Deficiency Foundation Miami Children’s Hospital Buffalo, NY Washington, DC Towson, MD Miami, FL Francisco A. -

My Beloved Neutrophil Dr Boxer 2014 Neutropenia Family Conference
The Beloved Neutrophil: Its Function in Health and Disease Stem Cell Multipotent Progenitor Myeloid Lymphoid CMP IL-3, SCF, GM-CSF CLP Committed Progenitor MEP GMP GM-CSF, IL-3, SCF EPO TPO G-CSF M-CSF IL-5 IL-3 SCF RBC Platelet Neutrophil Monocyte/ Basophil B-cells Macrophage Eosinophil T-Cells Mast cell NK cells Mature Cell Dendritic cells PRODUCTION AND KINETICS OF NEUTROPHILS CELLS % CELLS TIME Bone Marrow: Myeloblast 1 7 - 9 Mitotic Promyelocyte 4 Days Myelocyte 16 Maturation/ Metamyelocyte 22 3 – 7 Storage Band 30 Days Seg 21 Vascular: Peripheral Blood Seg 2 6 – 12 hours 3 Marginating Pool Apoptosis and ? Tissue clearance by 0 – 3 macrophages days PHAGOCYTOSIS 1. Mobilization 2. Chemotaxis 3. Recognition (Opsonization) 4. Ingestion 5. Degranulation 6. Peroxidation 7. Killing and Digestion 8. Net formation Adhesion: β 2 Integrins ▪ Heterodimer of a and b chain ▪ Tight adhesion, migration, ingestion, co- stimulation of other PMN responses LFA-1 Mac-1 (CR3) p150,95 a2b2 a CD11a CD11b CD11c CD11d b CD18 CD18 CD18 CD18 Cells All PMN, Dendritic Mac, mono, leukocytes mono/mac, PMN, T cell LGL Ligands ICAMs ICAM-1 C3bi, ICAM-3, C3bi other other Fibrinogen other GRANULOCYTE CHEMOATTRACTANTS Chemoattractants Source Activators Lipids PAF Neutrophils C5a, LPS, FMLP Endothelium LTB4 Neutrophils FMLP, C5a, LPS Chemokines (a) IL-8 Monocytes, endothelium LPS, IL-1, TNF, IL-3 other cells Gro a, b, g Monocytes, endothelium IL-1, TNF other cells NAP-2 Activated platelets Platelet activation Others FMLP Bacteria C5a Activation of complement Other Important Receptors on PMNs ñ Pattern recognition receptors – Detect microbes - Toll receptor family - Mannose receptor - bGlucan receptor – fungal cell walls ñ Cytokine receptors – enhance PMN function - G-CSF, GM-CSF - TNF Receptor ñ Opsonin receptors – trigger phagocytosis - FcgRI, II, III - Complement receptors – ñ Mac1/CR3 (CD11b/CD18) – C3bi ñ CR-1 – C3b, C4b, C3bi, C1q, Mannose binding protein From JG Hirsch, J Exp Med 116:827, 1962, with permission. -

