Calcium Channel
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

K+ Channel Modulators Product ID Product Name Description D3209 Diclofenac Sodium Salt NSAID; COX-1/2 Inhibitor, Potential K+ Channel Modulator
K+ Channel Modulators Product ID Product Name Description D3209 Diclofenac Sodium Salt NSAID; COX-1/2 inhibitor, potential K+ channel modulator. G4597 18β-Glycyrrhetinic Acid Triterpene glycoside found in Glycyrrhiza; 15-HPGDH inhibitor, hERG and KCNA3/Kv1.3 K+ channel blocker. A4440 Allicin Organosulfur found in garlic, binds DNA; inwardly rectifying K+ channel activator, L-type Ca2+ channel blocker. P6852 Propafenone Hydrochloride β-adrenergic antagonist, Kv1.4 and K2P2 K+ channel blocker. P2817 Phentolamine Hydrochloride ATP-sensitive K+ channel activator, α-adrenergic antagonist. P2818 Phentolamine Methanesulfonate ATP-sensitive K+ channel activator, α-adrenergic antagonist. T7056 Troglitazone Thiazolidinedione; PPARγ agonist, ATP-sensitive K+ channel blocker. G3556 Ginsenoside Rg3 Triterpene saponin found in species of Panax; γ2 GABA-A agonist, Kv7.1 K+ channel activator, α10 nAChR antagonist. P6958 Protopanaxatriol Triterpene sapogenin found in species of Panax; GABA-A/C antagonist, slow-activating delayed rectifier K+ channel blocker. V3355 Vindoline Semi-synthetic vinca alkaloid found in Catharanthus; Kv2.1 K+ channel blocker and H+/K+ ATPase inhibitor. A5037 Amiodarone Hydrochloride Voltage-gated Na+, Ca2+, K+ channel blocker, α/β-adrenergic antagonist, FIASMA. B8262 Bupivacaine Hydrochloride Monohydrate Amino amide; voltage-gated Na+, BK/SK, Kv1, Kv3, TASK-2 K+ channel inhibitor. C0270 Carbamazepine GABA potentiator, voltage-gated Na+ and ATP-sensitive K+ channel blocker. C9711 Cyclovirobuxine D Found in Buxus; hERG K+ channel inhibitor. D5649 Domperidone D2/3 antagonist, hERG K+ channel blocker. G4535 Glimepiride Sulfonylurea; ATP-sensitive K+ channel blocker. G4634 Glipizide Sulfonylurea; ATP-sensitive K+ channel blocker. I5034 Imiquimod Imidazoquinoline nucleoside analog; TLR-7/8 agonist, KCNA1/Kv1.1 and KCNA2/Kv1.2 K+ channel partial agonist, TREK-1/ K2P2 and TRAAK/K2P4 K+ channel blocker. -

Therapeutic Potential of RQ-00311651, a Novel T-Type Ca
Research Paper Therapeutic potential of RQ-00311651, a novel T-type Ca21 channel blocker, in distinct rodent models for neuropathic and visceral pain Fumiko Sekiguchia, Yuma Kawaraa, Maho Tsubotaa, Eri Kawakamia, Tomoka Ozakia, Yudai Kawaishia, Shiori Tomitaa, Daiki Kanaokaa, Shigeru Yoshidab, Tsuyako Ohkuboc, Atsufumi Kawabataa,* Abstract 21 T-type Ca channels (T channels), particularly Cav3.2 among the 3 isoforms, play a role in neuropathic and visceral pain. We thus characterized the effects of RQ-00311651 (RQ), a novel T-channel blocker, in HEK293 cells transfected with human Cav3.1 or 21 Cav3.2 by electrophysiological and fluorescent Ca signaling assays, and also evaluated the antiallodynic/antihyperalgesic activity of RQ in somatic, visceral, and neuropathic pain models in rodents. RQ-00311651 strongly suppressed T currents when tested at holding potentials of 265 ; 260 mV, but not 280 mV, in the Cav3.1- or Cav3.2-expressing cells. RQ-00311651 also inhibited high K1-induced Ca21 signaling in those cells. In mice, RQ, administered intraperitoneally (i.p.) at 5 to 20 mg/kg or orally at 20 to 40 mg/kg, significantly suppressed the somatic hyperalgesia and visceral pain-like nociceptive behavior/referred hyperalgesia caused by intraplantar and intracolonic administration of NaHS or Na2S, H2S donors, respectively, which involve the enhanced activity of Cav3.2 channels. RQ-00311651, given i.p. at 5 to 20 mg/kg, exhibited antiallodynic or antihyperalgesic activity in rats with spinal nerve injury–induced neuropathy or in rats and mice with paclitaxel-induced neuropathy. Oral and i.p. RQ at 10 to 20 mg/kg also suppressed the visceral nociceptive behavior and/or referred hyperalgesia accompanying cerulein-induced acute pancreatitis and cyclophosphamide-induced cystitis in mice. -

