Cyanohydrin - Wikipedia, the Free Encyclopedia
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Transport of Dangerous Goods
ST/SG/AC.10/1/Rev.16 (Vol.I) Recommendations on the TRANSPORT OF DANGEROUS GOODS Model Regulations Volume I Sixteenth revised edition UNITED NATIONS New York and Geneva, 2009 NOTE The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of the Secretariat of the United Nations concerning the legal status of any country, territory, city or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries. ST/SG/AC.10/1/Rev.16 (Vol.I) Copyright © United Nations, 2009 All rights reserved. No part of this publication may, for sales purposes, be reproduced, stored in a retrieval system or transmitted in any form or by any means, electronic, electrostatic, magnetic tape, mechanical, photocopying or otherwise, without prior permission in writing from the United Nations. UNITED NATIONS Sales No. E.09.VIII.2 ISBN 978-92-1-139136-7 (complete set of two volumes) ISSN 1014-5753 Volumes I and II not to be sold separately FOREWORD The Recommendations on the Transport of Dangerous Goods are addressed to governments and to the international organizations concerned with safety in the transport of dangerous goods. The first version, prepared by the United Nations Economic and Social Council's Committee of Experts on the Transport of Dangerous Goods, was published in 1956 (ST/ECA/43-E/CN.2/170). In response to developments in technology and the changing needs of users, they have been regularly amended and updated at succeeding sessions of the Committee of Experts pursuant to Resolution 645 G (XXIII) of 26 April 1957 of the Economic and Social Council and subsequent resolutions. -

Cyanovinylation of Aldehydes: Organocatalytic Multicomponent Synthesis of Conjugated Cyanomethyl Vinyl Ethers
molecules Article Cyanovinylation of Aldehydes: Organocatalytic Multicomponent Synthesis of Conjugated Cyanomethyl Vinyl Ethers Samuel Delgado-Hernández 1,2 , Fernando García-Tellado 1,* and David Tejedor 1,* 1 Instituto de Productos Naturales y Agrobiología, Consejo Superior de Investigaciones Científicas, Astrofísico Francisco Sánchez 3, 38206 La Laguna, Spain; [email protected] 2 Doctoral and Postgraduate School, Universidad de La Laguna, Apartado Postal 456, 38200 La Laguna, Spain * Correspondence: [email protected] (F.G.-T.); [email protected] (D.T.) Abstract: A novel organocatalytic multicomponent cyanovinylation of aldehydes was designed for the synthesis of conjugated cyanomethyl vinyl ethers. The reaction was implemented for the synthesis of a 3-substituted 3-(cyanomethoxy)acrylates, using aldehydes as substrates, acetone cyanohydrin as the cyanide anion source, and methyl propiolate as the source of the vinyl com- ponent. The multicomponent reaction is catalyzed by N-methyl morpholine (2.5 mol%) to deliver the 3-(cyanomethoxy)acrylates in excellent yields and with preponderance of the E-isomer. The multicomponent reaction manifold is highly tolerant to the structure and composition of the aldehyde (aliphatic, aromatic, heteroaromatics), and it is instrumentally simple (one batch, open atmospheres), economic (2.5 mol% catalyst, stoichiometric reagents), environmentally friendly (no toxic waste), and Citation: Delgado-Hernández, S.; sustainable (easy scalability). García-Tellado, F.; Tejedor, D. Cyanovinylation of -

Assessment of Portable HAZMAT Sensors for First Responders
