R.C. Systems PID Response Factors
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Optrel-Filter-Guide.Pdf
Guide to optrel filters This information is intended to provide an overview of the relevant factors that will help in the selection of a suitable fil- ter. If you lack the necessary information and/or are not sure about your choice, consult a professional first. Only with the right choice you can protect yourself from the health risks caused by pollutants in the ambient air. 1. information you need for a correct choose: • Pollutants in the ambient air • Concentration(s) of the pollutant(s) • Aggregate state(s) of the pollutant(s) (gaseous, solid, as a mixture) • Can the pollutants be detected without aids? (e.g. odor or taste) • The valid limit values e.g. AGW, OEL, ... • Oxygen content of the ambient air. There are locally different minimum concentrations apply (Germany at least 17 vol. %) • Should respiratory protection be combined with other be combined? (head, eye or hearing protection) 3. selection of the suitable filter device With the information from chapter 2, the necessary protection factor can be determined. The following chart shows the protection factor of the optrel respiratory protection equipment is listed: 2 optrel Filter Guide Device Marking Nominal protection factor Particle filtering devices e3000X in combination with TH3P 500 PAPR helmet panoramaxx se- ries, sphere series, clearmaxx series swiss air TH3P 500 e3000X in combination with TH2P 50 PAPR clearmaxx und industri- al hard hat e3000X in combination with TH1P 10 PAPR panoramaxx series and industrial hard hat Chart 1 with protection factors of optrel equipment (Germany) The NPF is derived from the maximum permissible leakage of the respective equipment. -

Analysis of Trace Hydrocarbon Impurities in 1,3-Butadiene Using Optimized Rt®-Alumina BOND/MAPD PLOT Columns by Rick Morehead, Jan Pijpelink, Jaap De Zeeuw, Tom Vezza
Petroleum & Petrochemical Applications Analysis of Trace Hydrocarbon Impurities in 1,3-Butadiene Using Optimized Rt®-Alumina BOND/MAPD PLOT Columns By Rick Morehead, Jan Pijpelink, Jaap de Zeeuw, Tom Vezza Abstract Identifying and quantifying trace impurities in 1,3-butadiene is critical in producing high quality synthetic rubber products. Stan- dard analytical methods employ alumina PLOT columns which yield good resolution for low molecular weight hydrocarbons, but suffer from irreproducibility and poor sensitivity for polar hydrocarbons. In this study, Rt®-Alumina BOND/MAPD PLOT columns were used to separate both common light polar contaminants, including methyl acetylene and propadiene, as well as 4-vinylcy- clohexene, which is a high molecular weight impurity that normally requires a second test on an alternative column. By using an extended temperature program that employs the full thermal range of the column, 4-vinylcyclohexene, as well as all of the typical low molecular weight impurities in 1,3-butadiene, can be analyzed in a single test. Introduction 1,3-butadiene is typically isolated from products of the naphtha steam cracking process. Prior to purification, 1,3-butadiene can be contaminated with significant amounts of isobutene as well as other C4 isomers. In addition to removing these C4 isomeric contaminants during purification, it is also important that 1,3-butadiene be free of propadiene and methyl acetylene, which can interfere with catalytic polymerization. Alumina PLOT columns are the most commonly used GC column for this application, but the determination of polar hydrocarbon impurities at trace levels can be quite challenging and is highly dependent on the deactiva- tion of the alumina surface. -

A Comprehensive Investigation on the Formation of Organo-Sulfur Molecules in Dark Clouds Via Neutral-Neutral Reactions
A&A 395, 1031–1044 (2002) Astronomy DOI: 10.1051/0004-6361:20021328 & c ESO 2002 Astrophysics A comprehensive investigation on the formation of organo-sulfur molecules in dark clouds via neutral-neutral reactions M. Yamada1, Y. Osamura1, and R. I. Kaiser2;? 1 Department of Chemistry, Rikkyo University, 3-34-1 Nishi-ikebukuro, Tokyo 171-8501, Japan 2 Department of Chemistry, University of York, YO10 5DD, UK Received 11 June 2002 / Accepted 9 September 2002 Abstract. Fifteen reactions on the doublet HCnS and singlet/triplet H2CnS(n = 1; 2) potential energy surfaces have been investi- gated theoretically via electronic structure calculations to unravel the formation of hydrogen deficient, sulfur bearing molecules via bimolecular reactions of two neutral species in cold molecular clouds. Various barrierless reaction pathways to synthe- size astronomically observed CnS and hitherto undetected HCnS(n = 1; 2) isomers are offered. These data present important 2 2 guidance to search for rotational transitions of the elusive thioformyl HCS(X A0) and thioketenyl HCCS(X Π) radicals in the interstellar medium. Key words. astrochemistry – ISM: molecules – molecular processes – methods: miscellaneous 1. Introduction terminated counterparts HCnSandCnSH have not been de- tected in the interstellar medium yet. However, these molecules Untangling the synthetic routes to form sulfur-carrying might represent the missing source of molecular-bound sul- molecules in the interstellar medium is a significant instru- fur in the interstellar medium. Hydrogen deficient organosul- ment to understand the chemical evolution and physical con- 2 2 fur molecules like HCS(X A0) and HCCS(X Π) resemble also ditions in star forming regions and in circumstellar envelopes important reaction intermediates which could lead to sulfur (Minh & van Dishoeck 2000). -

