Supplementary Material
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Role of Glutathionperoxidase 4 (GPX4) in Hematopoiesis and Leukemia
The role of glutathionperoxidase 4 (GPX4) in hematopoiesis and leukemia von Kira Célénie Stahnke Inaugral- Dissertation zur Erlangung der Doktorwürde der TierärztliChen Fakultät der Ludwig-Maximilians-Universität MünChen The role of glutathionperoxidase 4 (GPX4) in hematopoiesis and leukemia von Kira Célénie Stahnke aus Dortmund München 2016 Aus dem VeterinärwissensChaftliChen Department der TierärztliChen Fakultät der Ludwig-Maximilians-Universität München Lehrstuhl für Molekulare Tierzucht und Biotechnologie Arbeit angefertigt unter der Leitung von Univ.-Prof. Dr. Eckhard Wolf Angefertigt am Institut für Experimentelle TumorforsChung Universitätsklinikum Ulm Mentor: Prof. Dr. Christian Buske GedruCkt mit Genehmigung der TierärztliChen Fakultät der Ludwig-Maximilians-Universität München Dekan: Univ.-Prof. Dr. JoaChim Braun Berichterstatter: Univ.-Prof. Dr. Eckard Wolf Korreferent/en: Priv.-Doz. Dr. Bianka Schulz Tag der Promotion: 06.02.2016 Table of contents List of Abbreviations..............................................................................2 1 Introduction ........................................................................................6 1.1 ROS and oxidative stress in physiology and pathology..................................................... 6 1.1.1 SourCes of Reactive Oxygen SpeCies..................................................................................................6 1.1.2 PhysiologiCal role of ROS........................................................................................................................8 -

In Thyroid Cancer
Metere et al. Cancer Cell Int (2018) 18:7 https://doi.org/10.1186/s12935-018-0504-4 Cancer Cell International PRIMARY RESEARCH Open Access A possible role for selenoprotein glutathione peroxidase (GPx1) and thioredoxin reductases (TrxR1) in thyroid cancer: our experience in thyroid surgery Alessio Metere1* , Francesca Frezzotti1, Claire Elizabeth Graves2, Massimo Vergine1, Alessandro De Luca1, Donatella Pietraforte3 and Laura Giacomelli1 Abstract Background: Oxidative stress is responsible for some alterations in the chemical structure and, consequently, in the function of proteins, lipids, and DNA. Recent studies have linked oxidative stress to cancers, particularly thyroid cancer, but the mechanisms remain unclear. Here, we further characterize the role of oxidative stress in thyroid cancer by analyzing the expression of two selenium antioxidant molecules, glutathione peroxidase (GPx1) and thioredoxin reductase (TrxR1) in thyroid cancer cells. Methods: Samples of both healthy thyroid tissue and thyroid tumor were taken for analysis after total thyroidectomy. The expression of GPx1 and TrxR1 was revealed by Western blot analysis and quantifed by densitometric analy- ses, while the evaluation of free radicals was performed by Electron Paramagnetic Resonance (EPR)-spin trapping technique. Results: Our results show a decrease in the expression of GPx1 and TrxR1 ( 45.7 and 43.2% respectively, p < 0.01) in the thyroid cancer cells compared to the healthy cells. In addition, the EPR− technique− shows an increase of free radicals in tumor tissue, signifcantly higher than that found in healthy thyroid tissue ( 116.3%, p < 0.01). + Conclusions: Our fndings underscore the relationship between thyroid cancer and oxidative stress, showing the imbalance of the oxidant/antioxidant system in thyroid cancer tissue. -

