Japanese Guidelines for Adult Asthma 2020*
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
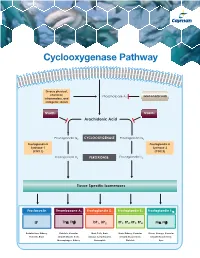
Cyclooxygenase Pathway
Cyclooxygenase Pathway Diverse physical, chemical, Phospholipase A Glucocorticoids inflammatory, and 2 mitogenic stimuli NSAIDs NSAIDs Arachidonic Acid Prostaglandin G2 CYCLOOXYGENASE Prostaglandin G2 Prostaglandin H Prostaglandin H Synthase-1 Synthase-2 (COX 1) (COX 2) Prostaglandin H2 PEROXIDASE Prostaglandin H2 Tissue Specific Isomerases Prostacyclin Thromboxane A2 Prostaglandin D2 Prostaglandin E2 Prostaglandin F2α IP TPα, TPβ DP1, DP2 EP1, EP2, EP3, EP4 FPα, FPβ Endothelium, Kidney, Platelets, Vascular Mast Cells, Brain, Brain, Kidney, Vascular Uterus, Airways, Vascular Platelets, Brain Smooth Muscle Cells, Airways, Lymphocytes, Smooth Muscle Cells, Smooth Muscle Cells, Macrophages, Kidney Eosinophils Platelets Eyes Prostacyclin Item No. Product Features Prostacyclin (Prostaglandin I2; PGI2) is formed from arachidonic acid primarily in the vascular endothelium and renal cortex by sequential 515211 6-keto • Sample Types: Culture Medium | Plasma Prostaglandin • Measure 6-keto PGF levels down to 6 pg/ml activities of COX and prostacyclin synthase. PGI2 is non-enzymatically 1α F ELISA Kit • Incubation : 18 hours | Development: 90-120 minutes | hydrated to 6-keto PGF1α (t½ = 2-3 minutes), and then quickly converted 1α Read: Colorimetric at 405-420 nm to the major metabolite, 2,3-dinor-6-keto PGF1α (t½= 30 minutes). Prostacyclin was once thought to be a circulating hormone that regulated • Assay 24 samples in triplicate or 36 samples in duplicate platelet-vasculature interactions, but the rate of secretion into circulation • NOTE: A portion of urinary 6-keto PGF1α is of renal origin coupled with the short half-life indicate that prostacyclin functions • NOTE : It has been found that normal plasma levels of 6-keto PGF may be low locally. -

Role of Thromboxane Receptor-Alpha in Prostate Cancer Progression Prasanna Ekambaram Wayne State University
Wayne State University Wayne State University Dissertations 1-1-2012 Role Of Thromboxane Receptor-Alpha In Prostate Cancer Progression Prasanna Ekambaram Wayne State University, Follow this and additional works at: http://digitalcommons.wayne.edu/oa_dissertations Recommended Citation Ekambaram, Prasanna, "Role Of Thromboxane Receptor-Alpha In Prostate Cancer Progression" (2012). Wayne State University Dissertations. Paper 591. This Open Access Dissertation is brought to you for free and open access by DigitalCommons@WayneState. It has been accepted for inclusion in Wayne State University Dissertations by an authorized administrator of DigitalCommons@WayneState. ROLE OF THROMBOXANE RECEPTOR-ALPHA IN PROSTATE CANCER PROGRESSION by PRASANNA EKAMBARAM DISSERTATION Submitted to the Graduate School of Wayne State University, Detroit, Michigan in partial fulfillment of the requirements for the degree of DOCTOR OF PHILOSOPHY 2012 MAJOR: CANCER BIOLOGY Approved by: ______________________________ Advisor Date ______________________________ ______________________________ ______________________________ ______________________________ © COPYRIGHT BY PRASANNA EKAMBARAM 2012 All rights reserved DEDICATION I would like to dedicate this dissertation to my wife, Arulselvi and my kids Tejas and Abhi for standing strong by my side at good times and difficult times in life and helping me work hard toward achieving my goal of being a research scientist; To my parents for their blessings, and prayers and for helping to come to Unites States to complete my graduate studies. ii ACKNOWLEDGMENTS Foremost, I would like to extend my gratitude to my mentor, Dr. Honn for giving me this wonderful opportunity to work in his laboratory and to be my mentor for my PhD program. I would like to thank him for his immense patience, the support and guidance through the years. -