The author(s) shown below used Federal funds provided by the U.S. Department of Justice and prepared the following final report: Document Title: Assessment of Portable HAZMAT Sensors for First Responders Author(s): Chad Huffman, Ph.D., Lars Ericson, Ph.D. Document No.: 246708 Date Received: May 2014 Award Number: 2010-IJ-CX-K024 This report has not been published by the U.S. Department of Justice. To provide better customer service, NCJRS has made this Federally- funded grant report available electronically. Opinions or points of view expressed are those of the author(s) and do not necessarily reflect the official position or policies of the U.S. Department of Justice. Assessment of Portable HAZMAT Sensors for First Responders DOJ Office of Justice Programs National Institute of Justice Sensor, Surveillance, and Biometric Technologies (SSBT) Center of Excellence (CoE) March 1, 2012 Submitted by ManTech Advanced Systems International 1000 Technology Drive, Suite 3310 Fairmont, West Virginia 26554 Telephone: (304) 368-4120 Fax: (304) 366-8096 Dr. Chad Huffman, Senior Scientist Dr. Lars Ericson, Director UNCLASSIFIED This project was supported by Award No. 2010-IJ-CX-K024, awarded by the National Institute of Justice, Office of Justice Programs, U.S. Department of Justice. The opinions, findings, and conclusions or recommendations expressed in this publication are those of the author(s) and do not necessarily reflect those of the Department of Justice. This document is a research report submitted to the U.S. Department of Justice. This report has not been published by the Department. Opinions or points of view expressed are those of the author(s) and do not necessarily reflect the official position or policies of the U.S. -
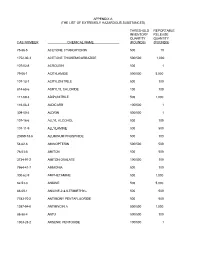
The List of Extremely Hazardous Substances)
APPENDIX A (THE LIST OF EXTREMELY HAZARDOUS SUBSTANCES) THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 75-86-5 ACETONE CYANOHYDRIN 500 10 1752-30-3 ACETONE THIOSEMICARBAZIDE 500/500 1,000 107-02-8 ACROLEIN 500 1 79-06-1 ACRYLAMIDE 500/500 5,000 107-13-1 ACRYLONITRILE 500 100 814-68-6 ACRYLYL CHLORIDE 100 100 111-69-3 ADIPONITRILE 500 1,000 116-06-3 ALDICARB 100/500 1 309-00-2 ALDRIN 500/500 1 107-18-6 ALLYL ALCOHOL 500 100 107-11-9 ALLYLAMINE 500 500 20859-73-8 ALUMINUM PHOSPHIDE 500 100 54-62-6 AMINOPTERIN 500/500 500 78-53-5 AMITON 500 500 3734-97-2 AMITON OXALATE 100/500 100 7664-41-7 AMMONIA 500 100 300-62-9 AMPHETAMINE 500 1,000 62-53-3 ANILINE 500 5,000 88-05-1 ANILINE,2,4,6-TRIMETHYL- 500 500 7783-70-2 ANTIMONY PENTAFLUORIDE 500 500 1397-94-0 ANTIMYCIN A 500/500 1,000 86-88-4 ANTU 500/500 100 1303-28-2 ARSENIC PENTOXIDE 100/500 1 THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 1327-53-3 ARSENOUS OXIDE 100/500 1 7784-34-1 ARSENOUS TRICHLORIDE 500 1 7784-42-1 ARSINE 100 100 2642-71-9 AZINPHOS-ETHYL 100/500 100 86-50-0 AZINPHOS-METHYL 10/500 1 98-87-3 BENZAL CHLORIDE 500 5,000 98-16-8 BENZENAMINE, 3-(TRIFLUOROMETHYL)- 500 500 100-14-1 BENZENE, 1-(CHLOROMETHYL)-4-NITRO- 500/500 500 98-05-5 BENZENEARSONIC ACID 10/500 10 3615-21-2 BENZIMIDAZOLE, 4,5-DICHLORO-2-(TRI- 500/500 500 FLUOROMETHYL)- 98-07-7 BENZOTRICHLORIDE 100 10 100-44-7 BENZYL CHLORIDE 500 100 140-29-4 BENZYL CYANIDE 500 500 15271-41-7 BICYCLO[2.2.1]HEPTANE-2-CARBONITRILE,5- -

Investigation of Cyano·Methylation Reaction by Cyano·Hydrine and Its Determination in Tobacco-Smoke. ( Strecker·Reactions )