Z944: an Oral T-Type Calcium Channel Modulator for the Treatment of Pain Margaret S
Z944: An oral T-type calcium channel modulator for the treatment of pain Margaret S. Lee, PhD Ion Channel Retreat 2014, June 25, 2014 T-type Calcium Channels: A Novel Target for Pain and Other CNS Disorders • T-type calcium channels are voltage gated and comprised of three subtypes: Cav3.1, Cav3.2 & Cav3.3 • Expressed in Central and Peripheral Nervous System, including primary afferent, dorsal horn neurons, thalamus and somatosensory cortex • Contribute to neuronal excitability, synaptic excitation, burst firing and action potential trains, and also lower threshold for action potentials Pain Signaling Thalamocortical Connectivity Source: Adapted from Zamponi, et al., Brain Res. Reviews. 2009 Source: Adapted from Park, et al., Frontiers Neural Circuits. 2013 • Rodent neuropathic and IBS pain models exhibit increased • Thalamocortical dysrythmia linked to CNS indications, e.g. T-type current density motor, neuropsychiatric and chronic pain syndromes • Gene knockout or antisense reduces pain in neuropathic, • Mutations in T-type channels are found in rodent and acute and visceral pain models human excitability disorders • T-type channel blockers attenuate neuropathic, • Approved anti-convulsants (e.g. ethosuximide, valproate) inflammatory, acute and visceral pain in animal models target T-type channels © Copyright Neuromed 2 Z944 is a Potent, Selective Blocker of T-type Calcium Channels IC50 (nM) Channel 30% Fold-Selectivity Closed Inactivated (30% Inactivated) CaV3.1(human, exogenous) 50 130 1 CaV3.2 (human, exogenous) 160 540 3.2 CaV3.3 (human, exogenous) 110 260 2.2 N-type (rat, exogenous) 11,000 150,000 220 L-type cardiac calcium (rat CaV1.2) 32,000 -- 640 Cardiac Sodium (human NaV1.5) 100,000 -- 2000 hERG channel (human) 7,800 -- 156 • Displays enhanced potency for the inactivated state across T-type channels • Z944 block of Cav3.2 is more pronounced during high-frequency firing • Z944 has >150-fold selectivity vs. -

GABA Receptors
D Reviews • BIOTREND Reviews • BIOTREND Reviews • BIOTREND Reviews • BIOTREND Reviews Review No.7 / 1-2011 GABA receptors Wolfgang Froestl , CNS & Chemistry Expert, AC Immune SA, PSE Building B - EPFL, CH-1015 Lausanne, Phone: +41 21 693 91 43, FAX: +41 21 693 91 20, E-mail: [email protected] GABA Activation of the GABA A receptor leads to an influx of chloride GABA ( -aminobutyric acid; Figure 1) is the most important and ions and to a hyperpolarization of the membrane. 16 subunits with γ most abundant inhibitory neurotransmitter in the mammalian molecular weights between 50 and 65 kD have been identified brain 1,2 , where it was first discovered in 1950 3-5 . It is a small achiral so far, 6 subunits, 3 subunits, 3 subunits, and the , , α β γ δ ε θ molecule with molecular weight of 103 g/mol and high water solu - and subunits 8,9 . π bility. At 25°C one gram of water can dissolve 1.3 grams of GABA. 2 Such a hydrophilic molecule (log P = -2.13, PSA = 63.3 Å ) cannot In the meantime all GABA A receptor binding sites have been eluci - cross the blood brain barrier. It is produced in the brain by decarb- dated in great detail. The GABA site is located at the interface oxylation of L-glutamic acid by the enzyme glutamic acid decarb- between and subunits. Benzodiazepines interact with subunit α β oxylase (GAD, EC 4.1.1.15). It is a neutral amino acid with pK = combinations ( ) ( ) , which is the most abundant combi - 1 α1 2 β2 2 γ2 4.23 and pK = 10.43. -