Improved Homology Modeling of the Human & Rat EP4 Prostanoid Receptors
Holt et al. BMC Molecular and Cell Biology (2019) 20:37 BMC Molecular and https://doi.org/10.1186/s12860-019-0212-5 Cell Biology RESEARCHARTICLE Open Access Improved homology modeling of the human & rat EP4 prostanoid receptors Melissa C. Holt, Chi S. Ho, M. Inés Morano, Stephen D. Barrett and Adam J. Stein* Abstract Background: The EP4 prostanoid receptor is one of four GPCRs that mediate the diverse actions of prostaglandin E2 (PGE2). Novel selective EP4 receptor agonists would assist to further elucidate receptor sub-type function and promote development of therapeutics for bone healing, heart failure, and other receptor associated conditions. The rat EP4 (rEP4) receptor has been used as a surrogate for the human EP4 (hEP4) receptor in multiple SAR studies. To better understand the validity of this traditional approach, homology models were generated by threading for both receptors using the RaptorX server. These models were fit to an implicit membrane using the PPM server and OPM database with refinement of intra and extracellular loops by Prime (Schrödinger). To understand the interaction between the receptors and known agonists, induced-fit docking experiments were performed using Glide and Prime (Schrödinger), with both endogenous agonists and receptor sub-type selective, small-molecule agonists. The docking scores and observed interactions were compared with radioligand displacement experiments and receptor (rat & human) activation assays monitoring cAMP. Results: Rank-ordering of in silico compound docking scores aligned well with in vitro activity assay EC50 and radioligand binding Ki. We observed variations between rat and human EP4 binding pockets that have implications in future small-molecule receptor-modulator design and SAR, specifically a S103G mutation within the rEP4 receptor. -

Q4 2019/20 Initiation
Duration Duration Duration between Research Integrate between between First Date Site Ethics d Date of Date Site Date Site Reasons Participan Confirme HRA Date Site Non- Date Site Committe Research Name of First Selected Selected Date Site Date Site Date Site for delay t d and Approval Confirmed Confirmat Ready To e Applicatio Trial Participant and Date and First Invited Selected Confirmed correspon Recruited First Date By Sponsor ion Status Start Reference n System Recruited Site Participan d to: ? Participan Number Number Confirme t t d Recruited Recruited VIVOTM non- invasive mapping 19/LO/03 Please Please 162140 257755 of Yes 03/07/2019 26 33 59 01/03/2019 05/05/2019 21/04/2019 15/05/2019 31/05/2019 31/05/2019 08 Select... Select... ventricula r arrhythmi a A Pilot Study to exPlOre the exisTence and impact of 19/LO/07 FRAILTY Please Please 162141 264744 Yes 20/08/2019 31 11 42 01/04/2019 09/07/2019 16/07/2019 24/07/2019 09/08/2019 09/08/2019 84 in Select... Select... patients over the age of 70 undergoin g cardiac interventi ons Duration Duration Duration between Research Integrate between between First Date Site Ethics d Date of Date Site Date Site Reasons Participan Confirme HRA Date Site Non- Date Site Committe Research Name of First Selected Selected Date Site Date Site Date Site for delay t d and Approval Confirmed Confirmat Ready To e Applicatio Trial Participant and Date and First Invited Selected Confirmed correspon Recruited First Date By Sponsor ion Status Start Reference n System Recruited Site Participan d to: ? Participan Number Number Confirme t t d Recruited Recruited "Pressure- controlled Intermitte nt Coronary Sinus 19/SC/00 Occlusion Please Please 162142 259683 Yes 23/09/2019 40 12 52 01/04/2019 02/08/2019 16/07/2019 14/08/2019 11/09/2019 16/09/2019 50 (PiCSO) in Select.. -

