Properties of Fluids
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Thermodynamics Notes
Thermodynamics Notes Steven K. Krueger Department of Atmospheric Sciences, University of Utah August 2020 Contents 1 Introduction 1 1.1 What is thermodynamics? . .1 1.2 The atmosphere . .1 2 The Equation of State 1 2.1 State variables . .1 2.2 Charles' Law and absolute temperature . .2 2.3 Boyle's Law . .3 2.4 Equation of state of an ideal gas . .3 2.5 Mixtures of gases . .4 2.6 Ideal gas law: molecular viewpoint . .6 3 Conservation of Energy 8 3.1 Conservation of energy in mechanics . .8 3.2 Conservation of energy: A system of point masses . .8 3.3 Kinetic energy exchange in molecular collisions . .9 3.4 Working and Heating . .9 4 The Principles of Thermodynamics 11 4.1 Conservation of energy and the first law of thermodynamics . 11 4.1.1 Conservation of energy . 11 4.1.2 The first law of thermodynamics . 11 4.1.3 Work . 12 4.1.4 Energy transferred by heating . 13 4.2 Quantity of energy transferred by heating . 14 4.3 The first law of thermodynamics for an ideal gas . 15 4.4 Applications of the first law . 16 4.4.1 Isothermal process . 16 4.4.2 Isobaric process . 17 4.4.3 Isosteric process . 18 4.5 Adiabatic processes . 18 5 The Thermodynamics of Water Vapor and Moist Air 21 5.1 Thermal properties of water substance . 21 5.2 Equation of state of moist air . 21 5.3 Mixing ratio . 22 5.4 Moisture variables . 22 5.5 Changes of phase and latent heats . -

Lecture 13: Earth Materials
Earth Materials Lecture 13 Earth Materials GNH7/GG09/GEOL4002 EARTHQUAKE SEISMOLOGY AND EARTHQUAKE HAZARD Hooke’s law of elasticity Force Extension = E × Area Length Hooke’s law σn = E εn where E is material constant, the Young’s Modulus Units are force/area – N/m2 or Pa Robert Hooke (1635-1703) was a virtuoso scientist contributing to geology, σ = C ε palaeontology, biology as well as mechanics ij ijkl kl ß Constitutive equations These are relationships between forces and deformation in a continuum, which define the material behaviour. GNH7/GG09/GEOL4002 EARTHQUAKE SEISMOLOGY AND EARTHQUAKE HAZARD Shear modulus and bulk modulus Young’s or stiffness modulus: σ n = Eε n Shear or rigidity modulus: σ S = Gε S = µε s Bulk modulus (1/compressibility): Mt Shasta andesite − P = Kεv Can write the bulk modulus in terms of the Lamé parameters λ, µ: K = λ + 2µ/3 and write Hooke’s law as: σ = (λ +2µ) ε GNH7/GG09/GEOL4002 EARTHQUAKE SEISMOLOGY AND EARTHQUAKE HAZARD Young’s Modulus or stiffness modulus Young’s Modulus or stiffness modulus: σ n = Eε n Interatomic force Interatomic distance GNH7/GG09/GEOL4002 EARTHQUAKE SEISMOLOGY AND EARTHQUAKE HAZARD Shear Modulus or rigidity modulus Shear modulus or stiffness modulus: σ s = Gε s Interatomic force Interatomic distance GNH7/GG09/GEOL4002 EARTHQUAKE SEISMOLOGY AND EARTHQUAKE HAZARD Hooke’s Law σij and εkl are second-rank tensors so Cijkl is a fourth-rank tensor. For a general, anisotropic material there are 21 independent elastic moduli. In the isotropic case this tensor reduces to just two independent elastic constants, λ and µ. -
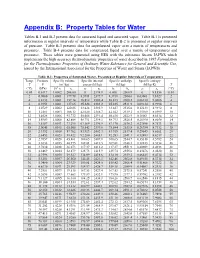
Appendix B: Property Tables for Water
Appendix B: Property Tables for Water Tables B-1 and B-2 present data for saturated liquid and saturated vapor. Table B-1 is presented information at regular intervals of temperature while Table B-2 is presented at regular intervals of pressure. Table B-3 presents data for superheated vapor over a matrix of temperatures and pressures. Table B-4 presents data for compressed liquid over a matrix of temperatures and pressures. These tables were generated using EES with the substance Steam_IAPWS which implements the high accuracy thermodynamic properties of water described in 1995 Formulation for the Thermodynamic Properties of Ordinary Water Substance for General and Scientific Use, issued by the International Associated for the Properties of Water and Steam (IAPWS). Table B-1: Properties of Saturated Water, Presented at Regular Intervals of Temperature Temp. Pressure Specific volume Specific internal Specific enthalpy Specific entropy T P (m3/kg) energy (kJ/kg) (kJ/kg) (kJ/kg-K) T 3 (°C) (kPa) 10 vf vg uf ug hf hg sf sg (°C) 0.01 0.6117 1.0002 206.00 0 2374.9 0.000 2500.9 0 9.1556 0.01 2 0.7060 1.0001 179.78 8.3911 2377.7 8.3918 2504.6 0.03061 9.1027 2 4 0.8135 1.0001 157.14 16.812 2380.4 16.813 2508.2 0.06110 9.0506 4 6 0.9353 1.0001 137.65 25.224 2383.2 25.225 2511.9 0.09134 8.9994 6 8 1.0729 1.0002 120.85 33.626 2385.9 33.627 2515.6 0.12133 8.9492 8 10 1.2281 1.0003 106.32 42.020 2388.7 42.022 2519.2 0.15109 8.8999 10 12 1.4028 1.0006 93.732 50.408 2391.4 50.410 2522.9 0.18061 8.8514 12 14 1.5989 1.0008 82.804 58.791 2394.1 58.793 2526.5 -