Dorset Medicines Advisory Group
DORSET CARDIOLOGY WORKING GROUP GUIDELINE FOR CALCIUM CHANNEL BLOCKERS IN HYPERTENSION SUMMARY The pan-Dorset cardiology working group continues to recommend the use of amlodipine (a third generation dihydropyridine calcium-channel blocker) as first choice calcium channel blocker on the pan-Dorset formulary for hypertension. Lercanidipine is second choice, lacidipine third choice and felodipine is fourth choice. This is due to preferable side effect profiles in terms of ankle oedema and relative costs of the preparations. Note: where angina is the primary indication or is a co-morbidity prescribers must check against the specific product characteristics (SPC) for an individual drug to confirm this is a licensed indication. N.B. Lacidipine and lercandipine are only licensed for use in hypertension. Chapter 02.06.02 CCBs section of the Formulary has undergone an evidence-based review. A comprehensive literature search was carried out on NHS Evidence, Medline, EMBASE, Cochrane Database, and UK Duets. This was for recent reviews or meta-analyses on calcium channel blockers from 2009 onwards (comparative efficacy and side effects) and randomised controlled trials (RCTs). REVIEW BACKGROUND Very little good quality evidence exists. No reviews, meta-analyses or RCTs were found covering all calcium channel blockers currently on the formulary. Another limitation was difficulty obtaining full text original papers for some of the references therefore having to use those from more obscure journals instead. Some discrepancies exist between classification of generations of dihydropyridine CCBs, depending upon the year of publication of the reference/authors’ interpretation. Dihydropyridine (DHP) CCBs tend to be more potent vasodilators than non-dihydropyridine (non-DHP) CCBs (diltiazem, verapamil), but the latter have greater inotropic effects. -

Flufenamic Acid, Mefenamic Acid and Niflumic Acid Inhibit Single Nonselective Cation Channels in the Rat Exocrine Pancreas
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector Volume 268, number 1, 79-82 FEBS 08679 July 1990 Flufenamic acid, mefenamic acid and niflumic acid inhibit single nonselective cation channels in the rat exocrine pancreas H. Gagelein*, D. Dahlem, H.C. Englert** and H.J. Lang** Max-Planck-Institutfir Biophysik, Kennedyallee 70, D-600 Frankfurt/Main 70, FRG Received 17 May 1990 The non-steroidal anti-inflammatory drugs, flufenamic acid, mefenamic acid and niflumic acid, block Ca2+-activated non-selective cation channels in inside-out patches from the basolateral membrane of rat exocrine pancreatic cells. Half-maximal inhibition was about 10 PM for flufenamic acid and mefenamic acid, whereas niflumic acid was less potent (IC,, about 50 PM). Indomethacin, aspirin, diltiazem and ibuprofen (100 /IM) had not effect. It is concluded that the inhibitory effect of flufenamate, mefenamate and niflumate is dependent on the specific structure, consisting of two phenyl rings linked by an amino bridge. Mefenamic acid; Flufenamic acid; Niflumic acid; Indomethacin; Non-selective cation channel; Rat exocrine pancreas 1. INTRODUCTION indomethacin, ibuprofen, diltiazem and acetylsalicylic acid (aspirin) were obtained from Sigma (Munich, FRG). The substances were dissolved in dimethylsulfoxide (DMSO, Merck, Darmstadt, FRG, Recently it was reported that the drug, 3 ’ ,5-dichloro- 0.1% of total volume) before addition to the measuring solution. diphenylamine-2-carboxylic acid (DCDPC), blocks DMSO alone had no effect on the single channel recordings. non-selective cation channels in the basolateral mem- 2.2. Methods brane of rat exocrine pancreatic cells [ 11. -