The Case for Lupus Nephritis
Journal of Clinical Medicine Review Expanding the Role of Complement Therapies: The Case for Lupus Nephritis Nicholas L. Li * , Daniel J. Birmingham and Brad H. Rovin Department of Internal Medicine, Division of Nephrology, The Ohio State University, Columbus, OH 43210, USA; [email protected] (D.J.B.); [email protected] (B.H.R.) * Correspondence: [email protected]; Tel.: +1-614-293-4997; Fax: +1-614-293-3073 Abstract: The complement system is an innate immune surveillance network that provides defense against microorganisms and clearance of immune complexes and cellular debris and bridges innate and adaptive immunity. In the context of autoimmune disease, activation and dysregulation of complement can lead to uncontrolled inflammation and organ damage, especially to the kidney. Systemic lupus erythematosus (SLE) is characterized by loss of tolerance, autoantibody production, and immune complex deposition in tissues including the kidney, with inflammatory consequences. Effective clearance of immune complexes and cellular waste by early complement components protects against the development of lupus nephritis, while uncontrolled activation of complement, especially the alternative pathway, promotes kidney damage in SLE. Therefore, complement plays a dual role in the pathogenesis of lupus nephritis. Improved understanding of the contribution of the various complement pathways to the development of kidney disease in SLE has created an opportunity to target the complement system with novel therapies to improve outcomes in lupus nephritis. In this review, we explore the interactions between complement and the kidney in SLE and their implications for the treatment of lupus nephritis. Keywords: lupus nephritis; complement; systemic lupus erythematosus; glomerulonephritis Citation: Li, N.L.; Birmingham, D.J.; Rovin, B.H. -
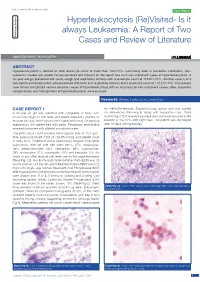
Hyperleukocytosis (Re)Visited- Is It Case Series Always Leukaemia: a Report of Two Pathology Section Cases and Review of Literature Short Communication
Review Article Clinician’s corner Original Article Images in Medicine Experimental Research Miscellaneous Letter to Editor DOI: 10.7860/JCDR/2020/40556.13409 Case Report Postgraduate Education Hyperleukocytosis (Re)Visited- Is it Case Series always Leukaemia: A Report of Two Pathology Section Cases and Review of Literature Short Communication ASHUTOSH RATH1, RICHA GUPTA2 ABSTRACT Hyperleukocytosis is defined as total leukocyte count of more than 100×109/L. Commonly seen in leukaemic conditions, non- leukaemic causes are usually not encountered and thought of. We report two such non-malignant cases of hyperleukocytosis. A six-year old girl presented with fever, cough and respiratory distress with a leukocyte count of 125.97×109/L. Another case is of a two-month old female infant, who presented with fever and respiratory distress and a leukocyte count of 112.27×109/L. The present case thrives to highlight various possible causes of hyperleukocytosis with an emphasis on non-malignant causes. Also, important complications and management of hyperleukocytosis are discussed. Keywords: Benign, Leukocytosis, Leukostasis CASE REPORT 1 for methicillin-resistant Staphylococcus aureus and was started A six-year-old girl was admitted with complaints of fever, non- on intravenous Vancomycin along with supportive care. Serial productive cough for one week and severe respiratory distress for monitoring of TLC revealed a gradual reduction and it returned to the the past one day. There was no other significant history. On physical baseline of 15×109/L after eight days. The patient was discharged examination, the patient had mild pallor. Respiratory examination after 10 days of hospital stay. -
Instant Notes: Immunology, Second Edition
Immunology Second Edition The INSTANT NOTES series Series Editor: B.D. Hames School of Biochemistry and Molecular Biology, University of Leeds, Leeds, UK Animal Biology 2nd edition Biochemistry 2nd edition Bioinformatics Chemistry for Biologists 2nd edition Developmental Biology Ecology 2nd edition Immunology 2nd edition Genetics 2nd edition Microbiology 2nd edition Molecular Biology 2nd edition Neuroscience Plant Biology Chemistry series Consulting Editor: Howard Stanbury Analytical Chemistry Inorganic Chemistry 2nd edition Medicinal Chemistry Organic Chemistry 2nd edition Physical Chemistry Psychology series Sub-series Editor: Hugh Wagner Dept of Psychology, University of Central Lancashire, Preston, UK Psychology Cognitive Psychology Forthcoming title Physiological Psychology Immunology Second Edition P.M. Lydyard Department of Immunology and Molecular Pathology, Royal Free and University College Medical School, University College London, London, UK A. Whelan Department of Immunology, Trinity College and St James’ Hospital, Dublin, Ireland and M.W. Fanger Department of Microbiology and Immunology, Dartmouth Medical School, Lebanon, New Hampshire, USA © Garland Science/BIOS Scientific Publishers Limited, 2004 First published 2000 This edition published in the Taylor & Francis e-Library, 2005. “To purchase your own copy of this or any of Taylor & Francis or Routledge’s collection of thousands of eBooks please go to www.eBookstore.tandf.co.uk.” Second edition published 2004 All rights reserved. No part of this book may be reproduced or -

