Downloaded From
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Dental Pathology, Wear and Developmental Defects in South African Hominins
Dental pathology, wear and developmental defects in South African hominins IAN EDWARD TOWLE A thesis submitted in partial fulfilment of the requirements of Liverpool John Moores University for the degree of Doctor of Philosophy June 2017 Abstract Studying different types of dental pathology, wear, and developmental defects can allow inferences into diet and behaviour in a variety of ways. In this project data on these different variables were collected for South African hominins and compared with extant primates. The species studied include Paranthropus robustus, Australopithecus africanus, A. sediba, early Homo, Homo naledi, baboons, chimpanzees and gorillas. Macroscopic examination of each specimen was performed, with a 10X hand lens used to verify certain pathologies. Variables recorded include antemortem chipping, enamel hypoplasia, caries, occlusal wear, tertiary dentine, abscesses, and periodontal disease. Clear differences in frequencies were found in the different South African hominin species. Homo naledi displays high rates of chipping, especially small fractures above molar wear facets, likely reflecting a diet containing high levels of contaminants. Other noteworthy results include the high levels of pitting enamel hypoplasia in P. robustus molars compared to other species, likely due to a species-specific enamel formation property or developmental disturbance. The low rates of chipping in P. robustus does not fit with this species being a hard food specialist. Instead, the wear best supports a diet of low-quality tough vegetation. Australopithecus africanus likely had a broad diet, with angled molar wear, lack of caries, and high chipping frequencies supporting this conclusion. Seven new carious lesions are described, two from H. naledi and five P. -
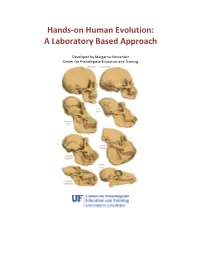
Hands-On Human Evolution: a Laboratory Based Approach
Hands-on Human Evolution: A Laboratory Based Approach Developed by Margarita Hernandez Center for Precollegiate Education and Training Author: Margarita Hernandez Curriculum Team: Julie Bokor, Sven Engling A huge thank you to….. Contents: 4. Author’s note 5. Introduction 6. Tips about the curriculum 8. Lesson Summaries 9. Lesson Sequencing Guide 10. Vocabulary 11. Next Generation Sunshine State Standards- Science 12. Background information 13. Lessons 122. Resources 123. Content Assessment 129. Content Area Expert Evaluation 131. Teacher Feedback Form 134. Student Feedback Form Lesson 1: Hominid Evolution Lab 19. Lesson 1 . Student Lab Pages . Student Lab Key . Human Evolution Phylogeny . Lab Station Numbers . Skeletal Pictures Lesson 2: Chromosomal Comparison Lab 48. Lesson 2 . Student Activity Pages . Student Lab Key Lesson 3: Naledi Jigsaw 77. Lesson 3 Author’s note Introduction Page The validity and importance of the theory of biological evolution runs strong throughout the topic of biology. Evolution serves as a foundation to many biological concepts by tying together the different tenants of biology, like ecology, anatomy, genetics, zoology, and taxonomy. It is for this reason that evolution plays a prominent role in the state and national standards and deserves thorough coverage in a classroom. A prime example of evolution can be seen in our own ancestral history, and this unit provides students with an excellent opportunity to consider the multiple lines of evidence that support hominid evolution. By allowing students the chance to uncover the supporting evidence for evolution themselves, they discover the ways the theory of evolution is supported by multiple sources. It is our hope that the opportunity to handle our ancestors’ bone casts and examine real molecular data, in an inquiry based environment, will pique the interest of students, ultimately leading them to conclude that the evidence they have gathered thoroughly supports the theory of evolution. -