Next Generation PLOT Alumina Technology for Accurate Measurement of Trace Polar Hydrocarbons in Hydrocarbon Streams
Next generation PLOT alumina technology for accurate measurement of trace polar hydrocarbons in hydrocarbon streams Jaap de Zeeuw, Tom Vezza, Bill Bromps, Rick Morehead and Gary Stidsen Restek Corporation, Bellefonte, USA 1 Next generation PLOT alumina technology for accurate measurement of trace polar hydrocarbons in hydrocarbon streams Jaap de Zeeuw, Tom Vezza, Bill Bromps, Rick More head and Gary Stidsen Restek Corporation Summary In light hydrocarbon analysis, the separation and detection of traces of polar hydrocarbons like acetylene, propadiene and methyl acetylene is very important. Using commercial alumina columns with KCl or Na2SO4 deactivation, often results in low response of polar hydrocarbons. Additionally, challenges are observed in response-in time stability. Solutions have been proposed to maximize response for components like methyl acetylene and propadiene, using alumina columns that were specially deactivated. Operating such columns showed still several challenges: Due to different deactivations, the retention and loadability of such alumina columns has been drastically reduced. A new Alumina deactivation technology was developed that combined the high response for polar hydrocarbon with maintaining the loadability. This allows the high response for components like methyl acetylene, acetylene and propadiene, also to be used for impurity analysis as well as TCD type applications. Such columns also showed excellent stability of response in time, which was superior then existing solutions. Additionally, it was observed that such alumina columns could be used up to 250 C, extending the Tmax by 50C. This allows higher hydrocarbon elution, faster stabilization and also widens application scope of any GC where multiple columns are used. In this poster the data is presented showing the improvements made in this important application field. -

Proposed 15-Day Modifications
INFORMAL PRELIMINARY REVIEW DRAFT Proposed 15-Day Modifications ATTACHMENT A PROPOSED 15-DAY MODIFICATIONS California Code of Regulations, Title 17, Division 3, Chapter 1, Subchapter 7.7, Article 1 Note: This document shows proposed modifications to the originally proposed amendments to the Regulation for the Reporting of Criteria Air Pollutants and Toxic Air Contaminants, as presented during the November 19, 2020, meeting of the California Air Resources Board. At that meeting, the Board directed staff to make modifications to the proposed amendments based on public comments received, and to provide these updates for public comment for a period of at least 15 days. The pre-existing regulation text is set forth below in normal type. The original proposed amendments are shown in underline formatting to indicate additions and strikeout to indicate deletions. The additional proposed modifications made available with the notice of public availability of modified text dated March XY, 2021, are shown in double- underline to indicate additions and double-strikeout to indicate deletions. The symbol “***” means that intervening text not proposed for amendment is not shown. PROPOSED AMENDMENTS TO THE REGULATION FOR THE REPORTING OF CRITERIA AIR POLLUTANTS AND TOXIC AIR CONTAMINANTS California Code of Regulations, Title 17, Division 3, Chapter 1, Subchapter 7.7, Articles 1 and 2 Amend Subchapter 7.7, Article 1, and sections 93400, 93401, 93402, 93403, 93404, 93405, 93406, 93407, 93408, 93409, 93410, title 17, California Code of Regulations, and adopt new Subchapter 7.7, Article 2, sections 93420, 93421, and new Subchapter 7.7, Article 2, Appendices A and B to title 17, California Code of Regulations, to read as follows: CARB / AQPSD 1 2/11/2021 INFORMAL PRELIMINARY REVIEW DRAFT Proposed 15-Day Modifications Subchapter 7.7: Regulation for the Reporting of Criteria Air Pollutants and Toxic Air Contaminants Article 1. -