Synthesis and Pharmacological Evaluation of Fenamate Analogues: 1,3,4-Oxadiazol-2-Ones and 1,3,4- Oxadiazole-2-Thiones
Scientia Pharmaceutica (Sci. Pharm.) 71,331-356 (2003) O Osterreichische Apotheker-Verlagsgesellschaft m. b.H., Wien, Printed in Austria Synthesis and Pharmacological Evaluation of Fenamate Analogues: 1,3,4-Oxadiazol-2-ones and 1,3,4- Oxadiazole-2-thiones Aida A. ~l-~zzoun~'*,Yousreya A ~aklad',Herbert ~artsch~,~afaaA. 2aghary4, Waleed M. lbrahims, Mosaad S. ~oharned~. Pharmaceutical Sciences Dept. (Pharmaceutical Chemistry goup' and Pharmacology group2), National Research Center, Tahrir St. Dokki, Giza, Egypt. 3~nstitutflir Pharmazeutische Chemie, Pharrnazie Zentrum der Universitilt Wien. 4~harmaceuticalChemistry Dept. ,' Organic Chemistry Dept. , Helwan University , Faculty of Pharmacy, Ein Helwan Cairo, Egypt. Abstract A series of fenamate pyridyl or quinolinyl analogues of 1,3,4-oxadiazol-2-ones 5a-d and 6a-r, and 1,3,4-oxadiazole-2-thiones 5e-g and 6s-v, respectively, have been synthesized and evaluated for their analgesic (hot-plate) , antiinflammatory (carrageenin induced rat's paw edema) and ulcerogenic effects as well as plasma prostaglandin E2 (PGE2) level. The highest analgesic activity was achieved with compound 5a (0.5 ,0.6 ,0.7 mrnolkg b.wt.) in respect with mefenamic acid (0.4 mmollkg b.wt.). Compounds 6h, 61 and 5g showed 93, 88 and 84% inhibition, respectively on the carrageenan-induced rat's paw edema at dose level of O.lrnrnol/kg b.wt, compared with 58% inhibition of mefenamic acid (0.2mmoll kg b.wt.). Moreover, the highest inhibitory activity on plasma PGE2 level was displayed also with 6h, 61 and 5g (71, 70,68.5% respectively, 0.lmmolkg b.wt.) compared with indomethacin (60%, 0.01 mmolkg b.wt.) as a reference drug. -
PMBJP Product.Pdf
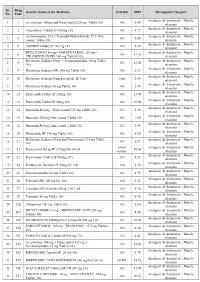
Sr. Drug Generic Name of the Medicine Unit Size MRP Therapeutic Category No. Code Analgesic & Antipyretic / Muscle 1 1 Aceclofenac 100mg and Paracetamol 325 mg Tablet 10's 10's 8.00 relaxants Analgesic & Antipyretic / Muscle 2 2 Aceclofenac Tablets IP 100mg 10's 10's 4.37 relaxants Acetaminophen 325 + Tramadol Hydrochloride 37.5 film Analgesic & Antipyretic / Muscle 3 4 10's 8.00 coated Tablet 10's relaxants Analgesic & Antipyretic / Muscle 4 5 ASPIRIN Tablets IP 150 mg 14's 14's 2.70 relaxants DICLOFENAC 50 mg+ PARACETAMOL 325 mg+ Analgesic & Antipyretic / Muscle 5 6 10's 11.30 CHLORZOXAZONE 500 mg Tablets 10's relaxants Diclofenac Sodium 50mg + Serratiopeptidase 10mg Tablet Analgesic & Antipyretic / Muscle 6 8 10's 12.00 10's relaxants Analgesic & Antipyretic / Muscle 7 9 Diclofenac Sodium (SR) 100 mg Tablet 10's 10's 6.12 relaxants Analgesic & Antipyretic / Muscle 8 10 Diclofenac Sodium 25mg per ml Inj. IP 3 ml 3 ml 2.00 relaxants Analgesic & Antipyretic / Muscle 9 11 Diclofenac Sodium 50 mg Tablet 10's 10's 2.90 relaxants Analgesic & Antipyretic / Muscle 10 12 Etoricoxilb Tablets IP 120mg 10's 10's 33.00 relaxants Analgesic & Antipyretic / Muscle 11 13 Etoricoxilb Tablets IP 90mg 10's 10's 25.00 relaxants Analgesic & Antipyretic / Muscle 12 14 Ibuprofen 400 mg + Paracetamol 325 mg Tablet 10's 15's 5.50 relaxants Analgesic & Antipyretic / Muscle 13 15 Ibuprofen 200 mg film coated Tablet 10's 10's 1.80 relaxants Analgesic & Antipyretic / Muscle 14 16 Ibuprofen 400 mg film coated Tablet 10's 15's 3.50 relaxants Analgesic & Antipyretic -