PERIODICA POLYTECHNICA SER. CHEM. ENG. VOL. 36, NO. 3, PP. 209-2113 {I992} INVESTIGATION OF CYANO·METHYLATION REACTION BY CYANO·HYDRINE AND ITS DETERMINATION IN TOBACCO-SMOKE. ( STRECKER·REACTIONS ) 3 Vikt6ria HORVATH,l Lajos TREZLl, Tibor SZARVAS2, Janos PIPEK , Csaba VIDA 1 and Krisztina BAUER4 1 Department of Organic Chemical Technology, 3Department of Physics Technical University of Budapest, 2 Institute of Isotopes of the Hungarian Academy of Sciences 4 Plant Protection Institute,Hungarian Academy of Sciences. Received: September 9, 1992 Abstract The results of our research work [1-7) of several years aimed at the binding of the unhealthy components of tobacco smoke directed our attention to the analysis and identification of the unknown peak observed during the high sensitivity radiochromatographic analysis of tobacco smoke condensates.It was a fruitless effort to remove the formaldehyde, the ac etaldehyde and hydrogen cyanide present in tobacco smoke, or even to try to eliminate them separately, during the analysis the new, unknown peak appeared again and again on the radiochromatogram. During our research work we stated, that we were facing the Strecker reaction [8-9), known since 1850, that is,with the formation of cyanohydrine of the aldehydes and hydrogen cyanide present in tobacco smoke, corresponding to the new peak. Cyanohydrines to react quickly and energetically with basic aminoacids, during which reaction cyanomethyl derivatives are being formed. This reaction is proceeding also under physiological circumstances with the basic amino groups of proteins, contributing to the development of respiratory and cardiovascular insufficiencies. By means of model reactions and using solutions of tobacco smoke gases the process of the reaction was proved in a primary way, also its circumstances having been cleared. -

CO2-Enabled Cyanohydrin Synthesis and Facile Homologation Reactions Martin Juhl†, Allan R
CO2-Enabled Cyanohydrin Synthesis and Facile Homologation Reactions Martin Juhl†, Allan R. Petersen†, Ji-Woong Lee*,† †Department of Chemistry, Nano-Science Center, University of Copenhagen, Universitetsparken 5, Copenhagen Ø, 2100, Denmark KEYWORDS: Carbon Dioxide, Cyanohydrin, Xanthate, Homologation ABSTRACT: Thermodynamic and kinetic control of a chemical process is the key to access desired products and states. Changes are made when desired product is not accessible; one may manipulate the reaction with additional reagents, catalysts and/or protect- ing groups. Here we report the use of carbon dioxide to direct reaction pathways in order to selectively afford desired products in high reaction rates while avoiding the formation of byproducts. The utility of CO2-mediated selective cyanohydrin synthesis was further showcased by broadening Kiliani-Fischer synthesis to offer an easy access to variety of polyols, cyanohydrins, linear al- kylnitriles, by simply starting from alkyl- and arylaldehydes, KCN and atmospheric pressure of CO2. 9 A chemical reaction is governed by kinetics and ther- investigated the role of CO2 in a cyanation reaction, where CO2 modynamics, and a simultaneous control of both parameters is can be used in catalytic amounts to facilitate the stereoselective a common practice in designing and optimizing chemical reac- transformation of activated electrophiles via 1,4-conjugate ad- tions. The manipulation of thermodynamic stability of reactants dition reactions. Cyanohydrin synthesis - 1,2-cyanide addition and products -

Bowman Chem 345 Lecture Notes by Topic Cyanohydrin
