BY FENNER A. CHACE, JR. Department of Invertebrate
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

A Classification of Living and Fossil Genera of Decapod Crustaceans
RAFFLES BULLETIN OF ZOOLOGY 2009 Supplement No. 21: 1–109 Date of Publication: 15 Sep.2009 © National University of Singapore A CLASSIFICATION OF LIVING AND FOSSIL GENERA OF DECAPOD CRUSTACEANS Sammy De Grave1, N. Dean Pentcheff 2, Shane T. Ahyong3, Tin-Yam Chan4, Keith A. Crandall5, Peter C. Dworschak6, Darryl L. Felder7, Rodney M. Feldmann8, Charles H. J. M. Fransen9, Laura Y. D. Goulding1, Rafael Lemaitre10, Martyn E. Y. Low11, Joel W. Martin2, Peter K. L. Ng11, Carrie E. Schweitzer12, S. H. Tan11, Dale Tshudy13, Regina Wetzer2 1Oxford University Museum of Natural History, Parks Road, Oxford, OX1 3PW, United Kingdom [email protected] [email protected] 2Natural History Museum of Los Angeles County, 900 Exposition Blvd., Los Angeles, CA 90007 United States of America [email protected] [email protected] [email protected] 3Marine Biodiversity and Biosecurity, NIWA, Private Bag 14901, Kilbirnie Wellington, New Zealand [email protected] 4Institute of Marine Biology, National Taiwan Ocean University, Keelung 20224, Taiwan, Republic of China [email protected] 5Department of Biology and Monte L. Bean Life Science Museum, Brigham Young University, Provo, UT 84602 United States of America [email protected] 6Dritte Zoologische Abteilung, Naturhistorisches Museum, Wien, Austria [email protected] 7Department of Biology, University of Louisiana, Lafayette, LA 70504 United States of America [email protected] 8Department of Geology, Kent State University, Kent, OH 44242 United States of America [email protected] 9Nationaal Natuurhistorisch Museum, P. O. Box 9517, 2300 RA Leiden, The Netherlands [email protected] 10Invertebrate Zoology, Smithsonian Institution, National Museum of Natural History, 10th and Constitution Avenue, Washington, DC 20560 United States of America [email protected] 11Department of Biological Sciences, National University of Singapore, Science Drive 4, Singapore 117543 [email protected] [email protected] [email protected] 12Department of Geology, Kent State University Stark Campus, 6000 Frank Ave. -