Equation of State of an Ideal Gas
ASME231 Atmospheric Thermodynamics NC A&T State U Department of Physics Dr. Yuh-Lang Lin http://mesolab.org [email protected] Lecture 3: Equation of State of an Ideal Gas As mentioned earlier, the state of a system is the condition of the system (or part of the system) at an instant of time measured by its properties. Those properties are called state variables, such as T, p, and V. The equation which relates T, p, and V is called equation of state. Since they are related by the equation of state, only two of them are independent. Thus, all the other thermodynamic properties will depend on the state defined by two independent variables by state functions. PVT system: The simplest thermodynamic system consists of a fixed mass of a fluid uninfluenced by chemical reactions or external fields. Such a system can be described by the pressure, volume and temperature, which are related by an equation of state f (p, V, T) = 0, (2.1) 1 where only two of them are independent. From physical experiments, the equation of state for an ideal gas, which is defined as a hypothetical gas whose molecules occupy negligible space and have no interactions, may be written as p = RT, (2.2) or p = RT, (2.3) where -3 : density (=m/V) in kg m , 3 -1 : specific volume (=V/m=1/) in m kg . R: specific gas constant (R=R*/M), -1 -1 -1 -1 [R=287 J kg K for dry air (Rd), 461 J kg K for water vapor for water vapor (Rv)]. -