Bowman Chem 345 Lecture Notes by Topic Cyanohydrin: Important approximate pKa’s to know for this section: pKa ~ 10 pKa ~ -5 pKa ~ 0 pKa ~ 5 pKa ~ 15 + H H O HCl H3O O 2 H SO 2 4 N R ROH R H OH R R=H or sp3 C O H C N Cyanide is a moderate base (pKa of HCN ~ 10). It is also a good nucleophile. Like Grignards and organolithiums, it can add to carbonyls. Standard conditions for cyanohydrin formation is a cyanide salt and a weak acid like acetic acid.1 KCN CN O cyanohydrin AcOH racemic OH O AcOH=acetic acid= OH Mechanism: The first step is cyanide attacking the carbonyl. Though this is not a favorable reaction pKa wise, it is still possible as an alcohol and HCN have very close pKa’s (the difference is ≤ 6). The second step is favorable as acetate is a weaker base than an alkoxide (negative O). This step you can write as reversible or not (though it is on the boundary reversible/irreversible boundary (>6 but ≤ 10). [Technically, cyanohydrins are slightly more acidic (pKa~13) than normal alcohols due to the inductive effect of the nitrile. One of the most common mistakes in this mechanism is to protonate the carbonyl. Though technically, acetic acid can barely protonate the carbonyl, the cyanide being a stronger base than acetate would rather irreversibly deprotonate a protonated carbonyl than add to the carbonyl carbon. 1 Brunner, A.; Hintermann, L. Chem. Euro. J. 2016, 22, 2787-2792 Bowman Chem 345 Lecture Notes by Topic Unlike Grignard and organolithium reactions, the cyanohydrin formation can be reversed. -

Process for Producing Methyl Methacrylate Verfahren Zur Herstellung Von Methylmethacrylat Procede Pour La Fabrication De Methacrylate De Methyle
~™ II 1 1 III II II 1 1 II II I Ml II I II I II (19) J European Patent Office Office europeen des brevets (11) EP 0 406 676 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publicationation and mention (51 ) |nt. CI.6: C07C 69/54, C07C 67/20 of the grant of the patent: 27.03.1996 Bulletin 1996/13 (21) Application number: 90112194.7 (22) Date of filing: 27.06.1990 (54) Process for producing methyl methacrylate Verfahren zur Herstellung von Methylmethacrylat Procede pour la fabrication de methacrylate de methyle (84) Designated Contracting States: • Ebata, Shuji, DE ES FR GB IT NL C/o Mitsubishi Gas Chem. Com. Tayuhama, Niigata-shi, Niigata-ken (JP) (30) Priority: 04.07.1989 JP 171190/89 (74) Representative: Turk, Gille, Hrabal, Leifert (43) Date of publication of application: Brucknerstrasse 20 09.01.1991 Bulletin 1991/02 D-40593 Dusseldorf (DE) (73) Proprietor: MITSUBISHI GAS CHEMICAL (56) References cited: COMPANY, INC. DE-A- 3 436 608 Chiyoda-ku, Tokyo (JP) • PATENT ABSTRACTS OF JAPAN vol. 14, no. 68 (72) Inventors: (C- 686)(401 1 ), 8 February 1 990; & JP-A-1 290653 • Higuchi, Hirofumi, (MITSUBISHI GAS CHEM) 22.11. 1989 C/o Mitsubishi Gas Chem. Com. Tayuhama, Niigata-shi, Niigata-ken (JP) Remarks: • Kida, Koichi, The file contains technical information submitted C/o Mitsubishi Gas Chem. Com. after the application was filed and not included in this Tayuhama, Niigata-shi, Niigata-ken (JP) specification CO CO CO o Note: Within nine months from the publication of the mention of the grant of the European patent, any person may give notice to the European Patent Office of opposition to the European patent granted. -

Provisional Peer-Reviewed Toxicity Values for Acetone Cyanohydrin (Casrn 75-86-5)