Two Freshwater Shrimp Species of the Genus Caridina (Decapoda, Caridea, Atyidae) from Dawanshan Island, Guangdong, China, with the Description of a New Species
A peer-reviewed open-access journal ZooKeys 923: 15–32 (2020) Caridina tetrazona 15 doi: 10.3897/zookeys.923.48593 RESEarcH articLE http://zookeys.pensoft.net Launched to accelerate biodiversity research Two freshwater shrimp species of the genus Caridina (Decapoda, Caridea, Atyidae) from Dawanshan Island, Guangdong, China, with the description of a new species Qing-Hua Chen1, Wen-Jian Chen2, Xiao-Zhuang Zheng2, Zhao-Liang Guo2 1 South China Institute of Environmental Sciences, Ministry of Ecology and Environment, Guangzhou 510520, Guangdong Province, China 2 Department of Animal Science, School of Life Science and Enginee- ring, Foshan University, Foshan 528231, Guangdong Province, China Corresponding author: Zhao-Liang Guo ([email protected]) Academic editor: I.S. Wehrtmann | Received 19 November 2019 | Accepted 7 February 2020 | Published 1 April 2020 http://zoobank.org/138A88CC-DF41-437A-BA1A-CB93E3E36D62 Citation: Chen Q-H, Chen W-J, Zheng X-Z, Guo Z-L (2020) Two freshwater shrimp species of the genus Caridina (Decapoda, Caridea, Atyidae) from Dawanshan Island, Guangdong, China, with the description of a new species. ZooKeys 923: 15–32. https://doi.org/10.3897/zookeys.923.48593 Abstract A faunistic and ecological survey was conducted to document the diversity of freshwater atyid shrimps of Dawanshan Island. Two species of Caridina that occur on this island were documented and discussed. One of these, Caridina tetrazona sp. nov. is described and illustrated as new to science. It can be easily distinguished from its congeners based on a combination of characters, which includes a short rostrum, the shape of the endopod of the male first pleopod, the segmental ratios of antennular peduncle and third maxilliped, the slender scaphocerite, and the absence of a median projection on the posterior margin. -
W7192e19.Pdf
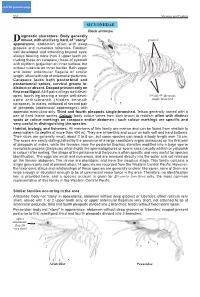
click for previous page 952 Shrimps and Prawns Sicyoniidae SICYONIIDAE Rock shrimps iagnostic characters: Body generally Drobust, with shell very hard, of “stony” grooves appearance; abdomen often with deep grooves and numerous tubercles. Rostrum well developed and extending beyond eyes, always bearing more than 3 upper teeth (in- cluding those on carapace); base of eyestalk with styliform projection on inner surface, but without tubercle on inner border. Both upper and lower antennular flagella of similar length, attached to tip of antennular peduncle. 1 Carapace lacks both postorbital and postantennal spines, cervical groove in- distinct or absent. Exopod present only on first maxilliped. All 5 pairs of legs well devel- 2 oped, fourth leg bearing a single well-devel- 3rd and 4th pleopods 4 single-branched oped arthrobranch (hidden beneath 3 carapace). In males, endopod of second pair 5 of pleopods (abdominal appendages) with appendix masculina only. Third and fourth pleopods single-branched. Telson generally armed with a pair of fixed lateral spines. Colour: body colour varies from dark brown to reddish; often with distinct spots or colour markings on carapace and/or abdomen - such colour markings are specific and very useful in distinguishing the species. Habitat, biology, and fisheries: All members of this family are marine and can be found from shallow to deep waters (to depths of more than 400 m). They are all benthic and occur on both soft and hard bottoms. Their sizes are generally small, about 2 to 8 cm, but some species can reach a body length over 15 cm. The sexes are easily distinguished by the presence of a large copulatory organ (petasma) on the first pair of pleopods of males, while the females have the posterior thoracic sternites modified into a large sperm receptacle process (thelycum) which holds the spermatophores or sperm sacs (usually whitish or yellowish in colour) after mating. -

Checklists of Crustacea Decapoda from the Canary and Cape Verde Islands, with an Assessment of Macaronesian and Cape Verde Biogeographic Marine Ecoregions
Zootaxa 4413 (3): 401–448 ISSN 1175-5326 (print edition) http://www.mapress.com/j/zt/ Article ZOOTAXA Copyright © 2018 Magnolia Press ISSN 1175-5334 (online edition) https://doi.org/10.11646/zootaxa.4413.3.1 http://zoobank.org/urn:lsid:zoobank.org:pub:2DF9255A-7C42-42DA-9F48-2BAA6DCEED7E Checklists of Crustacea Decapoda from the Canary and Cape Verde Islands, with an assessment of Macaronesian and Cape Verde biogeographic marine ecoregions JOSÉ A. GONZÁLEZ University of Las Palmas de Gran Canaria, i-UNAT, Campus de Tafira, 35017 Las Palmas de Gran Canaria, Spain. E-mail: [email protected]. ORCID iD: 0000-0001-8584-6731. Abstract The complete list of Canarian marine decapods (last update by González & Quiles 2003, popular book) currently com- prises 374 species/subspecies, grouped in 198 genera and 82 families; whereas the Cape Verdean marine decapods (now fully listed for the first time) are represented by 343 species/subspecies with 201 genera and 80 families. Due to changing environmental conditions, in the last decades many subtropical/tropical taxa have reached the coasts of the Canary Islands. Comparing the carcinofaunal composition and their biogeographic components between the Canary and Cape Verde ar- chipelagos would aid in: validating the appropriateness in separating both archipelagos into different ecoregions (Spalding et al. 2007), and understanding faunal movements between areas of benthic habitat. The consistency of both ecoregions is here compared and validated by assembling their decapod crustacean checklists, analysing their taxa composition, gath- ering their bathymetric data, and comparing their biogeographic patterns. Four main evidences (i.e. different taxa; diver- gent taxa composition; different composition of biogeographic patterns; different endemicity rates) support that separation, especially in coastal benthic decapods; and these parametres combined would be used as a valuable tool at comparing biotas from oceanic archipelagos. -