Pharmacology on Your Palms CLASSIFICATION of the DRUGS
Pharmacology on your palms CLASSIFICATION OF THE DRUGS DRUGS FROM DRUGS AFFECTING THE ORGANS CHEMOTHERAPEUTIC DIFFERENT DRUGS AFFECTING THE NERVOUS SYSTEM AND TISSUES DRUGS PHARMACOLOGICAL GROUPS Drugs affecting peripheral Antitumor drugs Drugs affecting the cardiovascular Antimicrobial, antiviral, Drugs affecting the nervous system Antiallergic drugs system antiparasitic drugs central nervous system Drugs affecting the sensory Antidotes nerve endings Cardiac glycosides Antibiotics CNS DEPRESSANTS (AFFECTING THE Antihypertensive drugs Sulfonamides Analgesics (opioid, AFFERENT INNERVATION) Antianginal drugs Antituberculous drugs analgesics-antipyretics, Antiarrhythmic drugs Antihelminthic drugs NSAIDs) Local anaesthetics Antihyperlipidemic drugs Antifungal drugs Sedative and hypnotic Coating drugs Spasmolytics Antiviral drugs drugs Adsorbents Drugs affecting the excretory system Antimalarial drugs Tranquilizers Astringents Diuretics Antisyphilitic drugs Neuroleptics Expectorants Drugs affecting the hemopoietic system Antiseptics Anticonvulsants Irritant drugs Drugs affecting blood coagulation Disinfectants Antiparkinsonian drugs Drugs affecting peripheral Drugs affecting erythro- and leukopoiesis General anaesthetics neurotransmitter processes Drugs affecting the digestive system CNS STIMULANTS (AFFECTING THE Anorectic drugs Psychomotor stimulants EFFERENT PART OF THE Bitter stuffs. Drugs for replacement therapy Analeptics NERVOUS SYSTEM) Antiacid drugs Antidepressants Direct-acting-cholinomimetics Antiulcer drugs Nootropics (Cognitive -
FIXED DOSE COMBINATIONS APPROVED by DCG (I) SINCE 1961 TILL 22Nd February 2019
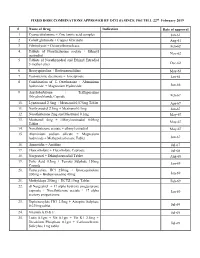
FIXED DOSE COMBINATIONS APPROVED BY DCG (I) SINCE 1961 TILL 22nd February 2019 # Name of Drug Indication Date of approval 1. Cyanocobalamine + Zinc tannic acid complex Jan-61 2. Cobalt glutamate + Copper Glycinate Aug-61 3. Fibrinolysin + Desoxyribonuclease Feb-62 4. Tablets of Norethisterone acetate + Ethinyl Nov-62 oestradiol 5. Tablets of Norethynodrel and Ethinyl Estradiol 3-methyl ether Dec-62 6. Broxyquinoline + Brobenzoxalidine May-63 7. Testosterone decanoate + Isocaproate Jan-64 8. Combination of L Oxethazaine + Aluminium hydroxide + Magnesium Hydroxide Jun-66 9. Amylobarbitone + Trifluperazine Dihydrochloride Capsule Feb-67 10. Lynestronol 2.5mg + Mestranol 0.075mg Tablet Apr-67 11. Northynodrel 2.5mg + Mestranol 0.1mg Jun-67 12. Norethisterone 2mg and Mestranol 0.1mg May-67 13. Mestranol 4mg + Ethinyloestradial 0.05mg May-67 Tablet 14. Norethisterane acetate + ethinyl estradiol May-67 15. Aluminium sodium silicate + Magnesium hydroxide + Methypolysiloxane Tablet Jun-67 16. Ammoidin + Amidine Jul-67 17. Fluocortolene + Flucortolene Caproate Jul-68 18. Norgestrel + Ethinyloestradiol Tablet Aug-68 19. Folic Acid 0.5mg + Ferrous Sulphate 150mg Jan-69 Capsule 20. Tetracycline HCl 250mg + Broxyquinoline 200mg + Brobenzoxadine 40mg Jan-69 21. Methyldopa 250mg + HCTZ 15mg Tablet Feb-69 22. dl Norgestrel + 17 alpha hydroxy progesterone caproate + Norethisterone acetate + 17 alpha Jan-69 acetoxy progesterone 23. Diphenoxylate HCl 2.5mg + Atropine Sulphate 0.025mg tablet Jul-69 24. Vitamin A,D & E Jul-69 25. Lutin 0.1gm + Vit 0.1gm + Vit K1 2.5mg + Dicalcium Phosphate 0.1gm + Carlozochrome Jul-69 Salicylate 1mg tablet 26. Vit K 1 5mg + Calcium Lactolionate 100 m g+ Carlozocrome Salicylate 2.5mg + Phenol 0.5% Jul-69 + Lignocaine Hcl 1% injection 27. -