A Machine Learning Tool for Predicting Protein Function Anna
The Woolf Classifier Building Pipeline: A Machine Learning Tool for Predicting Protein Function Anna Farrell-Sherman Submitted in Partial Fulfillment of the Prerequisite for Honors in Computer Science under the advisement of Eni Mustafaraj and Vanja Klepac-Ceraj May 2019 © 2019 Anna Farrell-Sherman Abstract Proteins are the machinery that allow cells to grow, reproduce, communicate, and create multicellular organisms. For all of their importance, scientists still have a hard time understanding the function of a protein based on its sequence alone. For my honors thesis in computer science, I created a machine learning tool that can predict the function of a protein based solely on its amino acid sequence. My tool gives scientists a structure in which to build a � Nearest Neighbor or random forest classifier to distinguish between proteins that can and cannot perform a given function. Using default Min-Max scaling, and the Matthews Correlation Coefficient for accuracy assessment, the Woolf Pipeline is built with simplified choices to guide users to success. i Acknowledgments There are so many people who made this thesis possible. First, thank you to my wonderful advisors, Eni and Vanja, who never gave up on me, and always pushed me to try my hardest. Thank you also to the other members of my committee, Sohie Lee, Shikha Singh, and Rosanna Hertz for supporting me through this process. To Kevin, Sophie R, Sophie E, and my sister Phoebe, thank you for reading my drafts, and advising me when the going got tough. To all my wonderful friends, who were always there with encouraging words, warm hugs, and many congratulations, I cannot thank you enough. -
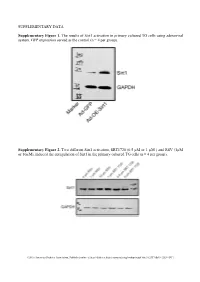
SUPPLEMENTARY DATA Supplementary Figure 1. The
SUPPLEMENTARY DATA Supplementary Figure 1. The results of Sirt1 activation in primary cultured TG cells using adenoviral system. GFP expression served as the control (n = 4 per group). Supplementary Figure 2. Two different Sirt1 activators, SRT1720 (0.5 µM or 1 µM ) and RSV (1µM or 10µM), induced the upregulation of Sirt1 in the primary cultured TG cells (n = 4 per group). ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. Primers used in qPCR Gene Name Primer Sequences Product Size (bp) Sirt1 F: tgccatcatgaagccagaga 241 (NM_001159589) R: aacatcgcagtctccaagga NOX4 F: tgtgcctttattgtgcggag 172 (NM_001285833.1) R: gctgatacactggggcaatg Supplementary Table 2. Antibodies used in Western blot or Immunofluorescence Antibody Company Cat. No Isotype Dilution Sirt1 Santa Cruz * sc-15404 Rabbit IgG 1/200 NF200 Sigma** N5389 Mouse IgG 1/500 Tubulin R&D# MAB1195 Mouse IgG 1/500 NOX4 Abcam† Ab133303 Rabbit IgG 1/500 NOX2 Abcam Ab129068 Rabbit IgG 1/500 phospho-AKT CST‡ #4060 Rabbit IgG 1/500 EGFR CST #4267 Rabbit IgG 1/500 Ki67 Santa Cruz sc-7846 Goat IgG 1/500 * Santa Cruz Biotechnology, Santa Cruz, CA, USA ** Sigma aldrich, Shanghai, China # R&D Systems Inc, Minneapolis, MN, USA † Abcam, Inc., Cambridge, MA, USA ‡ Cell Signaling Technology, Inc., Danvers, MA, USA ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary -

GPX4 at the Crossroads of Lipid Homeostasis and Ferroptosis Giovanni C
REVIEW GPX4 www.proteomics-journal.com GPX4 at the Crossroads of Lipid Homeostasis and Ferroptosis Giovanni C. Forcina and Scott J. Dixon* formation of toxic radicals (e.g., R-O•).[5] Oxygen is necessary for aerobic metabolism but can cause the harmful The eight mammalian GPX proteins fall oxidation of lipids and other macromolecules. Oxidation of cholesterol and into three clades based on amino acid phospholipids containing polyunsaturated fatty acyl chains can lead to lipid sequence similarity: GPX1 and GPX2; peroxidation, membrane damage, and cell death. Lipid hydroperoxides are key GPX3, GPX5, and GPX6; and GPX4, GPX7, and GPX8.[6] GPX1–4 and 6 (in intermediates in the process of lipid peroxidation. The lipid hydroperoxidase humans) are selenoproteins that contain glutathione peroxidase 4 (GPX4) converts lipid hydroperoxides to lipid an essential selenocysteine in the enzyme + alcohols, and this process prevents the iron (Fe2 )-dependent formation of active site, while GPX5, 6 (in mouse and toxic lipid reactive oxygen species (ROS). Inhibition of GPX4 function leads to rats), 7, and 8 use an active site cysteine lipid peroxidation and can result in the induction of ferroptosis, an instead. Unlike other family members, GPX4 (PHGPx) can act as a phospholipid iron-dependent, non-apoptotic form of cell death. This review describes the hydroperoxidase to reduce lipid perox- formation of reactive lipid species, the function of GPX4 in preventing ides to lipid alcohols.[7,8] Thus,GPX4ac- oxidative lipid damage, and the link between GPX4 dysfunction, lipid tivity is essential to maintain lipid home- oxidation, and the induction of ferroptosis. ostasis in the cell, prevent the accumula- tion of toxic lipid ROS and thereby block the onset of an oxidative, iron-dependent, non-apoptotic mode of cell death termed 1. -