Prawn Fauna (Crustacea: Decapoda) of India - an Annotated Checklist of the Penaeoid, Sergestoid, Stenopodid and Caridean Prawns
Available online at: www.mbai.org.in doi: 10.6024/jmbai.2012.54.1.01697-08 Prawn fauna (Crustacea: Decapoda) of India - An annotated checklist of the Penaeoid, Sergestoid, Stenopodid and Caridean prawns E. V. Radhakrishnan*1, V. D. Deshmukh2, G. Maheswarudu3, Jose Josileen 1, A. P. Dineshbabu4, K. K. Philipose5, P. T. Sarada6, S. Lakshmi Pillai1, K. N. Saleela7, Rekhadevi Chakraborty1, Gyanaranjan Dash8, C.K. Sajeev1, P. Thirumilu9, B. Sridhara4, Y Muniyappa4, A.D.Sawant2, Narayan G Vaidya5, R. Dias Johny2, J. B. Verma3, P.K.Baby1, C. Unnikrishnan7, 10 11 11 1 7 N. P. Ramachandran , A. Vairamani , A. Palanichamy , M. Radhakrishnan and B. Raju 1CMFRI HQ, Cochin, 2Mumbai RC of CMFRI, 3Visakhapatnam RC of CMFRI, 4Mangalore RC of CMFRI, 5Karwar RC of CMFRI, 6Tuticorin RC of CMFRI, 7Vizhinjam RC of CMFRI, 8Veraval RC of CMFRI, 9Madras RC of CMFRI, 10Calicut RC of CMFRI, 11Mandapam RC of CMFRI *Correspondence e-mail: [email protected] Received: 07 Sep 2011, Accepted: 15 Mar 2012, Published: 30 Apr 2012 Original Article Abstract Many penaeoid prawns are of considerable value for the fishing Introduction industry and aquaculture operations. The annual estimated average landing of prawns from the fishery in India was 3.98 The prawn fauna inhabiting the marine, estuarine and lakh tonnes (2008-10) of which 60% were contributed by freshwater ecosystems of India are diverse and fairly well penaeid prawns. An additional 1.5 lakh tonnes is produced from known. Significant contributions to systematics of marine aquaculture. During 2010-11, India exported US $ 2.8 billion worth marine products, of which shrimp contributed 3.09% in prawns of Indian region were that of Milne Edwards (1837), volume and 69.5% in value of the total export. -

Crangon Franciscorum Class: Multicrustacea, Malacostraca, Eumalacostraca
Phylum: Arthropoda, Crustacea Crangon franciscorum Class: Multicrustacea, Malacostraca, Eumalacostraca Order: Eucarida, Decapoda, Pleocyemata, Caridea Common gray shrimp Family: Crangonoidea, Crangonidae Taxonomy: Schmitt (1921) described many duncle segment (Wicksten 2011). Inner fla- shrimp in the genus Crago (e.g. Crago fran- gellum of the first antenna is greater than ciscorum) and reserved the genus Crangon twice as long as the outer flagellum (Kuris et for the snapping shrimp (now in the genus al. 2007) (Fig. 2). Alpheus). In 1955–56, the International Mouthparts: The mouth of decapod Commission on Zoological Nomenclature crustaceans comprises six pairs of appendag- formally reserved the genus Crangon for the es including one pair of mandibles (on either sand shrimps only. Recent taxonomic de- side of the mouth), two pairs of maxillae and bate revolves around potential subgeneric three pairs of maxillipeds. The maxillae and designation for C. franciscorum (C. Neocran- maxillipeds attach posterior to the mouth and gon franciscorum, C. franciscorum francis- extend to cover the mandibles (Ruppert et al. corum) (Christoffersen 1988; Kuris and Carl- 2004). Third maxilliped setose and with exo- ton 1977; Butler 1980; Wicksten 2011). pod in C. franciscorum and C. alaskensis (Wicksten 2011). Description Carapace: Thin and smooth, with a Size: Average body length is 49 mm for single medial spine (compare to Lissocrangon males and 68 mm for females (Wicksten with no gastric spines). Also lateral (Schmitt 2011). 1921) (Fig. 1), hepatic, branchiostegal and Color: White, mottled with small black spots, pterygostomian spines (Wicksten 2011). giving gray appearance. Rostrum: Rostrum straight and up- General Morphology: The body of decapod turned (Crangon, Kuris and Carlton 1977). -