Efficient Hydraulic Fluids Compressibility Is Still One of the Most Critically Important Factors
WORLDWIDE R. David Whitby Efficient hydraulic fluids Compressibility is still one of the most critically important factors. ydraulic fluids transmit power as a hydraulic system con- while phosphate and vegetable-oil esters have compressibilities H verts mechanical energy into fluid energy and subsequently similar to that of water. Polyalphaolefins are more compressible to mechanical work. To achieve this conversion efficiently, hydrau- than mineral oils. Water-glycol fluids are intermediate between lic fluids need to be relatively incompressible. Hydraulic fluids mineral oils and water. must also minimize wear, reduce friction, provide cooling and pre- Mineral oils are relatively incompressible, but volume reduc- vent rust and corrosion, be compatible with system components tions can be approximately 0.5% for pressures ranging from 6,900 and help keep the system free of deposits. kPa (1,000 psi) up to 27,600 kPa (4,000 psi). Compressibility in- Compressibility measures the rela- creases with pressure and temperature tive change in volume of a fluid or solid and has significant effects on high-pres- as a response to a change in pressure and sure fluid systems. Hydraulic oils typi- is the reciprocal of the volume-elastic cally contain 6% to 10% of dissolved air, modulus or bulk modulus of elasticity. which has no measurable effect on bulk Bulk modulus defines the pressure in- modulus provided it stays in solution. crease needed to cause a given relative Bulk modulus is an inherent property decrease in volume. of the hydraulic fluid and, therefore, is The bulk modulus of a fluid is nonlin- an inherent inefficiency of the hydraulic ear. -

Velocity, Density, Modulus of Hydrocarbon Fluids
Velocity, Density and Modulus of Hydrocarbon Fluids --Data Measurement De-hua Han*, HARC; M. Batzle, CSM Summary measured with pressure up to 55.2 Mpa (8000 Psi) and temperature up to 100 °C. Density and ultrasonic velocity of numerous hydrocarbon fluids (oil, oil based mud filtrate, hydrocarbon gases and Velocity of Dead Oil miscible CO2-oil) were measured at in situ conditions of pressure up to 50 Mpa and temperatures up to100 °C. Initial measurements were on the gas-free or ‘dead’ oils at Dynamic moduli are derived from velocities and densities. pressure and temperature (see Fig. 1). We used the Newly measured data refine correlations of velocity and following model to fit data: density to API gravity, Gas Oil ratio (GOR), Gas gravity and in situ pressure and temperature. Gas in solution is Vp (m/s) = A – B * T + C * P + D * T * P (1) largely responsible for reducing the bulk modulus of the live oil. Phase changes, such as exsolving gas during Here A is a pseudo velocity at 0 °C and room pressure (0 production, can dramatically lower velocities and modulus, Mpa, gauge), B is temperature gradient, C is pressure but is dependent on pressure conditions. Distinguish gas gradient and D is coefficient of coupled temperature and from liquid phase may not be possible at a high pressure. pressure effects. Fluids are often supercritical. With increasing pressure, a gas-like fluid can begin to behave like a liquid Dead & live oil of #1 B.P. = 1900 psi at 60 C Introduction 1800 1600 Hydrocarbon fluids are the primary targets of the seismic exploration. -