Effect of Rutin on Gentamicin-Induced Ototoxicity in Rats: a Biochemical and Histopathological Examination
ORIGINAL ARTICLE ENT UPDATES 11(1):8-13 DOI: 10.5152/entupdates.2021.887158 Effect of Rutin on Gentamicin-Induced Ototoxicity in Rats: A Biochemical and Histopathological Examination Abstract Objective: Gentamicin is a broad-spectrum aminoglycoside antibiotic administered parenterally for moderate to severe gram-negative infections. Ototoxicity is an im- portant side effect that limits gentamicin use. The aim of this study is to investigate the effect of rutin on gentamicin-induced ototoxicity in rats biochemically and histo- pathologically. Methods: Distilled water was administered by oral gavage to healthy controls (HG) and cobalt administered group (GC). 50 mg/kg rutin was administered by oral gavage to rutin + gentamicin (RGG) group. After one hour, 100 mg/kg gentamicin was injected intraperitoneally (i.p) to the RGG and GC animal groups. This procedure was repeated once a day for 14 days. Results: Malondialdehyde (MDA), nuclear factor-κB(NF-κB), tumor necrosis factor alpha (TNF-α) and interleukin-1 beta(IL-1β) levels in the cochlear nerve tissue of gen- tamicin-treated animals were significantly higher compared to healthy controls and rutin + gentamicin treated rats. On the other hand, the amount of total Glutathione (tGSH) was significantly lower compared to the control and rutin group. Histopatho- logical examination revealed degenerated myelinated nerve fibers in the gentamicin group and Schwann cell nuclei were generally not seen. There was a high accumula- tion of collagen fiber in the tissue and dilated blood capillaries. In the rutin group, my- elinated nerve fibers mostly exhibited normal morphology, Schwann cell nuclei were İsmail Salcan1 evident and the vessels were normal. -

Isobutylene Polymer Having Unsaturated Group and Preparation
Europaisches Patentamt European Patent Office Office europeen des brevets (11) EP 0 452 875 B2 (12) NEW EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) |nt CI.6: C08F 10/10, C08F 2/42 of the opposition decision: 18.08.1999 Bulletin 1999/33 (45) Mention of the grant of the patent: 09.08.1995 Bulletin 1995/32 (21) Application number: 91106042.4 (22) Date of filing: 16.04.1991 (54) Isobutylene polymer having unsaturated group and preparation thereof Isobutylenpolymer mit ungesattigter Gruppe und seine Herstellung Polymere de I'isobutylene ayant un groupe insature et sa preparation (84) Designated Contracting States: (74) Representative: BE CH DE ES FR GB IT LI NL SE Hansen, Bernd, Dr. Dipl.-Chem. et al Hoffmann Eitle, (30) Priority: 16.04.1990 JP 10102990 Patent- und Rechtsanwalte, 29.11.1990 JP 33636490 Postfach 81 04 20 81904 Miinchen (DE) (43) Date of publication of application: 23.10.1991 Bulletin 1991/43 (56) References cited: EP-A- 0 265 053 EP-B- 0 322 241 (73) Proprietor: Kanegafuchi Chemical Industry Co., CA-A- 467 049 GB-A- 1 059 580 Ltd. US-A- 4 31 6 973 US-A- 4 327 201 Kita-ku Osaka-shi Osaka-fu (JP) US- A- 4 524 1 88 US-A- 4 758 63 1 US-A- 4 929 683 (72) Inventors: • Noda, Koji Remarks: Kobe-shi, Hyogo-ken (JP) The file contains technical information submitted • Fujisawa, Hiroshi after the application was filed and not included in this Kobe-shi, Hyogo-ken (JP) specification • Yonezawa, Kazuya Kobe-shi, Hyogo-ken (JP) CM DO 10 Is- 00 CM IO ^- o a. -

