CAM Evolution in Bromeliads: Resolving Patterns of Ecological Opportunity, Speciation and Niche Conservatism
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

1 Retail Listings 2011 by USDA Zone, As of Sept 5 - Please Check for Current Availability
1 Retail listings 2011 by USDA zone, as of Sept 5 - please check for current availability USDA zone: 2 Alcea rosea 'Nigra' Classic hollyhock with dark maroon, nearly black flowers covering the 5-8 ft spires in July and August. They like well-drained soil and full to part sun with average summer water. Short-lived, they reseed easily establishing long-lived colonies. Frost hardy in USDA zone 2. 4in @ $3 Malvaceae Lindelofia longiflora Bright blue flowered cousin of a forget-me-not which blooms from late spring to frost. Long-live perennial, clumping to 2 ft by 2 ft in rich, moist soil in a half shady spot– think woodland. Great for a border that gets some water, but not much attention otherwise. Hardy to 25 below. 6in @ $12 Boraginaceae Physocarpus opulifolius 'Dart's Gold' golden ninebark Its golden foliage highlights the pure white, fragrant, summer flowers and brilliant red fruit in autumn. Peeling bark adds interest to this durable hedging plant or specimen, deciduous, to 5 ft tall and wide, smaller than the species. Out of the hottest afternoon sun seems to suit it best for foliage color. Can take a bit of drought, but best with a little summer water. Takes will to pruning. Frost hardy in USDA zone 2. 1g @ $12, 2g @ $22 Rosaceae Rosa glauca red leaf rose Grown as much for its foliage as its flowers this deciduous shrub, to 6 ft tall x 5 ft wide, has glaucous blue foliage and, in June, single pink flowers with white centers. Lovely rose hips follow and remain through the winter. -

New Jan16.2011
Spring 2011 Mail Order Catalog Cistus Nursery 22711 NW Gillihan Road Sauvie Island, OR 97231 503.621.2233 phone 503.621.9657 fax order by phone 9 - 5 pst, visit 10am - 5pm, fax, mail, or email: [email protected] 24-7-365 www.cistus.com Spring 2011 Mail Order Catalog 2 USDA zone: 2 Symphoricarpos orbiculatus ‘Aureovariegatus’ coralberry Old fashioned deciduous coralberry with knock your socks off variegation - green leaves with creamy white edges. Pale white-tinted-pink, mid-summer flowers attract bees and butterflies and are followed by bird friendly, translucent, coral berries. To 6 ft or so in most any normal garden conditions - full sun to part shade with regular summer water. Frost hardy in USDA zone 2. $12 Caprifoliaceae USDA zone: 3 Athyrium filix-femina 'Frizelliae' Tatting fern An unique and striking fern with narrow fronds, only 1" wide and oddly bumpy along the sides as if beaded or ... tatted. Found originally in the Irish garden of Mrs. Frizell and loved for it quirkiness ever since. To only 1 ft tall x 2 ft wide and deciduous, coming back slowly in spring. Best in bright shade or shade where soil is rich. Requires summer water. Frost hardy to -40F, USDA zone 3 and said to be deer resistant. $14 Woodsiaceae USDA zone: 4 Aralia cordata 'Sun King' perennial spikenard The foliage is golden, often with red stems, and dazzling on this big and bold perennial, quickly to 3 ft tall and wide, first discovered in a department store in Japan by nurseryman Barry Yinger. Spikes of aralia type white flowers in summer are followed by purple-black berries. -