ICES Marine Science Symposia
ICES mar. Sei. Symp., 199: 209-221. 1995 Decapod crustaceans in the diets of demersal fish in the Cantabrian Sea I. Olaso and E. Rodriguez-Marin Olaso, I., and Rodriguez-Marin, E. 1995. Decapod crustaceans in the diets of demer sal fish in the Cantabrian Sea. - ICES mar. Sei. Symp., 199: 209-221. The diets of 14 species of demersal fish, representing 81% of the fish biomass of the Cantabrian Sea (ICES Division VIIIc), were determined in the spring and autumn of 1988 from the analysis of 6536 stomachs. Decapod crustaceans represented 54.3% of the diet of these fish in frequency of occurrence and 22.1% in percentage by volume. The main prey taxa were: Solenocera membranacea, Alpeus glaber, the Crangonidae and Processidae families within the Natantia, the Paguridae and Galatheidae families within the Anomura, and Goneplax rhomboides and the Portunidae family within the Brachyura. Predator-prey linkages were described and each predator was assigned to the size group that would best demonstrate any size-related feeding pattern. I. Olaso and E. Rodriguez-Marin: Instituto Espanol de Oceanografia, Laboratorio Oceanogrâfico de Santander. Apdo240, 39080 Santander, Cantabria, Spain [tel: (+34) 42 2740431275033, fax: (+34) 42 275072], Introduction Studies of size, distribution, and abundance of prey and diet selection by predators were conducted because The Bay of Biscay forms a well-defined unit because of these parameters play a fundamental role in the selec its geographical location and semi-enclosed nature. The tion of diet and have the advantage of being easily southern part of this gulf, called the Cantabrian Sea, is a quantified (Murdoch and Oaken, 1975; Vince et al., transition zone and its fish and crustacean inhabitants 1976). -

Antifouling Adaptations of Marine Shrimp (Decapoda: Caridea): Gill Cleaning Mechanisms and Grooming of Brooded Embryos
Zoological Journal oj the Linnean Society, 6i: 281-305. With 12 figures April 1979 Antifouling adaptations of marine shrimp (Decapoda: Caridea): gill cleaning mechanisms and grooming of brooded embryos RAYMOND T. BAUER Biological Sciences, California Polytechnic State University, San Luis Obispo, California, U.S.A. Accepted for publication September 1977 Gills in the branchial chambers of caridean shrimps, as well as the brooded embryos in females, are subject to fouling by particulate debris and epizoites. Important mechanisms for cleaning the gills are brushing of the gills by the grooming or cleaning chelipeds in some species, while in others, setae from the bases of the thoracic legs brush up among the gills during movement of the limbs (epipod- setobranch complexes). Setae of cleaning chelipeds and of epipod-setobranch complexes show- similar ultrastructural adaptions for scraping gill surfaces. Ablation of the cleaning chelipeds ol the shrimp Heptacarpm pictus results in severe fouling of the gills in experimenials, while those of controls remain clean, Embrvos brooded by female carideans are often brushed and jostled by the grooming chelipeds. In H. pictui. removal of the cleaning chelae results in heavier microbial and sediment fouling than in controls. KEY WO RDS: - shrimp - gills - grooming - cleaning - cpipods - sctobranchs - fouling - Decapoda - Caridea. CONTENTS Introduction 281 Methods 282 Results 284 Gill cleaning by the chelipeds 284 Gill cleaning by the epipod-setobranch complex 289 Experiments on the adaptive value of cheliped brushing in Heptacarpus pictus 292 Cleaning of brooded embryos 296 Experiments on the adaptive value of cheliped brushing of eggs in Heptacarpus pictus 297 Discussion 299 Adaptive value of gill cleaning mechanisms in caridean shrimp 299 Adaptive value of embryo brushing by females 301 Acknowledgements 302 References 302 INTRODUCTION Grooming behaviour is a frequent activity of caridean shrimp which appears to prevent epizoic and sediment fouling of the body (Bauer, 1975; Bauer, 1977, Bauer, 1978). -