Density and Specific Weight Estimation for the Liquids and Solid Materials
Laboratory experiments Experiment no 2 – Density and specific weight estimation for the liquids and solid materials. 1. Theory Density is a physical property shared by all forms of matter (solids, liquids, and gases). In this lab investigation, we are mainly concerned with determining the density of solid objects; both regular-shaped and irregular-shaped. In general regular-shaped solid objects are those that have straight sides that can be measured using a metric ruler. These shapes include but are not limited to cubes and rectangular prisms. In general, irregular-shaped solid objects are those that do not have straight sides that cannot be measured with a metric ruler or slide caliper. The density of a material is defined as its mass per unit volume. The symbol of density is ρ (the Greek letter rho). m kg (1) V m3 The specific weight (also known as the unit weight) is the weight per unit volume of a material. The symbol of specific weight is γ (the Greek letter Gamma). W N (2) V m3 On the surface of the Earth, the weight W of an object is related to its mass m by: W = m · g, (3) where g is the acceleration due to the Earth's gravity, equal to about 9.81 ms-2. Using eq. 1, 2 and 3 we will obtain dependence between specific weight of the body and its density: g (4) Apparatus: Vernier caliper, balance with specific gravity platform (additional table in our case), 250 ml graduate beaker. Unknowns: a) Various solid samples (regular and irregular shaped) b) Light liquid sample (alcohol-water mixture) or heavy liquid sample (salt-water mixture). -
![[5] Magnetic Densimetry: Partial Specific Volume and Other Applications](https://docslib.b-cdn.net/cover/6742/5-magnetic-densimetry-partial-specific-volume-and-other-applications-566742.webp)
[5] Magnetic Densimetry: Partial Specific Volume and Other Applications
74 MOLECULAR WEIGHT DETERMINATIONS [5] [5] Magnetic Densimetry: Partial Specific Volume and Other Applications By D. W. KUPKE and J. W. BEAMS This chapter consists of three parts. Part I is a summary of the principles and current development of the magnetic densimeter, with which density values may be obtained conveniently on small volumes of solution with the speed and accuracy required for present-day ap- plications in protein chemistry. In Part II, the applications, including definitions, are outlined whereby the density property may be utilized for the study of protein solutions. Finally, in Part III, some practical aspects on the routine determination of densities of protein solutions by magnetic densimetry are listed. Part I. Magnetic Densimeter The magnetic densimeter measures by electromagnetic methods the vertical (up or down) force on a totally immersed buoy. From these measured forces together with the known mass and volume of the buoy, the density of the solution can be obtained by employing Archimedes' principle. In practice, it is convenient to eliminate evaluation of the mass and volume of the buoy and to determine the relation of the mag- netic to the mechanical forces on the buoy by direct calibration of the latter when it is immersed in liquids of known density. Several workers have devised magnetic float methods for determin- ing densities, but the method first described by Lamb and Lee I and later improved by MacInnes, Dayhoff, and Ray: is perhaps the most accurate means (~1 part in 106) devised up to that time for determining the densities of solutions. The magnetic method was not widely used, first because it was tedious and required considerable manipulative skill; second, the buoy or float was never stationary for periods long enough to allow ruling out of wall effects, viscosity perturbations, etc.; third, the technique required comparatively large volumes of the solution (350 ml) for accurate measurements. -

Hydraulics Manual Glossary G - 3
Glossary G - 1 GLOSSARY OF HIGHWAY-RELATED DRAINAGE TERMS (Reprinted from the 1999 edition of the American Association of State Highway and Transportation Officials Model Drainage Manual) G.1 Introduction This Glossary is divided into three parts: · Introduction, · Glossary, and · References. It is not intended that all the terms in this Glossary be rigorously accurate or complete. Realistically, this is impossible. Depending on the circumstance, a particular term may have several meanings; this can never change. The primary purpose of this Glossary is to define the terms found in the Highway Drainage Guidelines and Model Drainage Manual in a manner that makes them easier to interpret and understand. A lesser purpose is to provide a compendium of terms that will be useful for both the novice as well as the more experienced hydraulics engineer. This Glossary may also help those who are unfamiliar with highway drainage design to become more understanding and appreciative of this complex science as well as facilitate communication between the highway hydraulics engineer and others. Where readily available, the source of a definition has been referenced. For clarity or format purposes, cited definitions may have some additional verbiage contained in double brackets [ ]. Conversely, three “dots” (...) are used to indicate where some parts of a cited definition were eliminated. Also, as might be expected, different sources were found to use different hyphenation and terminology practices for the same words. Insignificant changes in this regard were made to some cited references and elsewhere to gain uniformity for the terms contained in this Glossary: as an example, “groundwater” vice “ground-water” or “ground water,” and “cross section area” vice “cross-sectional area.” Cited definitions were taken primarily from two sources: W.B. -