Thiol Peroxidases Mediate Specific Genome-Wide Regulation of Gene Expression in Response to Hydrogen Peroxide
Thiol peroxidases mediate specific genome-wide regulation of gene expression in response to hydrogen peroxide Dmitri E. Fomenkoa,1,2, Ahmet Koca,1, Natalia Agishevaa, Michael Jacobsena,b, Alaattin Kayaa,c, Mikalai Malinouskia,c, Julian C. Rutherfordd, Kam-Leung Siue, Dong-Yan Jine, Dennis R. Winged, and Vadim N. Gladysheva,c,2 aDepartment of Biochemistry, University of Nebraska, Lincoln, NE 68588-0664; bDepartment of Life Sciences, Wayne State College, Wayne, NE 68787; dDepartment of Medicine, University of Utah Health Sciences Center, Salt Lake City, UT 84132; eDepartment of Biochemistry, University of Hong Kong, Hong Kong, China; and cDivision of Genetics, Department of Medicine, Brigham and Women’s Hospital and Harvard Medical School, Boston, MA 02115 Edited by Joan Selverstone Valentine, University of California, Los Angeles, CA, and approved December 22, 2010 (received for review July 21, 2010) Hydrogen peroxide is thought to regulate cellular processes by and could withstand significant oxidative stress. It responded to direct oxidation of numerous cellular proteins, whereas antioxi- several redox stimuli by robust transcriptional reprogramming. dants, most notably thiol peroxidases, are thought to reduce However, it was unable to transcriptionally respond to hydrogen peroxides and inhibit H2O2 response. However, thiol peroxidases peroxide. The data suggested that thiol peroxidases transfer have also been implicated in activation of transcription factors oxidative signals from peroxides to target proteins, thus activating and signaling. It remains unclear if these enzymes stimulate or various transcriptional programs. This study revealed a previously inhibit redox regulation and whether this regulation is widespread undescribed function of these proteins, in addition to their roles or limited to a few cellular components. -

Micrornas As New Regulators of Neutrophil Extracellular Trap Formation
International Journal of Molecular Sciences Review MicroRNAs as New Regulators of Neutrophil Extracellular Trap Formation Sonia Águila † , Ascensión M. de los Reyes-García †, María P. Fernández-Pérez, Laura Reguilón-Gallego, Laura Zapata-Martínez, Inmaculada Ruiz-Lorente, Vicente Vicente , Rocío González-Conejero *,‡ and Constantino Martínez *,‡ Department of Hematology and Medical Oncology, Morales Meseguer University Hospital, Centro Regional de Hemodonación, Universidad de Murcia, IMIB, C/Ronda de Garay S/N, 30003 Murcia, Spain; [email protected] (S.Á.); [email protected] (A.M.d.l.R.-G.); [email protected] (M.P.F.-P.); [email protected] (L.R.-G.); [email protected] (L.Z.-M.); [email protected] (I.R.-L.); [email protected] (V.V.) * Correspondence: [email protected] (R.G.-C.); [email protected] (C.M.); Tel.: +34-968341990 (R.G.-C. & C.M.); Fax: +34-968261914 (R.G.-C. & C.M.) † These authors contributed equally to this work. ‡ These authors shared senior authorship. Abstract: Neutrophil extracellular traps (NETs) are formed after neutrophils expelled their chromatin content in order to primarily capture and eliminate pathogens. However, given their characteristics due in part to DNA and different granular proteins, NETs may induce a procoagulant response linking inflammation and thrombosis. Unraveling NET formation molecular mechanisms as well Citation: Águila, S.; de los as the intracellular elements that regulate them is relevant not only for basic knowledge but also Reyes-García, A.M.; Fernández-Pérez, to design diagnostic and therapeutic tools that may prevent their deleterious effects observed in M.P.; Reguilón-Gallego, L.; several inflammatory pathologies (e.g., cardiovascular and autoimmune diseases, cancer). -