Pharmaceutical Sciences
IAJPS 2018, 05 (01), 33-36 Naveen Kumar G.S et al ISSN 2349-7750 CODEN [USA]: IAJPBB ISSN: 2349-7750 INDO AMERICAN JOURNAL OF PHARMACEUTICAL SCIENCES Available online at: http://www.iajps.com Research Article DEVELOPMENT OF UV SPECTROPHOTOMETRIC METHOD FOR ESTIMATION OF SERATRODAST IN BULK AND PHARMACEUTICAL FORMULATIONS Naveen Kumar G S*, Harish K H, Hanumanthachar Joshi Sarada Vilas College of Pharmacy, K. M. Puram, Mysuru, India. Abstract: A simple UV spectrophotometric method has been developed for the estimation of Seratrodast pure form and pharmaceutical formulation. Seratrodast in bulk drug and pharmaceutical formulation and has an absorption maximum at 285nm in methanol. It obeys Beer’s law in the concentration range of 20 -100 µg/ml. The method was measured at its appropriate λmax against the reagent blank. The developed method was found to be precise, accurate and reproducible. Keywords: Beer’s law, Seratrodast, methanol and UV spectrophotometry *Corresponding author: Naveen Kumar G.S, QR code Sarada Vilas College of Pharmacy, K. M. Puram, Mysuru, India Email: [email protected], Please cite this article in press as Naveen Kumar G.S et al., Development of UV Spectrophotometric Method for Estimation of Seratrodastin Bulk and Pharmaceutical Formulations, Indo Am. J. P. Sci, 2018; 05(01). www.iajps.com Page 33 IAJPS 2018, 05 (01), 33-36 Naveen Kumar G.S et al ISSN 2349-7750 INTRODUCTION: Experimental methods Seratrodast is a thromboxane A2 (TXA2) receptor Preparation of Standard stock solution (TP receptor) antagonist used primarily in the Standard solution of seratrodast (1 mg/ml) was treatment of asthma. -

Patent Application Publication ( 10 ) Pub . No . : US 2019 / 0192440 A1
US 20190192440A1 (19 ) United States (12 ) Patent Application Publication ( 10) Pub . No. : US 2019 /0192440 A1 LI (43 ) Pub . Date : Jun . 27 , 2019 ( 54 ) ORAL DRUG DOSAGE FORM COMPRISING Publication Classification DRUG IN THE FORM OF NANOPARTICLES (51 ) Int . CI. A61K 9 / 20 (2006 .01 ) ( 71 ) Applicant: Triastek , Inc. , Nanjing ( CN ) A61K 9 /00 ( 2006 . 01) A61K 31/ 192 ( 2006 .01 ) (72 ) Inventor : Xiaoling LI , Dublin , CA (US ) A61K 9 / 24 ( 2006 .01 ) ( 52 ) U . S . CI. ( 21 ) Appl. No. : 16 /289 ,499 CPC . .. .. A61K 9 /2031 (2013 . 01 ) ; A61K 9 /0065 ( 22 ) Filed : Feb . 28 , 2019 (2013 .01 ) ; A61K 9 / 209 ( 2013 .01 ) ; A61K 9 /2027 ( 2013 .01 ) ; A61K 31/ 192 ( 2013. 01 ) ; Related U . S . Application Data A61K 9 /2072 ( 2013 .01 ) (63 ) Continuation of application No. 16 /028 ,305 , filed on Jul. 5 , 2018 , now Pat . No . 10 , 258 ,575 , which is a (57 ) ABSTRACT continuation of application No . 15 / 173 ,596 , filed on The present disclosure provides a stable solid pharmaceuti Jun . 3 , 2016 . cal dosage form for oral administration . The dosage form (60 ) Provisional application No . 62 /313 ,092 , filed on Mar. includes a substrate that forms at least one compartment and 24 , 2016 , provisional application No . 62 / 296 , 087 , a drug content loaded into the compartment. The dosage filed on Feb . 17 , 2016 , provisional application No . form is so designed that the active pharmaceutical ingredient 62 / 170, 645 , filed on Jun . 3 , 2015 . of the drug content is released in a controlled manner. Patent Application Publication Jun . 27 , 2019 Sheet 1 of 20 US 2019 /0192440 A1 FIG . -