Chapter 3 3.4-2 the Compressibility Factor Equation of State
Chapter 3 3.4-2 The Compressibility Factor Equation of State The dimensionless compressibility factor, Z, for a gaseous species is defined as the ratio pv Z = (3.4-1) RT If the gas behaves ideally Z = 1. The extent to which Z differs from 1 is a measure of the extent to which the gas is behaving nonideally. The compressibility can be determined from experimental data where Z is plotted versus a dimensionless reduced pressure pR and reduced temperature TR, defined as pR = p/pc and TR = T/Tc In these expressions, pc and Tc denote the critical pressure and temperature, respectively. A generalized compressibility chart of the form Z = f(pR, TR) is shown in Figure 3.4-1 for 10 different gases. The solid lines represent the best curves fitted to the data. Figure 3.4-1 Generalized compressibility chart for various gases10. It can be seen from Figure 3.4-1 that the value of Z tends to unity for all temperatures as pressure approach zero and Z also approaches unity for all pressure at very high temperature. If the p, v, and T data are available in table format or computer software then you should not use the generalized compressibility chart to evaluate p, v, and T since using Z is just another approximation to the real data. 10 Moran, M. J. and Shapiro H. N., Fundamentals of Engineering Thermodynamics, Wiley, 2008, pg. 112 3-19 Example 3.4-2 ---------------------------------------------------------------------------------- A closed, rigid tank filled with water vapor, initially at 20 MPa, 520oC, is cooled until its temperature reaches 400oC. -

Ideal Gasses Is Known As the Ideal Gas Law
ESCI 341 – Atmospheric Thermodynamics Lesson 4 –Ideal Gases References: An Introduction to Atmospheric Thermodynamics, Tsonis Introduction to Theoretical Meteorology, Hess Physical Chemistry (4th edition), Levine Thermodynamics and an Introduction to Thermostatistics, Callen IDEAL GASES An ideal gas is a gas with the following properties: There are no intermolecular forces, except during collisions. All collisions are elastic. The individual gas molecules have no volume (they behave like point masses). The equation of state for ideal gasses is known as the ideal gas law. The ideal gas law was discovered empirically, but can also be derived theoretically. The form we are most familiar with, pV nRT . Ideal Gas Law (1) R has a value of 8.3145 J-mol1-K1, and n is the number of moles (not molecules). A true ideal gas would be monatomic, meaning each molecule is comprised of a single atom. Real gasses in the atmosphere, such as O2 and N2, are diatomic, and some gasses such as CO2 and O3 are triatomic. Real atmospheric gasses have rotational and vibrational kinetic energy, in addition to translational kinetic energy. Even though the gasses that make up the atmosphere aren’t monatomic, they still closely obey the ideal gas law at the pressures and temperatures encountered in the atmosphere, so we can still use the ideal gas law. FORM OF IDEAL GAS LAW MOST USED BY METEOROLOGISTS In meteorology we use a modified form of the ideal gas law. We first divide (1) by volume to get n p RT . V we then multiply the RHS top and bottom by the molecular weight of the gas, M, to get Mn R p T . -

FORMULAS for CALCULATING the SPEED of SOUND Revision G
FORMULAS FOR CALCULATING THE SPEED OF SOUND Revision G By Tom Irvine Email: [email protected] July 13, 2000 Introduction A sound wave is a longitudinal wave, which alternately pushes and pulls the material through which it propagates. The amplitude disturbance is thus parallel to the direction of propagation. Sound waves can propagate through the air, water, Earth, wood, metal rods, stretched strings, and any other physical substance. The purpose of this tutorial is to give formulas for calculating the speed of sound. Separate formulas are derived for a gas, liquid, and solid. General Formula for Fluids and Gases The speed of sound c is given by B c = (1) r o where B is the adiabatic bulk modulus, ro is the equilibrium mass density. Equation (1) is taken from equation (5.13) in Reference 1. The characteristics of the substance determine the appropriate formula for the bulk modulus. Gas or Fluid The bulk modulus is essentially a measure of stress divided by strain. The adiabatic bulk modulus B is defined in terms of hydrostatic pressure P and volume V as DP B = (2) - DV / V Equation (2) is taken from Table 2.1 in Reference 2. 1 An adiabatic process is one in which no energy transfer as heat occurs across the boundaries of the system. An alternate adiabatic bulk modulus equation is given in equation (5.5) in Reference 1. æ ¶P ö B = ro ç ÷ (3) è ¶r ø r o Note that æ ¶P ö P ç ÷ = g (4) è ¶r ø r where g is the ratio of specific heats.