Chapter 2 Agammaglobulinemia: X-Linked and Autosomal Recessive
Agammaglobulinemia: X-Linked and Autosomal Recessive Chapter 2 Agammaglobulinemia: X-Linked and Autosomal Recessive The basic defect in both X-Linked Agammaglobulinemia and autosomal recessive agammaglobulinemia is a failure of B-lymphocyte precursors to mature into B-lymphocytes and ultimately plasma cells. Since they lack the cells that are responsible for producing immunoglobulins, these patients have severe deficiencies of all types of immunoglobulins. Definition of X-Linked Agammaglobulinemia (XLA) and Autosomal Recessive Agammaglobulinemia (ARA) X-Linked Agammaglobulinemia (XLA) was first are not coated with antibody. All of these actions described in 1952 by Dr. Ogden Bruton. This disease, prevent germs from invading body tissues where they sometimes called Bruton’s Agammaglobulinemia or may cause serious infections. (See chapter titled “The Congenital Agammaglobulinemia, was one of the first Immune System and Primary Immunodeficiency immunodeficiency diseases to be identified. XLA is an Diseases.”) inherited immunodeficiency disease in which patients The basic defect in XLA is an inability of the patient to lack the ability to produce antibodies, the proteins that produce antibodies. Antibodies are produced by make up the gamma globulin or immunoglobulin specialized cells in the body, called plasma cells. fraction of blood plasma. Plasma cells develop in an orderly sequence of steps Antibodies are an integral part of the body’s defense beginning with stem cells located in the bone marrow. mechanism against certain types of microorganisms or The stem cells give rise to immature lymphocytes, germs, like bacteria or viruses. Antibodies are important called pro-B-lymphocytes. Pro-B-lymphocytes next in the recovery from infections and protect against develop into pre-B-cells, which then give rise to getting certain infections more than once. -

Practice Parameter for the Diagnosis and Management of Primary Immunodeficiency
Practice parameter Practice parameter for the diagnosis and management of primary immunodeficiency Francisco A. Bonilla, MD, PhD, David A. Khan, MD, Zuhair K. Ballas, MD, Javier Chinen, MD, PhD, Michael M. Frank, MD, Joyce T. Hsu, MD, Michael Keller, MD, Lisa J. Kobrynski, MD, Hirsh D. Komarow, MD, Bruce Mazer, MD, Robert P. Nelson, Jr, MD, Jordan S. Orange, MD, PhD, John M. Routes, MD, William T. Shearer, MD, PhD, Ricardo U. Sorensen, MD, James W. Verbsky, MD, PhD, David I. Bernstein, MD, Joann Blessing-Moore, MD, David Lang, MD, Richard A. Nicklas, MD, John Oppenheimer, MD, Jay M. Portnoy, MD, Christopher R. Randolph, MD, Diane Schuller, MD, Sheldon L. Spector, MD, Stephen Tilles, MD, Dana Wallace, MD Chief Editor: Francisco A. Bonilla, MD, PhD Co-Editor: David A. Khan, MD Members of the Joint Task Force on Practice Parameters: David I. Bernstein, MD, Joann Blessing-Moore, MD, David Khan, MD, David Lang, MD, Richard A. Nicklas, MD, John Oppenheimer, MD, Jay M. Portnoy, MD, Christopher R. Randolph, MD, Diane Schuller, MD, Sheldon L. Spector, MD, Stephen Tilles, MD, Dana Wallace, MD Primary Immunodeficiency Workgroup: Chairman: Francisco A. Bonilla, MD, PhD Members: Zuhair K. Ballas, MD, Javier Chinen, MD, PhD, Michael M. Frank, MD, Joyce T. Hsu, MD, Michael Keller, MD, Lisa J. Kobrynski, MD, Hirsh D. Komarow, MD, Bruce Mazer, MD, Robert P. Nelson, Jr, MD, Jordan S. Orange, MD, PhD, John M. Routes, MD, William T. Shearer, MD, PhD, Ricardo U. Sorensen, MD, James W. Verbsky, MD, PhD GlaxoSmithKline, Merck, and Aerocrine; has received payment for lectures from Genentech/ These parameters were developed by the Joint Task Force on Practice Parameters, representing Novartis, GlaxoSmithKline, and Merck; and has received research support from Genentech/ the American Academy of Allergy, Asthma & Immunology; the American College of Novartis and Merck. -