Investigational Allergology and Clinical Immunology
Journal of Investigational Allergology and Clinical Immunology ISSN 1018-9068 Volume 31, Supplement 1, 2021 Official Organ of Spanish Society of Allergology and Clinical Immunology www.jiaci.org GEMA5.0 SPANISH GUIDELINE ON THE MANAGEMENT OF ASTHMA Journal of Investigational Allergology and Clinical Immunology Volume 31, Suppl. 1, 2021 Official Organ of Spanish Society of Allergology and Clinical Immunology Editors in Chief A.G. Oehling, Servicio de Alergología, Clínica Rotger, C/ Santiago Rusiñol 3, E-07012 Palma de Mallorca, Spain (E-mail [email protected]) J.M. Olaguibel, Unidad de Asma Grave, Servicio de Alergología, Complejo Hospitalario de Navarra, C/Irunlarrea s/n, E-31008 Pamplona, Spain (Tel. +34 948 255-400, Fax +34 948 296-500, E-mail [email protected]) Associate Editors I. Dávila, Hospital Clínico Universitario, Paseo San Vicente s/n, E-37007 Salamanca, Spain P.M. Gamboa, Servicio de Alergología, Hospital de Cruces, Plaza de Cruces, s/n, E-48903 Baracaldo, Bizkaia, Spain R. Lockey, University of South Florida College of Medicine, Division of Allergy and Immunology, VA Medical Center, 13000 North 30th Street, Tampa, FL 33612, USA V. del Pozo, Senior Research, Immunology IIS-FJD, Avda. Reyes Católicos 2, E-28040 Madrid, Spain J. Sastre, Servicio de Alergia, Fundación Jiménez Díaz, Avda. Reyes Católicos 2, E-28040 Madrid, Spain J.M. Zubeldia, Servicio de Alergología, Hospital G.U. Gregorio Marañón, C/ Dr. Esquerdo 46, E-28007 Madrid, Spain Founding Editor A.K. Oehling †, Department of Allergology and Clinical Immunology, Clínica Universidad de Navarra, Apartado 4209, E-31008 Pamplona, Spain Editorial Assistant G. Betelu, Department of Allergology and Clinical Immunology, Clínica Universidad de Navarra, Apartado 4209, E-31008 Pamplona, Spain (Tel. -

PDCO Minutes of the 20-23 February 2018 Meeting
23 February 2018 EMA/PDCO/113780/2018 Inspections, Human Medicines Pharmacovigilance and Committees Division Paediatric Committee (PDCO) Minutes of the meeting on 20-23 February 2018 Chair: Dirk Mentzer – Vice-Chair: Koenraad Norga 20 February 2018, 14:00- 17:00, room 3A 21 February 2018, 08:30- 19:00, room 3A 22 February 2018, 08:30- 19:00, room 3A 23 February 2018, 08:30- 13:00, room 3A Disclaimers Some of the information contained in this set of minutes is considered commercially confidential or sensitive and therefore not disclosed. With regard to intended therapeutic indications or procedure scopes listed against products, it must be noted that these may not reflect the full wording proposed by applicants and may also vary during the course of the review. Additional details on some of these procedures will be published in the PDCO Committee meeting reports (after the PDCO Opinion is adopted), and on the Opinions and decisions on paediatric investigation plans webpage (after the EMA Decision is issued). Of note, this set of minutes is a working document primarily designed for PDCO members and the work the Committee undertakes. Further information with relevant explanatory notes can be found at the end of this document. Note on access to documents Some documents mentioned in the minutes cannot be released at present following a request for access to documents within the framework of Regulation (EC) No 1049/2001 as they are subject to on-going procedures for which a final decision has not yet been adopted. They will become public when adopted or considered public according to the principles stated in the Agency policy on access to documents (EMA/127362/2006). -