Human Evolution: a Paleoanthropological Perspective - F.H
PHYSICAL (BIOLOGICAL) ANTHROPOLOGY - Human Evolution: A Paleoanthropological Perspective - F.H. Smith HUMAN EVOLUTION: A PALEOANTHROPOLOGICAL PERSPECTIVE F.H. Smith Department of Anthropology, Loyola University Chicago, USA Keywords: Human evolution, Miocene apes, Sahelanthropus, australopithecines, Australopithecus afarensis, cladogenesis, robust australopithecines, early Homo, Homo erectus, Homo heidelbergensis, Australopithecus africanus/Australopithecus garhi, mitochondrial DNA, homology, Neandertals, modern human origins, African Transitional Group. Contents 1. Introduction 2. Reconstructing Biological History: The Relationship of Humans and Apes 3. The Human Fossil Record: Basal Hominins 4. The Earliest Definite Hominins: The Australopithecines 5. Early Australopithecines as Primitive Humans 6. The Australopithecine Radiation 7. Origin and Evolution of the Genus Homo 8. Explaining Early Hominin Evolution: Controversy and the Documentation- Explanation Controversy 9. Early Homo erectus in East Africa and the Initial Radiation of Homo 10. After Homo erectus: The Middle Range of the Evolution of the Genus Homo 11. Neandertals and Late Archaics from Africa and Asia: The Hominin World before Modernity 12. The Origin of Modern Humans 13. Closing Perspective Glossary Bibliography Biographical Sketch Summary UNESCO – EOLSS The basic course of human biological history is well represented by the existing fossil record, although there is considerable debate on the details of that history. This review details both what is firmly understood (first echelon issues) and what is contentious concerning humanSAMPLE evolution. Most of the coCHAPTERSntention actually concerns the details (second echelon issues) of human evolution rather than the fundamental issues. For example, both anatomical and molecular evidence on living (extant) hominoids (apes and humans) suggests the close relationship of African great apes and humans (hominins). That relationship is demonstrated by the existing hominoid fossil record, including that of early hominins. -

Using Sub-Species Level Phylogenies
1 Reconstructing the ancestral phenotypes of great apes and 2 humans (Homininae) using sub-species level phylogenies 3 Keaghan Yaxley1 and Robert Foley2 4 1Leverhulme Centre for Human Evolutionary Studies, University of Cambridge, UK 5 2Leverhulme Centre for Human Evolutionary Studies, University of Cambridge, UK 6 7 8 1 9 Abstract 10 By their close affinity, the African great apes are of interest to the study of human evolution. 11 While numerous researchers have described the ancestors we share with these species with 12 reference to extant great apes, few have done so with phylogenetic comparative methods. 13 One obstacle to the application of these techniques is the within-species phenotypic variation 14 found in this group. Here we leverage this variation, modelling common ancestors using 15 Ancestral State Reconstructions (ASRs) with reference to subspecies level trait data. A 16 subspecies level phylogeny of the African great apes and humans was estimated from full- 17 genome mtDNA sequences and used to implement ASRs for fifteen continuous traits known 18 to vary between great ape subspecies. While including within-species phenotypic variation 19 increased phylogenetic signal for our traits and improved the performance of our ASRs, 20 whether this was done through the inclusion of subspecies phylogeny or through the use of 21 existing methods made little difference. Our ASRs corroborate previous findings that the Last 22 Common Ancestor (LCA) of humans, chimpanzees and bonobos was a chimp-like animal, 23 but also suggest that the LCA of humans, chimpanzees, bonobos and gorillas was an animal 24 unlike any extant African great ape. -

Environmental Hypotheses of Hominin Evolution
YEARBOOK OF PHYSICAL ANTHROPOLOGY 41:93–136 (1998) Environmental Hypotheses of Hominin Evolution RICHARD POTTS Human Origins Program, National Museum of Natural History, Smithsonian Institution, Washington, DC 20560-0112 KEY WORDS climate; habitat; adaptation; variability selection; fossil humans; stone tools ABSTRACT The study of human evolution has long sought to explain major adaptations and trends that led to the origin of Homo sapiens. Environmental scenarios have played a pivotal role in this endeavor. They represent statements or, more commonly, assumptions concerning the adap- tive context in which key hominin traits emerged. In many cases, however, these scenarios are based on very little if any data about the past settings in which early hominins lived. Several environmental hypotheses of human evolution are presented in this paper. Explicit test expectations are laid out, and a preliminary assessment of the hypotheses is made by examining the environmental records of Olduvai, Turkana, Olorgesailie, Zhoukoudian, Combe Grenal, and other hominin localities. Habitat-specific hypotheses have pre- vailed in almost all previous accounts of human adaptive history. The rise of African dry savanna is often cited as the critical event behind the develop- ment of terrestrial bipedality, stone toolmaking, and encephalized brains, among other traits. This savanna hypothesis has been countered recently by the woodland/forest hypothesis, which claims that Pliocene hominins had evolved in and were primarily attracted to closed habitats. The ideas that human evolution was fostered by cold habitats in higher latitudes or by seasonal variations in tropical and temperate zones also have their propo- nents. An alternative view, the variability selection hypothesis, states that large disparities in environmental conditions were responsible for important episodes of adaptive evolution. -