Aldrich Raman
Aldrich Raman Library Listing – 14,033 spectra This library represents the most comprehensive collection of FT-Raman spectral references available. It contains many common chemicals found in the Aldrich Handbook of Fine Chemicals. To create the Aldrich Raman Condensed Phase Library, 14,033 compounds found in the Aldrich Collection of FT-IR Spectra Edition II Library were excited with an Nd:YVO4 laser (1064 nm) using laser powers between 400 - 600 mW, measured at the sample. A Thermo FT-Raman spectrometer (with a Ge detector) was used to collect the Raman spectra. The spectra were saved in Raman Shift format. Aldrich Raman Index Compound Name Index Compound Name 4803 ((1R)-(ENDO,ANTI))-(+)-3- 4246 (+)-3-ISOPROPYL-7A- BROMOCAMPHOR-8- SULFONIC METHYLTETRAHYDRO- ACID, AMMONIUM SALT PYRROLO(2,1-B)OXAZOL-5(6H)- 2207 ((1R)-ENDO)-(+)-3- ONE, BROMOCAMPHOR, 98% 12568 (+)-4-CHOLESTEN-3-ONE, 98% 4804 ((1S)-(ENDO,ANTI))-(-)-3- 3774 (+)-5,6-O-CYCLOHEXYLIDENE-L- BROMOCAMPHOR-8- SULFONIC ASCORBIC ACID, 98% ACID, AMMONIUM SALT 11632 (+)-5-BROMO-2'-DEOXYURIDINE, 2208 ((1S)-ENDO)-(-)-3- 97% BROMOCAMPHOR, 98% 11634 (+)-5-FLUORODEOXYURIDINE, 769 ((1S)-ENDO)-(-)-BORNEOL, 99% 98+% 13454 ((2S,3S)-(+)- 11633 (+)-5-IODO-2'-DEOXYURIDINE, 98% BIS(DIPHENYLPHOSPHINO)- 4228 (+)-6-AMINOPENICILLANIC ACID, BUTANE)(N3-ALLYL)PD(II) CL04, 96% 97 8167 (+)-6-METHOXY-ALPHA-METHYL- 10297 ((3- 2- NAPHTHALENEACETIC ACID, DIMETHYLAMINO)PROPYL)TRIPH 98% ENYL- PHOSPHONIUM BROMIDE, 12586 (+)-ANDROSTA-1,4-DIENE-3,17- 99% DIONE, 98% 13458 ((R)-(+)-2,2'- 963 (+)-ARABINOGALACTAN BIS(DIPHENYLPHOSPHINO)-1,1'- -

Model Vulcanization Systems for Butyl Rubber and Halobutyl Rubber Manual
Exxon™ butyl and halobutyl rubber Model vulcanization systems for butyl rubber and halobutyl rubber manual Country name(s) 2 - Model vulcanization systems for butyl rubber and halobutyl rubber manual Model vulcanization systems for butyl rubber and halobutyl rubber manual - 3 Abstract The vulcanization of isobutylene-co-isoprene rubber (IIR), brominated isobutylene-co-isoprene rubber (BIIR), chlorinated isobutylene-co-isoprene rubber (CIIR), and brominated isobutylene-co-para-methylstyrene elastomer (BIMSM) differs from that of general-purpose rubbers (GPR). Butyl rubber has approximately 2% unsaturation in the backbone. Halobutyl rubber (BIIR and CIIR) incorporates the butyl backbone with either bromine or chlorine, which significantly increases the chemical reactivity of the isoprenyl units located in the butyl backbone. Similarly, in BIMSM the bromine atom is bonded to the para-methylstyrene (PMS) group, thus affording the completely saturated polymer backbone a site of chemical reactivity. Utilization of the unique attributes of butyl rubber and halobutyl rubbers with their minimal backbone unsaturation and of BIMSM elastomers with no backbone unsaturation is found in many areas of industry. These properties are excellent vapor impermeation, resistance to heat degradation, and improved chemical resistance as compared to general-purpose rubbers. However, this low amount of reactivity requires special consideration to vulcanize these isobutylene-based polymers. The type of vulcanization system selected is a function of the composite structure in which it is used, and the cured product performance requirements. Therefore, vulcanization systems vary and may include an accelerator package along with resins, zinc oxide, zinc oxide and sulfur, and quinoid systems. This review will discuss the types and selection of appropriate vulcanization systems for isobutylene-based elastomers. -