Complement Herbert L
Host Defense 2011 Complement Herbert L. Mathews, Ph.D. COMPLEMENT Date: 4/11/11 Reading Assignment: Janeway’s Immunobiology, 7th Edition, pp. 54-55, 61-82, 406- 409, 514-515. Figures: (Unless otherwise noted) Janeway’s Immunobiology, 7th Edition, Murphy et al., Garland Publishing. KEY CONCEPTS AND LEARNING OBJECTIVES You will be able to describe the mechanism and consequences of the activation of the complement system. To attain the goals for these lectures you will be able to: a. List the components of the complement system. b. Describe the three activation pathways for complement. c. Explain the consequences of complement activation. d. Describe the consequence of complement deficiency. Page 1 Host Defense 2011 Complement Herbert L. Mathews, Ph.D. CONTENT SUMMARY Introduction Nomenclature Activation of Complement The classical pathway The mannan-binding lectin pathway The alternative pathway Biological Consequence of Complement Activation Cell lysis and viral neutralization Opsonization Clearance of Immune Complexes Inflammation Regulation of Complement Activation Human Complement Component Deficiencies Page 2 Host Defense 2011 Complement Herbert L. Mathews, Ph.D. Introduction The complement system is a group of more than 30 plasma and membrane proteins that play a critical role in host defense. When activated, complement components interact in a highly regulated fashion to generate products that: Recruit inflammatory cells (promoting inflammation). Opsonize microbial pathogens and immune complexes (facilitating antigen clearance). Kill microbial pathogens (via a lytic mechanism known as the membrane attack complex). Generate an inflammatory response. Complement activation takes place on antigenic surfaces. However, the activation of complement generates several soluble fragments that have important biologic activity. -

Ic3b Catalog Number: A115 Sizes Available: 250 Μg/Vial Concentration
Name: iC3b Catalog Number: A115 Sizes Available: 250 µg/vial Concentration: 1.0 mg/mL (see Certificate of Analysis for actual concentration) Form: Frozen liquid Purity: >90% by SDS-PAGE Buffer: 10 mM sodium phosphate, 145 mM NaCl, pH 7.2 Extinction Coeff. A280 nm = 1.03 at 1.0 mg/mL Molecular Weight: 176,000 Da (3 chains) Preservative: None, 0.22 µm filtered Storage: -70oC or below. Avoid freeze/thaw. Source: Normal human serum (shown by certified tests to be negative for HBsAg and for antibodies to HCV, HIV-1 and HIV-II). Precautions: Use normal precautions for handling human blood products. Origin: Manufactured in the USA. General Description iC3b (inactivated C3b) is derived from C3b. Conversion of C3b to iC3b destroys almost all of the functional binding sites present on C3b. C3b itself is produced by all three pathways of complement (Law, S.K.A. and Reid, K.B.M. (1995)) when native C3 is cleaved releasing C3a. iC3b is prepared by cleavage of C3b by factor I in the presence of factor H. Cleavage by factors H and I occurs rapidly when the C3b is free in solution and is slower when it is attached to a surface. Other cofactors for factor I also permit cleavage if C3b to iC3b and these include the two membrane proteins CR1 (CD35) and MCP (CD46). Factor I can cleave C3b in two places in the alpha chain and if both sites are cleaved a small fragment (C3f, 2,000 Da) is released. If the C3b precursor was attached to a surface, the iC3b remains on that surface. -

Antibody-Dependent Cellular Cytotoxicity in Primary Immunodeficiency Diseases and with Normal Leukocyte Subpopulations: IMPORTANCE of the TYPE of TARGET
Antibody-Dependent Cellular Cytotoxicity in Primary Immunodeficiency Diseases and with Normal Leukocyte Subpopulations: IMPORTANCE OF THE TYPE OF TARGET S. Ozden Sanal, Rebecca H. Buckley J Clin Invest. 1978;61(1):1-10. https://doi.org/10.1172/JCI108907. To gain insight into a possible role for antibody-dependent cell-mediated cytotoxicity in vivo, we examined the ability of leukocytes from 28 patients with primary immunodeficiency and from 20 normal controls to lyse three different types of antibody-coated targets in vitro. Mean cytotoxic indices ±1 SD elicited by unfractionated mononuclear cells from normal controls were 28.74±13.26 for human HLA antibody-coated lymphocyte targets, 42.79±8.27 for rabbit IgG antibody- coated chicken erythrocyte targets, and 47.58±10.34 for human anti-CD (Ripley)-coated O+ erythrocyte targets. Significantly (P=<0.05) lower than normal mean cytotoxic indices against lymphocyte targets were seen with effector cells from 10 patients with X-linked agammaglobulinemia (3.7±4.33), in 10 with common variable agammaglobulinemia (16.05±7.74), in 3 with immunodeficiency with hyper IgM (18.41±4.88), and in 2 with severe combined immunodeficiency (3.94±0.3). Antibody-dependent cytotoxicity against chicken erythrocytes was significantly (P=<0.05) lower than normal only in the common variable agammaglobulinemic group (33.33±12.3) and against human erythrocytes only in the common variable (34.36±9.59) and hyper IgM (27.54±0.66) groups. Rosette and anti-F(ab′)2 depletion studies with normal leukocytes indicated that a nonadherent, nonphagocytic, non-Ig-bearing, non-C receptor-bearing, Fc receptor- bearing lymphocyte was the only effector capable of lysing HLA antiboyd-coated lymphocyte targets. -