A Description of the Geological Context, Discrete Traits, and Linear Morphometrics of the Middle Pleistocene Hominin from Dali, Shaanxi Province, China
AMERICAN JOURNAL OF PHYSICAL ANTHROPOLOGY 150:141–157 (2013) A Description of the Geological Context, Discrete Traits, and Linear Morphometrics of the Middle Pleistocene Hominin from Dali, Shaanxi Province, China Xinzhi Wu1 and Sheela Athreya2* 1Laboratory for Human Evolution, Institute of Vertebrate Paleontology and Paleoanthropology, Chinese Academy of Sciences, Beijing 100044, China 2Department of Anthropology, Texas A&M University, College Station, TX 77843 KEY WORDS Homo heidelbergensis; Homo erectus; Asia ABSTRACT In 1978, a nearly complete hominin Afro/European Middle Pleistocene Homo and align it fossil cranium was recovered from loess deposits at the with Asian H. erectus.Atthesametime,itdisplaysa site of Dali in Shaanxi Province, northwestern China. more derived morphology of the supraorbital torus and It was subsequently briefly described in both English supratoral sulcus and a thinner tympanic plate than and Chinese publications. Here we present a compre- H. erectus, a relatively long upper (lambda-inion) occi- hensive univariate and nonmetric description of the pital plane with a clear separation of inion and opis- specimen and provide comparisons with key Middle thocranion, and an absolute and relative increase in Pleistocene Homo erectus and non-erectus hominins brain size, all of which align it with African and Euro- from Eurasia and Africa. In both respects we find pean Middle Pleistocene Homo. Finally, traits such as affinities with Chinese H. erectus as well as African the form of the frontal keel and the relatively short, and European Middle Pleistocene hominins typically broad midface align Dali specifically with other referred to as Homo heidelbergensis.Specifically,the Chinese specimens from the Middle Pleistocene Dali specimen possesses a low cranial height, relatively and Late Pleistocene, including H. -

Part 7 Humans
How do we recognise th 6 Extinction a human? • Walking on two legs (bipedal) We humans have destroyed over 80% of the habitat All non-human great apes are in the 34 continental biodiversity hotspots on Earth critically endangered. The biggest • Large brain relative to body size Biodiversity (see Part 2 of this series). threat is the loss of their habitat • Language, Symbolism, Happiness due to commercial logging, and the conversion of rainforest into palm oil plantations and biofuel Croplands of the Grazing agriculture. This in turn has led to lands take Earth occupy an an increase in human populations School activity & Extinction area about the up an area in forest areas and has led to an about the Part 7 size of South increase in bush meat trade. Why not set up a weekly Compiled by Dr John Anderson, Hominins America size of Africa debate on Democracy. Prof Francis Thackeray (ESI, Wits, anthropology) We humans, Homo sapiens, have arrived very late in the history of life. We’ve been Commercial Logging – The Why should we see the & Katherine Visser (Jhb Zoo, curator primates); cutting down of forest trees to layout by Ditshego Madopi & Anmari Hanekom around on Earth for only some 200,000 years. Our subtribe, the Hominina, have 7 bil world as ours alone? Bring all the cities sell wood and make room for Wouldn’t it be a good been around for some 7 million years; and our subfamily, the Homininae including Homo sapiens population Chimps agriculture is making less and less and towns and explosion population plan to give our close the chimps and gorillas, for some 10 million years. -

A Unique Middle Miocene European Hominoid and the Origins of the Great Ape and Human Clade Salvador Moya` -Sola` A,1, David M
A unique Middle Miocene European hominoid and the origins of the great ape and human clade Salvador Moya` -Sola` a,1, David M. Albab,c, Sergio Alme´ cijac, Isaac Casanovas-Vilarc, Meike Ko¨ hlera, Soledad De Esteban-Trivignoc, Josep M. Roblesc,d, Jordi Galindoc, and Josep Fortunyc aInstitucio´Catalana de Recerca i Estudis Avanc¸ats at Institut Catala`de Paleontologia (ICP) and Unitat d’Antropologia Biolo`gica (Dipartimento de Biologia Animal, Biologia Vegetal, i Ecologia), Universitat Auto`noma de Barcelona, Edifici ICP, Campus de Bellaterra s/n, 08193 Cerdanyola del Valle`s, Barcelona, Spain; bDipartimento di Scienze della Terra, Universita`degli Studi di Firenze, Via G. La Pira 4, 50121 Florence, Italy; cInstitut Catala`de Paleontologia, Universitat Auto`noma de Barcelona, Edifici ICP, Campus de Bellaterra s/n, 08193 Cerdanyola del Valle`s, Barcelona, Spain; and dFOSSILIA Serveis Paleontolo`gics i Geolo`gics, S.L. c/ Jaume I nu´m 87, 1er 5a, 08470 Sant Celoni, Barcelona, Spain Edited by David Pilbeam, Harvard University, Cambridge, MA, and approved March 4, 2009 (received for review November 20, 2008) The great ape and human clade (Primates: Hominidae) currently sediments by the diggers and bulldozers. After 6 years of includes orangutans, gorillas, chimpanzees, bonobos, and humans. fieldwork, 150 fossiliferous localities have been sampled from the When, where, and from which taxon hominids evolved are among 300-m-thick local stratigraphic series of ACM, which spans an the most exciting questions yet to be resolved. Within the Afro- interval of 1 million years (Ϸ12.5–11.3 Ma, Late Aragonian, pithecidae, the Kenyapithecinae (Kenyapithecini ؉ Equatorini) Middle Miocene). -