Dispensing Solutions for Cyanoacrylate Adhesives White
Dispensing Solutions for Cyanoacrylate Adhesives Contents How Cyanoacrylates Work .......................................................................3 Advantages of CAs ...................................................................................4 CA Dispensing Challenges .......................................................................4 Best Practices for Dispensing CAs .....................................................5-12 Best Systems for Dispensing CAs ....................................................13-14 Useful Resources ...................................................................................15 Introduction Cyanoacrylate adhesives, also known as CAs or cyanos, are highly effective at bonding many types of materials together in assembly processes. Often referred to as super glues, they exhibit high bond strength and fast cure times that help manufacturers speed production processes for higher throughput yields. This makes them an ideal choice for assembling products in a variety of industries, including automotive, electronics, life sciences, defense, and consumer goods. Though beneficial, these moisture-cure adhesives can be a challenge, especially when your assembly process requires precise, repeatable dispensing. This paper outlines proper handling methods and dispensing solutions for successful CA dispensing. Find out how to minimize material waste by more than 60% while also minimizing operator exposure to the adhesive. Speed your production processes while producing higher quality parts with -

An Overview on “Stages of Anesthesia and Some Novel General Anesthetics Drug”
Rohit Jaysing Bhor. et al. /Asian Journal of Research in Chemistry and Pharmaceutical Sciences. 5(4), 2017, 132-140. Review Article ISSN: 2349 – 7106 Asian Journal of Research in Chemistry and Pharmaceutical Sciences Journal home page: www.ajrcps.com AN OVERVIEW ON “STAGES OF ANESTHESIA AND SOME NOVEL GENERAL ANESTHETICS DRUG” Rohit Jaysing Bhor *1 , Bhadange Shubhangi 1, C. J. Bhangale 1 1* Department of Pharmaceutical Chemistry, PRES’s College of Pharmacy Chincholi, Tal-Sinner, Dist-Nasik, Maharashtra, India. ABSTRACT Anesthesia is a painless performance of medical producers. There are both major and minor risks of anesthesia. Anesthesia is a state of temporary induced loss of sensation or awareness. It gives analgesia i.e. relief from pain or prevention of pain and paralysis. General anaesthesia is a medically induced state of unconsciousness. It gives loss of protective reflux. It is carried out to allow medical procedures or medical surgery. It can be classified into 3 types like Intravenous Anesthetics Drug; Miscellaneous Drug; and Inhalational anesthetic Drug. Sodium thiopental is an ultra-short-acting barbiturate and has been used commonly in the induction phase of anesthesia. Methohexital is an example of barbiturates derivatives. It is classified as short-acting, and has a rapid onset of action. KEYWORDS Anesthesia, Sodium thiopental, Methohexital and Propofol. Author for Correspondence: INTRODUCTON Anesthesia is a state of temporary induced loss of sensation or awareness. It gives analgesia i.e. relief 1 Rohit Jaysing Bhor, from pain or prevention of pain and paralysis . A patient under the effects of anesthetic drug is known Department of Pharmaceutical Chemistry, as anesthetized. -
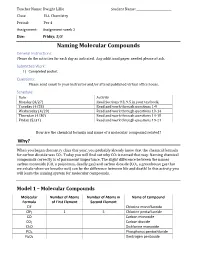
Naming Molecular Compounds General Instructions: Please Do the Activities for Each Day As Indicated
Teacher Name: Dwight Lillie Student Name: ________________________ Class: ELL Chemistry Period: Per 4 Assignment: Assignment week 2 Due: Friday, 5/8 Naming Molecular Compounds General Instructions: Please do the activities for each day as indicated. Any additional paper needed please attach. Submitted Work: 1) Completed packet. Questions: Please send email to your instructor and/or attend published virtual office hours. Schedule: Date Activity Monday (4/27) Read Sections 9.3, 9.5 in your textbook. Tuesday (4/28) Read and work through questions 1-9 Wednesday (4/29) Read and work through questions 10-14 Thursday (4/30) Read and work through questions 14-18 Friday (5/31) Read and work through questions 19-21 How are the chemical formula and name of a molecular compound related? Why? When you began chemistry class this year, you probably already knew that the chemical formula for carbon dioxide was CO2. Today you will find out why CO2 is named that way. Naming chemical compounds correctly is of paramount importance. The slight difference between the names carbon monoxide (CO, a poisonous, deadly gas) and carbon dioxide (CO2, a greenhouse gas that we exhale when we breathe out) can be the difference between life and death! In this activity you will learn the naming system for molecular compounds. Model 1 – Molecular Compounds Molecular Number of Atoms Number of Atoms in Name of Compound Formula of First Element Second Element ClF Chlorine monofluoride ClF5 1 5 Chlorine pentafluoride CO Carbon monoxide CO2 Carbon dioxide Cl2O Dichlorine monoxide PCl5 Phosphorus pentachloride N2O5 Dinitrogen pentoxide 1. Fill in the table to indicate the number of atoms of each type in the molecular formula.