Lactoperoxidase Antibacterial System: Natural Occurrence, Biological Functions and Practical Applications
724 Journal of Food Protection, Vol. 47. No. 9, Pages 724-732 (September 1984) Copyright®, International Association of Milk, Food, and Environmental Sanitarians Lactoperoxidase Antibacterial System: Natural Occurrence, Biological Functions and Practical Applications BRUNO REITER1 and GORAN HARNULV2* Downloaded from http://meridian.allenpress.com/jfp/article-pdf/47/9/724/1650811/0362-028x-47_9_724.pdf by guest on 29 September 2021 National Institute for Research in Dairying, Shinfield, Reading, RG2 9AT, England and Alfa-Laval Agri International AS, P.O. Box 39, S-I47 00 Tumba, Sweden (Received for publication January 30, 1984) ABSTRACT (37,80). The various biological functions of the LP sys tem, which have now been established, will be discussed In the present review dealing with the antibacterial lac below. toperoxidase (LP) system, it is shown that the two reactants In recent years, much work has been devoted to practi thiocyanate (SCN~) and hydrogen peroxide (H202) as well as cal applications of the LP system. The effect against oral the catalytic enzyme lactoperoxidase (LP) are widely distributed streptococci (5,19,33,101,105,107) has led to the de in nature and that evidence for the activity of the LP system velopment and marketing of a toothpaste containing nec in animals, including man, is accumulating. The in vitro effects on bacterial and animal cells are discussed and the unique ac essary ingredients to activate the LP system. Promising tion of the LP system on the bacterial cytoplasmic membrane results have also been obtained when including activating is pointed out. Some practical applications are also presented, components in the feed to calves (81,83,86) with the aim with particular emphses on the possibility of utilizing the LP of potentiating the LP system in the intestinal tract. -

The C718T Polymorphism in the 3″-Untranslated
Hypertension Research (2012) 35, 507–512 & 2012 The Japanese Society of Hypertension All rights reserved 0916-9636/12 www.nature.com/hr ORIGINAL ARTICLE The C718T polymorphism in the 3¢-untranslated region of glutathione peroxidase-4 gene is a predictor of cerebral stroke in patients with essential hypertension Alexey V Polonikov1, Ekaterina K Vialykh2, Mikhail I Churnosov3, Thomas Illig4, Maxim B Freidin5, Oksana V Vasil¢eva1, Olga Yu Bushueva1, Valentina N Ryzhaeva1, Irina V Bulgakova1 and Maria A Solodilova1 In the present study we have investigated the association of three single nucleotide polymorphisms in glutathione peroxidase (GPx) genes GPX1 rs1050450 (P198L), GPX3 rs2070593 (G930A) and GPX4 rs713041 (T718C) with the risk of cerebral stroke (CS) in patients with essential hypertension (EH). A total of 667 unrelated EH patients of Russian origin, including 306 hypertensives (the EH–CS group) who suffered from CS and 361 people (the EH–CS group) who did not have cerebrovascular accidents, were enrolled in the study. The variant allele 718C of the GPX4 gene was found to be significantly associated with an increased risk of CS in hypertensive patients (odds ratio (OR) 1.53, 95% confidence interval (CI) 1.23–1.90, Padj¼0.0003). The prevalence of the 718TC and 718CC genotypes of the GPX4 gene was higher in the EH–CS group than the EH-alone group (OR¼2.12, 95%CI 1.42–3.16, Padj¼0.0018). The association of the variant GPX4 genotypes with the increased risk of CS in hypertensives remained statistically significant after adjusting for confounding variables such as sex, body mass index (BMI), blood pressure and antihypertensive medication use (OR¼2.18, 95%CI 1.46–3.27, P¼0.0015). -

Programmed Cell-Death by Ferroptosis: Antioxidants As Mitigators
International Journal of Molecular Sciences Review Programmed Cell-Death by Ferroptosis: Antioxidants as Mitigators Naroa Kajarabille 1 and Gladys O. Latunde-Dada 2,* 1 Nutrition and Obesity Group, Department of Nutrition and Food Sciences, University of the Basque Country (UPV/EHU), 01006 Vitoria, Spain; [email protected] 2 King’s College London, Department of Nutritional Sciences, Faculty of Life Sciences and Medicine, Franklin-Wilkins Building, 150 Stamford Street, London SE1 9NH, UK * Correspondence: [email protected] Received: 9 September 2019; Accepted: 2 October 2019; Published: 8 October 2019 Abstract: Iron, the fourth most abundant element in the Earth’s crust, is vital in living organisms because of its diverse ligand-binding and electron-transfer properties. This ability of iron in the redox cycle as a ferrous ion enables it to react with H2O2, in the Fenton reaction, to produce a hydroxyl radical ( OH)—one of the reactive oxygen species (ROS) that cause deleterious oxidative damage • to DNA, proteins, and membrane lipids. Ferroptosis is a non-apoptotic regulated cell death that is dependent on iron and reactive oxygen species (ROS) and is characterized by lipid peroxidation. It is triggered when the endogenous antioxidant status of the cell is compromised, leading to lipid ROS accumulation that is toxic and damaging to the membrane structure. Consequently, oxidative stress and the antioxidant levels of the cells are important modulators of lipid peroxidation that induce this novel form of cell death. Remedies capable of averting iron-dependent lipid peroxidation, therefore, are lipophilic antioxidants, including vitamin E, ferrostatin-1 (Fer-1), liproxstatin-1 (Lip-1) and possibly potent bioactive polyphenols. -