EPA/690/R-12/001F l Final 10-01-2012 Provisional Peer-Reviewed Toxicity Values for Acetone Cyanohydrin (CASRN 75-86-5) Superfund Health Risk Technical Support Center National Center for Environmental Assessment Office of Research and Development U.S. Environmental Protection Agency Cincinnati, OH 45268 AUTHORS, CONTRIBUTORS, AND REVIEWERS CHEMICAL MANAGER Scott C. Wesselkamper, PhD National Center for Environmental Assessment, Cincinnati, OH DRAFT DOCUMENT PREPARED BY ICF International 9300 Lee Highway Fairfax, VA 22031 PRIMARY INTERNAL REVIEWERS Zheng (Jenny) Li, PhD, DABT National Center for Environmental Assessment, Washington, DC Anuradha Mudipalli, MSc, PhD National Center for Environmental Assessment, Research Triangle Park, NC This document was externally peer reviewed under contract to Eastern Research Group, Inc. 110 Hartwell Avenue Lexington, MA 02421-3136 Questions regarding the contents of this document may be directed to the U.S. EPA Office of Research and Development’s National Center for Environmental Assessment, Superfund Health Risk Technical Support Center (513-569-7300). ii Acetone cyanohydrin TABLE OF CONTENTS COMMONLY USED ABBREVIATIONS ................................................................................... iv BACKGROUND .............................................................................................................................1 DISCLAIMERS ...............................................................................................................................1 QUESTIONS REGARDING -
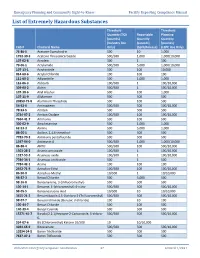
List of Extremely Hazardous Substances
Emergency Planning and Community Right-to-Know Facility Reporting Compliance Manual List of Extremely Hazardous Substances Threshold Threshold Quantity (TQ) Reportable Planning (pounds) Quantity Quantity (Industry Use (pounds) (pounds) CAS # Chemical Name Only) (Spill/Release) (LEPC Use Only) 75-86-5 Acetone Cyanohydrin 500 10 1,000 1752-30-3 Acetone Thiosemicarbazide 500/500 1,000 1,000/10,000 107-02-8 Acrolein 500 1 500 79-06-1 Acrylamide 500/500 5,000 1,000/10,000 107-13-1 Acrylonitrile 500 100 10,000 814-68-6 Acrylyl Chloride 100 100 100 111-69-3 Adiponitrile 500 1,000 1,000 116-06-3 Aldicarb 100/500 1 100/10,000 309-00-2 Aldrin 500/500 1 500/10,000 107-18-6 Allyl Alcohol 500 100 1,000 107-11-9 Allylamine 500 500 500 20859-73-8 Aluminum Phosphide 500 100 500 54-62-6 Aminopterin 500/500 500 500/10,000 78-53-5 Amiton 500 500 500 3734-97-2 Amiton Oxalate 100/500 100 100/10,000 7664-41-7 Ammonia 500 100 500 300-62-9 Amphetamine 500 1,000 1,000 62-53-3 Aniline 500 5,000 1,000 88-05-1 Aniline, 2,4,6-trimethyl- 500 500 500 7783-70-2 Antimony pentafluoride 500 500 500 1397-94-0 Antimycin A 500/500 1,000 1,000/10,000 86-88-4 ANTU 500/500 100 500/10,000 1303-28-2 Arsenic pentoxide 100/500 1 100/10,000 1327-53-3 Arsenous oxide 100/500 1 100/10,000 7784-34-1 Arsenous trichloride 500 1 500 7784-42-1 Arsine 100 100 100 2642-71-9 Azinphos-Ethyl 100/500 100 100/10,000 86-50-0 Azinphos-Methyl 10/500 1 10/10,000 98-87-3 Benzal Chloride 500 5,000 500 98-16-8 Benzenamine, 3-(trifluoromethyl)- 500 500 500 100-14-1 Benzene, 1-(chloromethyl)-4-nitro- 500/500 -

Processes for Producing Alpha-Cyanohydrin Esters and Alpha-Hydroxy Acids