BIOLÓGICA VENEZUELICA Es Editada Por Dirección Postal De Los Mismos
7 M BIOLÓGICA II VENEZUELICA ^^.«•r-íí-yííT"1 VP >H wv* "V-i-, •^nru-wiA ">^:^;iW SWv^X/^ií. UN I VE RSIDA P CENTRAL DÉ VENEZUELA ^;."rK\'':^>:^:;':••'': ; .-¥•-^>v^:v- ^ACUITAD DE CIENCIAS INSilTÜTO DÉ Z00LOGIA TROPICAL: •RITiTRnTOrr ACTA BIOLÓGICA VENEZUELICA es editada por Dirección postal de los mismos. Deberá suministrar el Instituto de Zoología Tropical, Facultad, de Ciencias se en página aparte el título del trabajo en inglés en de la Universidad Central de Venezuela y tiene por fi caso de no estar el manuscritp elaborado en ese nalidad la publicación de trabajos originales sobre zoo idioma. logía, botánica y ecología. Las descripciones de espe cies nuevas de la flora y fauna venezolanas tendrán Resúmenes: Cada resumen no debe exceder 2 pági prioridad de publicación. Los artículos enviados no de nas tamaño carta escritas a doble espacio. Deberán berán haber sido publicados previamente ni estar sien elaborarse en castellano e ingles, aparecer en este do considerados para tal fin en otras revistas. Los ma mismo orden y en ellos deberá indicarse el objetivo nuscritos deberán elaborarse en castellano o inglés y y los principales resultados y conclusiones de la co no deberán exceder 40 páginas tamaño carta, escritas municación. a doble espacio, incluyendo bibliografía citada, tablas y figuras. Ilustraciones: Todas las ilustraciones deberán ser llamadas "figuras" y numeradas en orden consecuti ACTA BIOLÓGICA VENEZUELICA se edita en vo (Ejemplo Fig. 1. Fig 2a. Fig 3c.) el número, así co cuatro números que constituyen un volumen, sin nin mo también el nombre del autor deberán ser escritos gún compromiso de fecha fija de publicación. -

Zootaxa, Pontoniine Shrimps (Decapoda: Palaemonidae)
Zootaxa 1137: 1–36 (2006) ISSN 1175-5326 (print edition) www.mapress.com/zootaxa/ ZOOTAXA 1137 Copyright © 2006 Magnolia Press ISSN 1175-5334 (online edition) Pontoniine shrimps (Decapoda: Palaemonidae) from the island of Socotra, with descriptions of new species of Dactylonia Fransen, 2002 and Periclimenoides Bruce, 1990 A. J. BRUCE Queensland Museum, P.O. Box 3300, South Brisbane, Australia 4101. E-mail: [email protected] Table of contents Abstract ............................................................................................................................................. 2 Introduction ....................................................................................................................................... 2 Taxonomy .......................................................................................................................................... 2 Conchodytes meleagrinae Peters, 1852 ............................................................................................ 3 Coralliocaris sp. ................................................................................................................................ 3 Dactylonia carinicula sp. nov. .......................................................................................................... 4 Key to the Indo-West Pacific Species of Dactylonia Fransen, 2002 .............................................. 13 Harpiliopsis depressa (Stimpson, 1860) .........................................................................................14 -