European Society for Immunodeficiencies (ESID)
Journal of Clinical Immunology https://doi.org/10.1007/s10875-020-00754-1 ORIGINAL ARTICLE European Society for Immunodeficiencies (ESID) and European Reference Network on Rare Primary Immunodeficiency, Autoinflammatory and Autoimmune Diseases (ERN RITA) Complement Guideline: Deficiencies, Diagnosis, and Management Nicholas Brodszki1 & Ashley Frazer-Abel2 & Anete S. Grumach3 & Michael Kirschfink4 & Jiri Litzman5 & Elena Perez6 & Mikko R. J. Seppänen7 & Kathleen E. Sullivan8 & Stephen Jolles9 Received: 5 June 2019 /Accepted: 20 January 2020 # The Author(s) 2020 Abstract This guideline aims to describe the complement system and the functions of the constituent pathways, with particular focus on primary immunodeficiencies (PIDs) and their diagnosis and management. The complement system is a crucial part of the innate immune system, with multiple membrane-bound and soluble components. There are three distinct enzymatic cascade pathways within the complement system, the classical, alternative and lectin pathways, which converge with the cleavage of central C3. Complement deficiencies account for ~5% of PIDs. The clinical consequences of inherited defects in the complement system are protean and include increased susceptibility to infection, autoimmune diseases (e.g., systemic lupus erythematosus), age-related macular degeneration, renal disorders (e.g., atypical hemolytic uremic syndrome) and angioedema. Modern complement analysis allows an in-depth insight into the functional and molecular basis of nearly all complement deficiencies. However, therapeutic options remain relatively limited for the majority of complement deficiencies with the exception of hereditary angioedema and inhibition of an overactivated complement system in regulation defects. Current management strategies for complement disor- ders associated with infection include education, family testing, vaccinations, antibiotics and emergency planning. Keywords Complement . -

Fracp Lecture 2010 Immune Deficiency 3
FRACP LECTURE 2010 IMMUNE DEFICIENCY 3 DR MARNIE ROBINSON PAEDIATRIC IMMUNOLOGIST/ALLERGIST IMMUNOLOGY LECTURE 3 • Neutrophil defects • INTERFERON –Y /IL‐12 pathway defect • Dysregulatory immune dfiideficienc ies NEUTROPHIL DEFECTS • Neutropaenia – Allo immune /au to immune – Kostmann, WHIM – cyclllical • Chronic granulomatous disease • Leukocyte adhesion deficiency • Neutrophil specific granule deficiency • Chediak –higashi syndrome AUTOIMMUNE NEUTROPAENIA • Antibodies against different neutrophil antigen • Aeitiology unknown • Slightly more common in females • Present with skin and upper respiratory tract infections (pneumonia /menin gitis /se psis less common) • Neutrophil count usually <0. 5 but may increase during infection • Treatment with G‐CSF (IVIG) • Usually remits spontaneously by <24 months ALLOIMMUNE NEUTROPAENIA • Caused by transplacental transfer of maternal against the FcyRIIIb isotypes of NA 1 and NA2 causing immune destruction of neutrophils • Incidence of 1/500 • Usually presents in first weeks of life • Present with omphalitis, cellulitis, pneumonia • Diagnosed by detection of neutrophil specific alloantibodies in maternal blood • Treat with G‐CSF • resolves with waning of maternal antibodies KOSTMANN’S SYNDROME • Bone marrow granulocyte arrest at promyeolocyte or myelocyte stage • Present early in life (usually <6 months) • Present with omphalitis , respiratory tract ifinfect ions, skin and liver abscesses • Increased susceptibility to AML • Treatment is with G‐CSF CYCLICAL NEUTROPAENIA • Defect in elastase 2 • Sporadic