Structures of the Human PGD2 Receptor CRTH2 Reveal Novel Mechanisms for Ligand Recognition
Article Structures of the Human PGD2 Receptor CRTH2 Reveal Novel Mechanisms for Ligand Recognition Graphical Abstract Authors Lei Wang, Dandan Yao, R.N.V. Krishna Deepak, ..., Weimin Gong, Zhiyi Wei, Cheng Zhang Correspondence [email protected] (Z.W.), [email protected] (C.Z.) In Brief Wang et al. reported crystal structures of antagonist-bound human CRTH2 as a new asthma drug target. Chemically diverse antagonists occupy a similar semi-occluded pocket with distinct binding modes. Structural analysis suggests a potential ligand entry port and an opposite charge attraction-facilitated binding process for the endogenous CRTH2 ligand prostaglandin D2. Highlights d Crystal structures of antagonist-bound human CRTH2 are solved d A well-structured N terminus covers ligand binding pocket d Conserved and divergent binding features of CRTH2 antagonists are revealed d A multiple-step binding process of prostaglandin D2 is proposed Wang et al., 2018, Molecular Cell 72, 48–59 October 4, 2018 ª 2018 Elsevier Inc. https://doi.org/10.1016/j.molcel.2018.08.009 Molecular Cell Article Structures of the Human PGD2 Receptor CRTH2 Reveal Novel Mechanisms for Ligand Recognition Lei Wang,1,7 Dandan Yao,2,3,7 R.N.V. Krishna Deepak,4 Heng Liu,1 Qingpin Xiao,1,5 Hao Fan,4 Weimin Gong,2,6 Zhiyi Wei,5,* and Cheng Zhang1,8,* 1Department of Pharmacology and Chemical Biology, School of Medicine, University of Pittsburgh, Pittsburgh, PA 15261, USA 2Key Laboratory of RNA Biology, Institute of Biophysics, Chinese Academy of Sciences, Beijing 100101, China 3University -

Mechanisms of Cyclooxygenase-2-Dependent Human Aortic Smooth Muscle Cell Phenotypic Modulation
University of Kentucky UKnowledge Theses and Dissertations--Pharmacy College of Pharmacy 2014 MECHANISMS OF CYCLOOXYGENASE-2-DEPENDENT HUMAN AORTIC SMOOTH MUSCLE CELL PHENOTYPIC MODULATION Oreoluwa O. Adedoyin University of Kentucky, [email protected] Right click to open a feedback form in a new tab to let us know how this document benefits ou.y Recommended Citation Adedoyin, Oreoluwa O., "MECHANISMS OF CYCLOOXYGENASE-2-DEPENDENT HUMAN AORTIC SMOOTH MUSCLE CELL PHENOTYPIC MODULATION" (2014). Theses and Dissertations--Pharmacy. 34. https://uknowledge.uky.edu/pharmacy_etds/34 This Doctoral Dissertation is brought to you for free and open access by the College of Pharmacy at UKnowledge. It has been accepted for inclusion in Theses and Dissertations--Pharmacy by an authorized administrator of UKnowledge. For more information, please contact [email protected]. STUDENT AGREEMENT: I represent that my thesis or dissertation and abstract are my original work. Proper attribution has been given to all outside sources. I understand that I am solely responsible for obtaining any needed copyright permissions. I have obtained needed written permission statement(s) from the owner(s) of each third-party copyrighted matter to be included in my work, allowing electronic distribution (if such use is not permitted by the fair use doctrine) which will be submitted to UKnowledge as Additional File. I hereby grant to The University of Kentucky and its agents the irrevocable, non-exclusive, and royalty-free license to archive and make accessible my work in whole or in part in all forms of media, now or hereafter known. I agree that the document mentioned above may be made available immediately for worldwide access unless an embargo applies. -