Crustacea, Decapoda, Dendrobranchiata and Caridea) from Off Northeastern Japan
Bull. Natl. Mus. Nat. Sci., Ser. A, 42(1), pp. 23–48, February 22, 2016 Additional Records of Deep-water Shrimps (Crustacea, Decapoda, Dendrobranchiata and Caridea) from off Northeastern Japan Tomoyuki Komai1 and Hironori Komatsu2 1 Natural History Museum and Institute, Chiba, 955–2 Aoba-cho, Chiba 260–8682, Japan E-mail: [email protected] 2 Department of Zoology, National Museum of Nature and Science, 4–1–1 Amakubo, Tsukuba, Ibaraki 305–0005, Japan E-mail: [email protected] (Received 5 October 2015; accepted 22 December 2015) Abstract Four deep-water species of shrimps are newly recorded from off Tohoku District, northeastern Japan: Hepomadus gracialis Spence Bate, 1888 (Dendrobranchiata, Aristeidae), Pasiphaea exilimanus Komai, Lin and Chan, 2012 (Caridea, Pasiphaeidae), Nematocarcinus longirostris Spence Bate, 1888 (Caridea, Nematocarcinidae), and Glyphocrangon caecescens Anonymous, 1891 (Caridea, Glyphocrangonidae). Of them, the bathypelagic P. exilimanus is new to the Japanese fauna. The other three species are abyssobenthic, extending to depths greater than 3000 m, and thus have been rarely collected. The newly collected samples of H. gracialis enable us to reassess diagnostic characters of the species. Nematocarcinus longirostris is rediscovered since the original description, and the taxonomy of the species is reviewed. Glyphocrangon caecescens is the sole representative of the family extending to off northern Japan. Key words : Hepomadus gracialis, Pasiphaea exilimanus, Nematocarcinus longirostris, Glypho- crangon caecescens, -

(Caridea: Alpheidae, Palaemonidae) on the Brazilian Coast
An Acad Bras Cienc (2021) 93(2): e20190634 DOI 10.1590/0001-3765202120190634 Anais da Academia Brasileira de Ciências | Annals of the Brazilian Academy of Sciences Printed ISSN 0001-3765 I Online ISSN 1678-2690 www.scielo.br/aabc | www.fb.com/aabcjournal ANIMAL SCIENCE Range extensions of three marine Running title: RANGE shrimps (Caridea: Alpheidae, EXTENSIONS OF THREE CARIDEANS FROM BRAZIL Palaemonidae) on the Brazilian coast LUCIANE A.A. FERREIRA, CECILI B. MENDES & PAULO P.G. PACHELLE Academy Section: ANIMAL Abstract: Three caridean shrimps have their distribution range extended on the SCIENCE Brazilian coast. Alpheus carlae Anker, 2012 (Alpheidae), previously reported from Ceará to São Paulo, and Typton fapespae Almeida, Anker & Mantelatto, 2014 (Palaemonidae), previously known only from Rio de Janeiro and São Paulo, are both now reported from e20190634 Santa Catarina, the new southernmost record of these species in the Atlantic Ocean. Athanas nitescens (Leach, 1813) (Alpheidae), an invasive species from the eastern Atlantic fi rst reported from São Paulo in 2012 based on a single male, is now confi rmed to have 93 established populations in Brazil with the fi nding of ovigerous females on the coast of (2) Rio de Janeiro. Illustrations for all three species are provided based on the new material. 93(2) Key words: Biodiversity, Crustacea, Decapoda, southwestern Atlantic, intertidal. DOI 10.1590/0001-3765202120190634 INTRODUCTION studies have been published dealing with new records, fi lling distributional gaps for various The infraorder Caridea Dana, 1852 comprises the species and thus providing valuable information second most speciose infraorder of decapod for future marine biodiversity assessments (e.g., crustaceans with over 3400 described species Cardoso 2009, Pachelle et al.