ISB: Atlas of Florida Vascular Plants
Longleaf Pine Preserve Plant List Acanthaceae Asteraceae Wild Petunia Ruellia caroliniensis White Aster Aster sp. Saltbush Baccharis halimifolia Adoxaceae Begger-ticks Bidens mitis Walter's Viburnum Viburnum obovatum Deer Tongue Carphephorus paniculatus Pineland Daisy Chaptalia tomentosa Alismataceae Goldenaster Chrysopsis gossypina Duck Potato Sagittaria latifolia Cow Thistle Cirsium horridulum Tickseed Coreopsis leavenworthii Altingiaceae Elephant's foot Elephantopus elatus Sweetgum Liquidambar styraciflua Oakleaf Fleabane Erigeron foliosus var. foliosus Fleabane Erigeron sp. Amaryllidaceae Prairie Fleabane Erigeron strigosus Simpson's rain lily Zephyranthes simpsonii Fleabane Erigeron vernus Dog Fennel Eupatorium capillifolium Anacardiaceae Dog Fennel Eupatorium compositifolium Winged Sumac Rhus copallinum Dog Fennel Eupatorium spp. Poison Ivy Toxicodendron radicans Slender Flattop Goldenrod Euthamia caroliniana Flat-topped goldenrod Euthamia minor Annonaceae Cudweed Gamochaeta antillana Flag Pawpaw Asimina obovata Sneezeweed Helenium pinnatifidum Dwarf Pawpaw Asimina pygmea Blazing Star Liatris sp. Pawpaw Asimina reticulata Roserush Lygodesmia aphylla Rugel's pawpaw Deeringothamnus rugelii Hempweed Mikania cordifolia White Topped Aster Oclemena reticulata Apiaceae Goldenaster Pityopsis graminifolia Button Rattlesnake Master Eryngium yuccifolium Rosy Camphorweed Pluchea rosea Dollarweed Hydrocotyle sp. Pluchea Pluchea spp. Mock Bishopweed Ptilimnium capillaceum Rabbit Tobacco Pseudognaphalium obtusifolium Blackroot Pterocaulon virgatum -

O GÊNERO NIDULARIUM LEM. (BROMELIACEAE) NO ESTADO DO PARANÁ
Acta boI. bras. II (2): 1997 237 o GÊNERO NIDULARIUM LEM. (BROMELIACEAE) NO ESTADO DO PARANÁ Rosângela Capuano Tardivo 1 Armando Carlos Cervi 1,2 Recebido em 13/06/96, Aceito 31/12/97 RESUMO-(O gênero Nidu/arium Lem. (Bromeliaceae) no Estado do Paraná), Este trabalho é um estudo taxonõmico das espécies do gênero Nidu/arium no Estado do Paraná. O gênero está representado por seis espécies, três variedades e uma forma: N billbergioides (Schult. f) L. B. Sm. f billbergioides; N campo-alegrense Lem.; N exostigmum Tardivo; N gracile Tardivo; N innocentii Leme varo innocentii; N innocentii var. paxianum (Mez) L. B. Sm.; N innocentii Lem. va ro wittmac/danum (Harms) L. B. Sm. e N procerum Lindman. São apresentadas chaves de identificação, descrições, ilustrações e distribuição geográfica dos táxons estudados Palavras-chave: Nidu/arium, Bromeliaceae, taxonomüi ABSCTRACf - (The genus Nidu/arium Lem, (Bromeliaceae) in Paraná State). This work is a taxonomic study of Nidularium species in Paraná State. The genus is represented by six species, three varieties and one form: N billbergioides (Schult. f) L. B. Sm. f, billbergioides; N campo-alegrense Leme; N exostigmum Tardivo; N gracile Tardivo; N innocentii Lem, varo innocentii, N innocentii varo paxianum (Mez) L. B. Sm.; Ninnocentii var. wittmackianum (Harms) L. B. Sm. e N procerum Lindman. Identification keys, descriptions, illustrations and geographical distribuiton of the taxa studied are presented. Key words: Nidularium, Bromeliaceae, taxonomy Introdução A família Bromeliaceae possui cerca de -

Bromeliads Bromeliads Are a Family of Plants (Bromeliaceae, the Pineapple Family) Native to Tropical North and South America
A Horticulture Information article from the Wisconsin Master Gardener website, posted 19 March 2012 Bromeliads Bromeliads are a family of plants (Bromeliaceae, the pineapple family) native to tropical North and South America. Europeans fi rst found out about bromeliads on Columbus’ second trip to the New World in 1493, where the pineapple (Ananas sp.) was being cultivated by the Carib tribe in the West Indies. The commercial pineapple (Ananas comosus) is native to southern Brazil and Paraguay. After the colonization of the New World it was rapidly transported to all areas of the tropics, and now is widely grown in tropical and sub- tropical areas. The only A collection of bromeliads placed on a tree at Costa Flores, Costa Rica. bromeliad to occur north of the tropics is Spanish “moss” (Tillandsia usneoides). It is neither Spanish nor a moss, but an epiphytic bromeliad. It doesn’t look much like a typical Commercial pineapple, Ananas comosus, bromeliad, though, with its long scaly stems and reduced in the fi eld. fl owers. Bromeliads are monocots, many of which, like their grass relatives, have a special form of photosynthesis that uses a variation of the more usual biochemical pathways to allow them to use water more effi ciently. Even though they come from the tropics, this helps those that are epiphytes contend with life in the treetops where there is limited water and a real danger of drying out. There are about 2500 species Many bromeliads are tropical and several thousand hybrids epiphytes. and cultivars. Many have brightly colored leaves, fl owers or fruit, and range in size from moss-like species of Tillandsia to the enormous Puya raimondii from the Andes which produces a fl owering stem up to 15 feet tall. -