Mandibular Ramus Shape of Australopithecus Sediba Suggests a Single Variable Species
Journal of Human Evolution 100 (2016) 54e64 Contents lists available at ScienceDirect Journal of Human Evolution journal homepage: www.elsevier.com/locate/jhevol Mandibular ramus shape of Australopithecus sediba suggests a single variable species * Terrence B. Ritzman a, b, c, d, , 1, Claire E. Terhune e, 1, Philipp Gunz f, Chris A. Robinson g a Department of Neuroscience, Washington University School of Medicine, 660 S. Euclid Ave., St. Louis, MO, USA b Department of Archaeology, University of Cape Town, Private Bag, Rondebosch, Cape Town, 7701, South Africa c Human Evolution Research Institute, University of Cape Town, Private Bag, Rondebosch, Cape Town, 7701, South Africa d School of Human Evolution, Arizona State University, School of Human Evolution and Social Change Building, P.O. Box 872402, Tempe, AZ, 85287, USA e Department of Anthropology, 330 Old Main, University of Arkansas, Fayetteville, 72701, USA f Department of Human Evolution, Max Planck Institute for Evolutionary Anthropology, Deutscher Platz 6, Leipzig, 04103, Germany g Department of Biological Sciences, Bronx Community College, City University of New York, 2155 University Avenue, Bronx, NY, 10453, USA article info abstract Article history: The fossils from Malapa cave, South Africa, attributed to Australopithecus sediba, include two partial Received 15 December 2015 skeletonsdMH1, a subadult, and MH2, an adult. Previous research noted differences in the mandibular Accepted 1 September 2016 rami of these individuals. This study tests three hypotheses that could explain these differences. The first two state that the differences are due to ontogenetic variation and sexual dimorphism, respec- tively. The third hypothesis, which is relevant to arguments suggesting that MH1 belongs in the genus Keywords: Australopithecus and MH2 in Homo, is that the differences are due to the two individuals representing Australopithecus sediba more than one taxon. -

Morphological Affinities of Homo Naledi with Other Plio
Anais da Academia Brasileira de Ciências (2017) 89(3 Suppl.): 2199-2207 (Annals of the Brazilian Academy of Sciences) Printed version ISSN 0001-3765 / Online version ISSN 1678-2690 http://dx.doi.org/10.1590/0001-3765201720160841 www.scielo.br/aabc | www.fb.com/aabcjournal Morphological affinities ofHomo naledi with other Plio- Pleistocene hominins: a phenetic approach WALTER A. NEVES1, DANILO V. BERNARDO2 and IVAN PANTALEONI1 1Instituto de Biociências, Universidade de São Paulo, Departamento de Genética e Biologia Evolutiva, Laboratório de Estudos Evolutivos e Ecológicos Humanos, Rua do Matão, 277, sala 218, Cidade Universitária, 05508-090 São Paulo, SP, Brazil 2Instituto de Ciências Humanas e da Informação, Universidade Federal do Rio Grande, Laboratório de Estudos em Antropologia Biológica, Bioarqueologia e Evolução Humana, Área de Arqueologia e Antropologia, Av. Itália, Km 8, Carreiros, 96203-000 Rio Grande, RS, Brazil Manuscript received on December 2, 2016; accepted for publication on February 21, 2017 ABSTRACT Recent fossil material found in Dinaledi Chamber, South Africa, was initially described as a new species of genus Homo, namely Homo naledi. The original study of this new material has pointed to a close proximity with Homo erectus. More recent investigations have, to some extent, confirmed this assignment. Here we present a phenetic analysis based on dentocranial metric variables through Principal Components Analysis and Cluster Analysis based on these fossils and other Plio-Pleistocene hominins. Our results concur that the Dinaledi fossil hominins pertain to genus Homo. However, in our case, their nearest neighbors are Homo habilis and Australopithecus sediba. We suggest that Homo naledi is in fact a South African version of Homo habilis, and not a new species. -