(CRTH2) Antagonists in Asthma: a Systematic Review and Meta-Analysis Protocol AUTHORS Liu, Wei; Min, Jie; Jiang, Hongli; Mao, Bing
BMJ Open: first published as 10.1136/bmjopen-2017-020882 on 20 April 2018. Downloaded from PEER REVIEW HISTORY BMJ Open publishes all reviews undertaken for accepted manuscripts. Reviewers are asked to complete a checklist review form (http://bmjopen.bmj.com/site/about/resources/checklist.pdf) and are provided with free text boxes to elaborate on their assessment. These free text comments are reproduced below. ARTICLE DETAILS TITLE (PROVISIONAL) Chemoattractant Receptor-Homologous Molecule Expressed on Th2 Cells (CRTH2) antagonists in asthma: a systematic review and meta-analysis protocol AUTHORS liu, wei; Min, Jie; Jiang, Hongli; Mao, Bing VERSION 1 – REVIEW REVIEWER Hugo Farne Imperial College London United Kingdom REVIEW RETURNED 05-Jan-2018 GENERAL COMMENTS This is an interesting proposal as the findings have indeed been contradictory. My major comment is about the language - this paper would benefit significantly from proof-reading by a native English speaker (e.g. first para page 8). http://bmjopen.bmj.com/ Other comments: Strengths & Limitations (p5): - As there are no head-to-head trials of CRTH2 antagonists, subgroup analyses cannot directly assess the efficacy of CRTH2 antagonists relative to each other - By having no cut-off in terms of minimum duration of treatment (e.g. 12 weeks or 16 weeks), the study may include trials on September 28, 2021 by guest. Protected copyright. that were underpowered, which in turn may dilute any effect – I would note this. Introduction (p7) - CRTH2 is also expressed on Type 2 Innate Lymphoid Cells (ILC2s) - CRTH2 is not expressed on mast cells (except intracellularly, with unknown function) so production of cytokines from mast cells is not a consequence of PGD2 binding to CRTH2 on mast cells - The rationale for the CRTH2 receptor as a drug target in asthma is not explained fully. -

Fevipiprant (QAW039), a Slowly Dissociating Crth2 Antagonist with the Potential for Improved Clinical Efficacy S
Supplemental material to this article can be found at: http://molpharm.aspetjournals.org/content/suppl/2016/02/25/mol.115.101832.DC1 1521-0111/89/5/593–605$25.00 http://dx.doi.org/10.1124/mol.115.101832 MOLECULAR PHARMACOLOGY Mol Pharmacol 89:593–605, May 2016 Copyright ª 2016 by The American Society for Pharmacology and Experimental Therapeutics Fevipiprant (QAW039), a Slowly Dissociating CRTh2 Antagonist with the Potential for Improved Clinical Efficacy s David A. Sykes, Michelle E. Bradley, Darren M. Riddy, Elizabeth Willard, John Reilly, Asadh Miah, Carsten Bauer, Simon J. Watson, David A. Sandham, Gerald Dubois, and Steven J. Charlton Novartis Institutes for Biomedical Research, Horsham, West Sussex, UK (D.A.S., M.E.B., D.M.R., E.W., J.R., A.M., S.W., D.A.S., G.D., S.J.C.); Novartis Institutes for Biomedical Research, Cambridge, Massachusetts (D.A.Sa., J.R.); Novartis Institutes for Biomedical Research, Basel, Switzerland (C.B.); School of Life Sciences, Queen’s Medical Centre, University of Nottingham, Downloaded from Nottingham, UK (D.A.Sy., S.J.C.) Received September 22, 2015; accepted February 22, 2016 ABSTRACT Here we describe the pharmacologic properties of a series of reversed by increasing concentrations of PGD2 after an initial molpharm.aspetjournals.org clinically relevant chemoattractant receptor-homologous mole- 15-minute incubation period. This behavior is consistent with cules expressed on T-helper type 2 (CRTh2) receptor antago- its relatively slow dissociation from the human CRTh2 receptor. nists, including fevipiprant (NVP-QAW039 or QAW039), which is In contrast for the other ligands tested this time-dependent currently in development for the treatment of allergic diseases.