Taxonomic Revision of the Chilean Puya Species (Puyoideae
Taxonomic revision of the Chilean Puya species (Puyoideae, Bromeliaceae), with special notes on the Puya alpestris-Puya berteroniana species complex Author(s): Georg Zizka, Julio V. Schneider, Katharina Schulte and Patricio Novoa Source: Brittonia , 1 December 2013, Vol. 65, No. 4 (1 December 2013), pp. 387-407 Published by: Springer on behalf of the New York Botanical Garden Press Stable URL: https://www.jstor.org/stable/24692658 JSTOR is a not-for-profit service that helps scholars, researchers, and students discover, use, and build upon a wide range of content in a trusted digital archive. We use information technology and tools to increase productivity and facilitate new forms of scholarship. For more information about JSTOR, please contact [email protected]. Your use of the JSTOR archive indicates your acceptance of the Terms & Conditions of Use, available at https://about.jstor.org/terms New York Botanical Garden Press and Springer are collaborating with JSTOR to digitize, preserve and extend access to Brittonia This content downloaded from 146.244.165.8 on Sun, 13 Dec 2020 04:26:58 UTC All use subject to https://about.jstor.org/terms Taxonomic revision of the Chilean Puya species (Puyoideae, Bromeliaceae), with special notes on the Puya alpestris-Puya berteroniana species complex Georg Zizka1'2, Julio V. Schneider1'2, Katharina Schulte3, and Patricio Novoa4 1 Botanik und Molekulare Evolutionsforschung, Senckenberg Gesellschaft für Naturforschung and Johann Wolfgang Goethe-Universität, Senckenberganlage 25, 60325, Frankfurt am Main, Germany; e-mail: [email protected]; e-mail: [email protected] 2 Biodiversity and Climate Research Center (BIK-F), Senckenberganlage 25, 60325, Frankfurt am Main, Germany 3 Australian Tropical Herbarium and Tropical Biodiversity and Climate Change Centre, James Cook University, PO Box 6811, Caims, QLD 4870, Australia; e-mail: [email protected] 4 Jardin Botânico Nacional, Camino El Olivar 305, El Salto, Vina del Mar, Chile Abstract. -