Catalysis of Peroxide Reduction by Fast Reacting Protein Thiols Focus Review †,‡ †,‡ ‡,§ ‡,§ ∥ Ari Zeida, Madia Trujillo, Gerardo Ferrer-Sueta, Ana Denicola, Darío A
Review Cite This: Chem. Rev. 2019, 119, 10829−10855 pubs.acs.org/CR Catalysis of Peroxide Reduction by Fast Reacting Protein Thiols Focus Review †,‡ †,‡ ‡,§ ‡,§ ∥ Ari Zeida, Madia Trujillo, Gerardo Ferrer-Sueta, Ana Denicola, Darío A. Estrin, and Rafael Radi*,†,‡ † ‡ § Departamento de Bioquímica, Centro de Investigaciones Biomedicaś (CEINBIO), Facultad de Medicina, and Laboratorio de Fisicoquímica Biologica,́ Facultad de Ciencias, Universidad de la Republica,́ 11800 Montevideo, Uruguay ∥ Departamento de Química Inorganica,́ Analítica y Química-Física and INQUIMAE-CONICET, Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires, 2160 Buenos Aires, Argentina ABSTRACT: Life on Earth evolved in the presence of hydrogen peroxide, and other peroxides also emerged before and with the rise of aerobic metabolism. They were considered only as toxic byproducts for many years. Nowadays, peroxides are also regarded as metabolic products that play essential physiological cellular roles. Organisms have developed efficient mechanisms to metabolize peroxides, mostly based on two kinds of redox chemistry, catalases/peroxidases that depend on the heme prosthetic group to afford peroxide reduction and thiol-based peroxidases that support their redox activities on specialized fast reacting cysteine/selenocysteine (Cys/Sec) residues. Among the last group, glutathione peroxidases (GPxs) and peroxiredoxins (Prxs) are the most widespread and abundant families, and they are the leitmotif of this review. After presenting the properties and roles of different peroxides in biology, we discuss the chemical mechanisms of peroxide reduction by low molecular weight thiols, Prxs, GPxs, and other thiol-based peroxidases. Special attention is paid to the catalytic properties of Prxs and also to the importance and comparative outlook of the properties of Sec and its role in GPxs. -

Significance of Peroxidase in Eosinophils Margaret A
University of Colorado, Boulder CU Scholar Series in Biology Ecology & Evolutionary Biology Spring 4-1-1958 Significance of peroxidase in eosinophils Margaret A. Kelsall Follow this and additional works at: http://scholar.colorado.edu/sbio Recommended Citation Kelsall, Margaret A., "Significance of peroxidase in eosinophils" (1958). Series in Biology. 14. http://scholar.colorado.edu/sbio/14 This Article is brought to you for free and open access by Ecology & Evolutionary Biology at CU Scholar. It has been accepted for inclusion in Series in Biology by an authorized administrator of CU Scholar. For more information, please contact [email protected]. SIGNIFICANCE OF PEROXIDASE IN EOSINOPHILS M a rg a ret A . K e lsa ll Peroxidase-bearing granules are the primary component and product of eosino phils. The physiological significance of eosinophils is, therefore, considered to be related to the ability of this cell to synthesize, store, and transport peroxidase and to release the peroxidase-positive granules into body fluids by a lytic process that is controlled by hormones, by variations in the histamine-epinephrine balance, and by several other stimuli. Peroxidase occurs not only in eosinophils, but also in neutrophils and blood platelets; but it is not present in most cells of animal tissues. The purpose of this work is to consider, as a working hypothesis, that the function of eosinophils is to produce, store, and transport peroxidase to catalyze oxidations. Many of the aerobic dehydrogenases that catalyze reactions in which hydrogen peroxide is produced are involved in protein catabolism. Therefore, relations between eosinophils and several normal and pathological conditions of increased protein catabolism are emphasized, and also the significance of peroxi dase in eosinophils and other leukocytes to H 20 2 produced by irradiation is considered.