Drug Name Plate Number Well Location % Inhibition, Screen Axitinib 1 1 20 Gefitinib (ZD1839) 1 2 70 Sorafenib Tosylate 1 3 21 Cr
Drug Name Plate Number Well Location % Inhibition, Screen Axitinib 1 1 20 Gefitinib (ZD1839) 1 2 70 Sorafenib Tosylate 1 3 21 Crizotinib (PF-02341066) 1 4 55 Docetaxel 1 5 98 Anastrozole 1 6 25 Cladribine 1 7 23 Methotrexate 1 8 -187 Letrozole 1 9 65 Entecavir Hydrate 1 10 48 Roxadustat (FG-4592) 1 11 19 Imatinib Mesylate (STI571) 1 12 0 Sunitinib Malate 1 13 34 Vismodegib (GDC-0449) 1 14 64 Paclitaxel 1 15 89 Aprepitant 1 16 94 Decitabine 1 17 -79 Bendamustine HCl 1 18 19 Temozolomide 1 19 -111 Nepafenac 1 20 24 Nintedanib (BIBF 1120) 1 21 -43 Lapatinib (GW-572016) Ditosylate 1 22 88 Temsirolimus (CCI-779, NSC 683864) 1 23 96 Belinostat (PXD101) 1 24 46 Capecitabine 1 25 19 Bicalutamide 1 26 83 Dutasteride 1 27 68 Epirubicin HCl 1 28 -59 Tamoxifen 1 29 30 Rufinamide 1 30 96 Afatinib (BIBW2992) 1 31 -54 Lenalidomide (CC-5013) 1 32 19 Vorinostat (SAHA, MK0683) 1 33 38 Rucaparib (AG-014699,PF-01367338) phosphate1 34 14 Lenvatinib (E7080) 1 35 80 Fulvestrant 1 36 76 Melatonin 1 37 15 Etoposide 1 38 -69 Vincristine sulfate 1 39 61 Posaconazole 1 40 97 Bortezomib (PS-341) 1 41 71 Panobinostat (LBH589) 1 42 41 Entinostat (MS-275) 1 43 26 Cabozantinib (XL184, BMS-907351) 1 44 79 Valproic acid sodium salt (Sodium valproate) 1 45 7 Raltitrexed 1 46 39 Bisoprolol fumarate 1 47 -23 Raloxifene HCl 1 48 97 Agomelatine 1 49 35 Prasugrel 1 50 -24 Bosutinib (SKI-606) 1 51 85 Nilotinib (AMN-107) 1 52 99 Enzastaurin (LY317615) 1 53 -12 Everolimus (RAD001) 1 54 94 Regorafenib (BAY 73-4506) 1 55 24 Thalidomide 1 56 40 Tivozanib (AV-951) 1 57 86 Fludarabine -

Neurochemical Mechanisms Underlying Alcohol Withdrawal
Neurochemical Mechanisms Underlying Alcohol Withdrawal John Littleton, MD, Ph.D. More than 50 years ago, C.K. Himmelsbach first suggested that physiological mechanisms responsible for maintaining a stable state of equilibrium (i.e., homeostasis) in the patient’s body and brain are responsible for drug tolerance and the drug withdrawal syndrome. In the latter case, he suggested that the absence of the drug leaves these same homeostatic mechanisms exposed, leading to the withdrawal syndrome. This theory provides the framework for a majority of neurochemical investigations of the adaptations that occur in alcohol dependence and how these adaptations may precipitate withdrawal. This article examines the Himmelsbach theory and its application to alcohol withdrawal; reviews the animal models being used to study withdrawal; and looks at the postulated neuroadaptations in three systems—the gamma-aminobutyric acid (GABA) neurotransmitter system, the glutamate neurotransmitter system, and the calcium channel system that regulates various processes inside neurons. The role of these neuroadaptations in withdrawal and the clinical implications of this research also are considered. KEY WORDS: AOD withdrawal syndrome; neurochemistry; biochemical mechanism; AOD tolerance; brain; homeostasis; biological AOD dependence; biological AOD use; disorder theory; biological adaptation; animal model; GABA receptors; glutamate receptors; calcium channel; proteins; detoxification; brain damage; disease severity; AODD (alcohol and other drug dependence) relapse; literature review uring the past 25 years research- science models used to study with- of the reasons why advances in basic ers have made rapid progress drawal neurochemistry as well as a research have not yet been translated Din understanding the chemi- reluctance on the part of clinicians to into therapeutic gains and suggests cal activities that occur in the nervous consider new treatments.