Patentamt Europaisches |||| ||| 1 1|| ||| ||| || || ||| ||| || ||| || || || (19) J European Patent Office Office europeen des brevets (11) EP 0 926 1 34 A2 (12) EUROPEAN PATENT APPLICATION (43) Date of publication:ation: (51) Int. CI.6: C07C 253/08 30.06.1999 Bulletin 1999/26 (21) Application number: 98123461.0 (22) Dateof filing: 11.12.1998 (84) Designated Contracting States: (72) Inventors: AT BE CH CY DE DK ES Fl FR GB GR IE IT LI LU • Ishii, Yasutaka MC NL PT SE Takatsuki.shi, Osaka 569-1 1 1 2 (JP) Designated Extension States: • Nakano, Tatsuya AL LT LV MK RO SI Himeji-shi, Hyogo 670-0094 (JP) (30) Priority: 22.12.1997 JP 35339597 (74) Representative: Grunecker, Kinkeldey, (71) Applicant: Stockmair & Schwanhausser Daicel Chemical Industries, Ltd. Anwaltssozietat Osaka 590-8501 (JP) Maximilianstrasse 58 80538 Munchen (DE) (54) Processes for producing alpha-cyanohydrin esters and alpha-hydroxy acids (57) The present invention relates to a process for the preparation of compounds of formula (4) wherein a carbonyl compound of the formula (3) is reacted in the presence of a cyanogenation agent with an enol ester of the formula (1) or with an oxime ester of the formula (2): s CO 1 ^s r1— c-o-c-cC. (2) O CN O 7 F K , R7—C-R8 (3) R C— O-C— R (4) wherein R1 through R8 have meanings given in the application. The compound of formula (4) can be hydrolysed to obtain the corresponding compound of formula (5): fl7— C-OH (5) CM < CO CO CM <7> O Q_ LU Printed by Xerox (UK) Business Services 2.16.7/3.6 EP 0 926 134 A2 Description FIELD OF THE INVENTION 5 [0001] The present invention relates to processes for producing an a-cyanohydrin ester and an a-hydroxy acid, or salts thereof using an enol ester compound or an oxime ester compound, a carbonyl compound and a cyanogenation agent. -

Stereoselective Synthesis of Spirooxindole Amides and Cyanohydrin Alkyl Ethers by Chunliang Lu B.S., China University of Mining
Stereoselective Synthesis of Spirooxindole Amides and Cyanohydrin Alkyl Ethers by Chunliang Lu B.S., China University of Mining and Technology, Xuzhou, China, 2004 M.S., Dalian University of Technology, Dalian, China, 2007 Submitted to the Graduate Faculty of the Dietrich School of Arts and Sciences in partial fulfillment of the requirements for the degree of Doctor of Philosophy University of Pittsburgh 2013 UNIVERSITY OF PITTSBURGH DIETRICH SCHOOL OF ARTS AND SCIENCES This dissertation was presented by Chunliang Lu It was defended on October 1, 2013 and approved by Dr. Kay M. Brummond, Professor, Department of Chemistry Dr. Theodore Cohen, Professor, Department of Chemistry Dr. Xiang-Qun Xie, Professor, Department of Pharmaceutical Sciences Dissertation Advisor: Dr. Paul E. Floreancig, Professor, Department of Chemistry ii Copyright © by Chunliang Lu 2013 iii Stereoselective Synthesis of Spirooxindole Amides and Cyanohydrin Alkyl Ethers Chunliang Lu, PhD University of Pittsburgh, 2013 A new family of spirooxindole amides were synthesized by a sequence of hydrozirconation, acylation, and intramolecular cyclization reactions. Three of the four possible diastereomers can be obtained as the major isomers through this process. The spirooxindole structure has many points for diversification, and a 37-membered library was synthesized through this approach by collaborators. A comparison with known compound collections showed that this new spirooxindole library possessed good chemical diversity. Cyanohydrin alkyl ethers, the key intermediate in the above multi-component hydrozironation reaction, were effectively synthesized through a Brønsted acid-mediated hydrocyanation of vinyl ethers. The enantiomerically enriched product can be obtained by asymmetric hydrocyanation of vinyl ethers catalyzed by a chiral Brønsted acid, and the catalyst can be regenerated by PhOH.