Water Relations of Bromeliaceae in Their Evolutionary Context
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Apollo Botanical Journal of the Linnean Society, 2016, 181, 415–440. With 2 figures Think tank: water relations of Bromeliaceae in their evolutionary context JAMIE MALES* Department of Plant Sciences, University of Cambridge, Downing Street, Cambridge CB2 3EA, UK Received 31 July 2015; revised 28 February 2016; accepted for publication 1 March 2016 Water relations represent a pivotal nexus in plant biology due to the multiplicity of functions affected by water status. Hydraulic properties of plant parts are therefore likely to be relevant to evolutionary trends in many taxa. Bromeliaceae encompass a wealth of morphological, physiological and ecological variations and the geographical and bioclimatic range of the family is also extensive. The diversification of bromeliad lineages is known to be correlated with the origins of a suite of key innovations, many of which relate directly or indirectly to water relations. However, little information is known regarding the role of change in morphoanatomical and hydraulic traits in the evolutionary origins of the classical ecophysiological functional types in Bromeliaceae or how this role relates to the diversification of specific lineages. In this paper, I present a synthesis of the current knowledge on bromeliad water relations and a qualitative model of the evolution of relevant traits in the context of the functional types. I use this model to introduce a manifesto for a new research programme on the integrative biology and evolution of bromeliad water-use strategies. The need for a wide-ranging survey of morphoanatomical and hydraulic traits across Bromeliaceae is stressed, as this would provide extensive insight into structure– function relationships of relevance to the evolutionary history of bromeliads and, more generally, to the evolutionary physiology of flowering plants. -
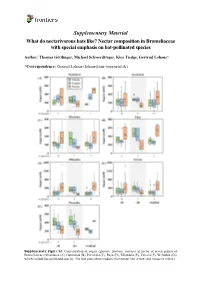
Supplementary Material What Do Nectarivorous Bats Like? Nectar Composition in Bromeliaceae with Special Emphasis on Bat-Pollinated Species
Supplementary Material What do nectarivorous bats like? Nectar composition in Bromeliaceae with special emphasis on bat-pollinated species Author: Thomas Göttlinger, Michael Schwerdtfeger, Kira Tiedge, Gertrud Lohaus* *Correspondence: Gertrud Lohaus ([email protected]) Supplementary Figure S1: Concentration of sugars (glucose, fructose, sucrose) in nectar of seven genera of Bromeliaceae (Alcantarea (A), Guzmania (B), Pitcairnia (C), Puya (D), Tillandsia (E), Vriesea (F), Werauhia (G)) which include bat-pollinated species. The box plots show medians (horizontal line in box) and means (x in box). Supplementary Material What do nectarivorous bats like? Nectar composition in Bromeliaceae with special emphasis on bat-pollinated species Author: Thomas Göttlinger, Michael Schwerdtfeger, Kira Tiedge, Gertrud Lohaus* *Correspondence: Gertrud Lohaus ([email protected]) Supplementary Figure S2: Concentration of amino acids (ala, arg, asn, asp, gaba, gln, glu, gly, his, iso, leu, lys, met, phe, pro, ser, thr, trp, tyr, val) in nectar of seven genera of Bromeliaceae (Alcantarea (A), Guzmania (B), Pitcairnia (C), Puya (D), Tillandsia (E), Vriesea (F), Werauhia (G)), which include bat-pollinated species. The box plots show medians (horizontal line in box) and means (x in box). Supplementary Material What do nectarivorous bats like? Nectar composition in Bromeliaceae with special emphasis on bat-pollinated species Author: Thomas Göttlinger, Michael Schwerdtfeger, Kira Tiedge, Gertrud Lohaus* *Correspondence: Gertrud Lohaus ([email protected]) Supplementary Figure S3: Cation concentrations (Ca2+, K+, Na+, Mg2+) in nectar of seven genera of Bromeliaceae (Alcantarea (A), Guzmania (B), Pitcairnia (C), Puya (D), Tillandsia (E), Vriesea (F), Werauhia (G)), which include bat-pollinated species. The box plots show medians (horizontal line in box) and means (x in box). -

2Do(). '!Phe . Famuy . Are Generally Con§Picu Mono
Rev. Bio\. Trop., 46(3):493-513, 1998 Current. floristk and phytogeographk knowledge of Mexican Bromeliaceae Adolfo Espejo Serna yAna Rosa López-Ferrari1 I Herbario Metropolitano, Depart¡unento de Biología,C.B.S., Universidad Autónoma Metropolitana, Unidad Jztapalapa, Apartado Postal 55,535,09340 México, D. F.,Fax 7244688, e-m<'lil: [email protected] Rece.ived 6-XI-1997. Corrected 28-V-1998. Accepted 19-VI-1998. Abstract: A current floristicand phytogeographic knowledge of native Mexican Bromeliaceae is presented. There are 22 genera of Brorlleliaceae recorded from the country Iha! ¡nelude 326 species. The genus Ursulaea with 2 species is endemic to Mexico, wbíle Hechtiawith 48 oC its 50 specíesbas its principal centerof diversity in the country. 7illandsia (175 spp), Hechtia (48 spp) and Pitcairnia (46 spp) are tbe genera with tbe greatest number of species. We present a comparative análysisof Mexican Bromeliaceae with tbat of other American regions that buve recently published accounts Cor the Family, .particularlythe Mesomerican area,Venezu¡:la, Ecuador, and tbeGuianas.Our results ledus to the cOI1e1usiontbat alltbese floras sbould be considered as distinct. We obse,rve a progressive decre¡¡¡se ofthe Simpson index value related wit� tbe remoteness of the Mexican area. A general análysisof tlrpspeCies numbers of Mexican bromeliad genera shows adistinct preference oftbespeci es forconiferousand oakfo,rests'; folÍowed by t�opical caduci ' folious forests. There is also significan! r¡:presentation of tbe family ifi'o ther vegetation types such as doud forests and tropical perennifolious forests. Generally Mel\ican Bromeliacea¡: speeies hav¡: scárceand sparse populationsandin manyc ases they inbabit diffs,bluffs and scaIJÍs in restrlcted areas,Col1cerning tbe. -