New Fossils of Australopithecus Sediba Reveal a Nearly Complete Lower Back
bioRxiv preprint doi: https://doi.org/10.1101/2021.05.27.445933; this version posted May 29, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. 1 New fossils of Australopithecus sediba reveal a nearly complete lower back 2 Scott A. Williams1,2,3*, Thomas C. Prang4, Marc R. Meyer5, Thierra K. Nalley6, Renier Van Der 3 Merwe3, Christopher Yelverton7, Daniel García-Martínez3,8,9, Gabrielle A. Russo10, Kelly R. 4 Ostrofsky11, Jennifer Eyre12, Mark Grabowski13, Shahed Nalla3,14, Markus Bastir3,8, Peter 5 Schmid3,15, Steven E. Churchill3,16, Lee R. Berger3 6 1Center for the Study of Human Origins, Department of Anthropology, New York University, 7 New York, NY 10003, USA 8 2New York Consortium in Evolutionary Primatology, New York, NY 10024, USA 9 3Centre for the Exploration of the Deep Human Journey, University of the Witwatersrand, 10 Private Bag 3, Wits 2050, Johannesburg, South Africa 11 4Department of Anthropology, Texas A&M University, College Station, TX 77843, USA 12 5Department of Anthropology, Chaffey College, Rancho Cucamonga, CA 91737, USA 13 6Western University of Health Sciences, College of Osteopathic Medicine of the Pacific, 14 Department of Medical Anatomical Sciences, Pomona, CA 91766, USA 15 7Department of Chiropractic, Faculty of Health Sciences, University of Johannesburg, 16 Johannesburg, South Africa 17 8Departamento de Paleobiología, Museo Nacional de Ciencias Naturales (CSIC), José Gutiérrez 18 Abascal 2, 28006, Madrid, Spain 19 9Centro Nacional de Investigación sobre la Evolución Humana (CENIEH), Paseo Sierra de 20 Atapuerca, 3, 09002, Burgos, Spain 21 10Department of Anthropology, Stony Brook University, Stony Brook, NY 11790, ISA 22 11Department of Anatomy, College of Osteopathic Medicine, New York Institute of Technology, 23 Old Westbury, NY 11569, USA 1 bioRxiv preprint doi: https://doi.org/10.1101/2021.05.27.445933; this version posted May 29, 2021. -

A Volumetric Technique for Fossil Body Mass Estimation Applied to Australopithecus
*Revised Manuscript Unmarked Click here to view linked References 1 A volumetric technique for fossil body mass estimation applied to Australopithecus 2 afarensis 3 4 Charlotte A. Brasseya, *, +, Thomas O’Mahoneyb, +, Andrew T. Chamberlainb, William I. 5 Sellersb 6 7 aSchool of Science and the Environment, Manchester Metropolitan University, Chester 8 Street, Manchester, M1 5GD, UK 9 bSchool of Earth and Environmental Sciences, University of Manchester, Oxford Road, 10 Manchester, M13 9PT, UK 11 12 *Corresponding author; [email protected] 13 +These authors contributed equally to this work 14 15 16 Keywords: Lucy; reconstruction; convex hull; hominin; primate; 1 17 Abstract 18 Fossil body mass estimation is a wellestablished practice within the field of physical 19 anthropology. Previous studies have relied upon traditional allometric approaches, in which 20 the relationship between one/several skeletal dimensions and body mass in a range of 21 modern taxa is used in a predictive capacity. The lack of relatively complete skeletons has 22 thus far limited the potential application of alternative mass estimation techniques, such as 23 volumetric reconstruction, to fossil hominins. Yet across vertebrate palaeontology more 24 broadly, novel volumetric approaches are resulting in predicted values for fossil body mass 25 very different to those estimated by traditional allometry. Here we present a new digital 26 reconstruction of Australopithecus afarensis (A.L. 288-1; ‘Lucy’) and a convex hull-based 27 volumetric estimate of body mass. The technique relies upon identifying a predictable 28 relationship between the ‘shrink-wrapped’ volume of the skeleton and known body mass in a 29 range of modern taxa, and subsequent application to an articulated model of the fossil taxa 30 of interest.