Epilist 1.0: a Global Checklist of Vascular Epiphytes
Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2021 EpiList 1.0: a global checklist of vascular epiphytes Zotz, Gerhard ; Weigelt, Patrick ; Kessler, Michael ; Kreft, Holger ; Taylor, Amanda Abstract: Epiphytes make up roughly 10% of all vascular plant species globally and play important functional roles, especially in tropical forests. However, to date, there is no comprehensive list of vas- cular epiphyte species. Here, we present EpiList 1.0, the first global list of vascular epiphytes based on standardized definitions and taxonomy. We include obligate epiphytes, facultative epiphytes, and hemiepiphytes, as the latter share the vulnerable epiphytic stage as juveniles. Based on 978 references, the checklist includes >31,000 species of 79 plant families. Species names were standardized against World Flora Online for seed plants and against the World Ferns database for lycophytes and ferns. In cases of species missing from these databases, we used other databases (mostly World Checklist of Selected Plant Families). For all species, author names and IDs for World Flora Online entries are provided to facilitate the alignment with other plant databases, and to avoid ambiguities. EpiList 1.0 will be a rich source for synthetic studies in ecology, biogeography, and evolutionary biology as it offers, for the first time, a species‐level overview over all currently known vascular epiphytes. At the same time, the list represents work in progress: species descriptions of epiphytic taxa are ongoing and published life form information in floristic inventories and trait and distribution databases is often incomplete and sometimes evenwrong. -

Anatomy of the Floral Scape of Bromeliaceae1 SUZANA LÚCIA PROENÇA2,3 and MARIA DAS GRAÇAS SAJO2
Revista Brasil. Bot., V.31, n.3, p.399-408, jul.-set. 2008 Anatomy of the floral scape of Bromeliaceae1 SUZANA LÚCIA PROENÇA2,3 and MARIA DAS GRAÇAS SAJO2 (received: July 04, 2007; accepted: June 05, 2008) ABSTRACT – (Anatomy of the floral scape of Bromeliaceae). This paper describes the anatomy of the floral scape for 12 species of Bromeliaceae, belonging to the subfamilies Bromelioideae, Tillandsioideae and Pitcairnioideae. Although all the scapes have a similar organization, there are variations in the structure of the epidermis, cortex and vascular cylinder. Such variations are described for the studied scapes and, when considered together they can help to identify the species. These aspects are described for each scape and discussed under a taxonomic point of view. Key words - anatomy, Bromeliaceae, floral scape RESUMO – (Anatomia do escapo floral de Bromeliaceae). Este trabalho descreve a anatomia do escapo floral de doze espécies de Bromeliaceae pertencentes às subfamílias Bromelioideae, Tillandsioideae e Pitcairnioideae e tem como objetivo ampliar o conhecimento anatômico da família e desse órgão em particular. Embora todos os escapos apresentem uma organização similar, observam-se variações na estrutura da epiderme, do córtex e do cilindro vascular. Tais variações são descritas para os escapos estudados e, quando são analisadas em conjunto, podem auxiliar na identificação das espécies. Esses aspectos são descritos para cada um dos escapos e discutidos dentro de um contexto taxonômico. Palavras-chave - anatomia, Bromeliaceae, escapo floral Introduction There are few studies on the floral scape anatomy of Bromeliaceae, the more important is Tomlinson’s revision Bromeliaceae comprises about 2,600 Neotropical (1969) of the results of Mez (1896 apud Tomlinson 1969), species, except for Pitcairnia feliciana (A. -

Aechmea Information Compiled by Theresa M
Aechmea Information compiled by Theresa M. Bert, Ph.D. (corresponding author) and Harry E. Luther, Director, Mulford B. Foster Bromeliad Identification Center (last update: January 2005) Welcome to the Aechmea species list. All taxonomic entities for the genus Aechmea listed in Luther (2004) & new species & taxonomic revisions since that publication up to September 2004 are included here. The information provided for each taxon is summarized from the references & citations provided at the end of the list. In the table, the citations are denoted by superscripted numbers. This information is not all-inclusive of everything that is known about each species, but much information is included. We did not include information on citings during personal expeditions unless they were documented in the literature & also provided unique information on the biology, ecology, or taxonomy of the species. Nor did we include information on cultivation. This is a dynamic table. As authoritative information becomes available, we will update this table. We also invite input. If you know of a well-documented fact about a species in this list, please provide the corresponding author with the information & the literature citation in which that information appears. (We reserve the right to accept or deny inclusion of any information provided to us.) We also welcome your thoughts on the type of information that should be included in this list. Blank fields denote no information is available. All currently recognized taxonomic entities of each species are listed, including subspecies, varieties, & forms. When the lower taxonomic level of these plants is the same as the species, only the species name is given (e.g., Aechmea distichantha var.