Adiabatic Bulk Moduli
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
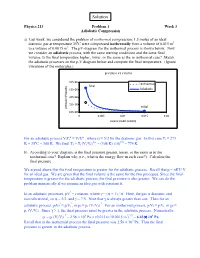
3-1 Adiabatic Compression
Solution Physics 213 Problem 1 Week 3 Adiabatic Compression a) Last week, we considered the problem of isothermal compression: 1.5 moles of an ideal diatomic gas at temperature 35oC were compressed isothermally from a volume of 0.015 m3 to a volume of 0.0015 m3. The pV-diagram for the isothermal process is shown below. Now we consider an adiabatic process, with the same starting conditions and the same final volume. Is the final temperature higher, lower, or the same as the in isothermal case? Sketch the adiabatic processes on the p-V diagram below and compute the final temperature. (Ignore vibrations of the molecules.) α α For an adiabatic process ViTi = VfTf , where α = 5/2 for the diatomic gas. In this case Ti = 273 o 1/α 2/5 K + 35 C = 308 K. We find Tf = Ti (Vi/Vf) = (308 K) (10) = 774 K. b) According to your diagram, is the final pressure greater, lesser, or the same as in the isothermal case? Explain why (i.e., what is the energy flow in each case?). Calculate the final pressure. We argued above that the final temperature is greater for the adiabatic process. Recall that p = nRT/ V for an ideal gas. We are given that the final volume is the same for the two processes. Since the final temperature is greater for the adiabatic process, the final pressure is also greater. We can do the problem numerically if we assume an idea gas with constant α. γ In an adiabatic processes, pV = constant, where γ = (α + 1) / α. -

Lecture 13: Earth Materials
Earth Materials Lecture 13 Earth Materials GNH7/GG09/GEOL4002 EARTHQUAKE SEISMOLOGY AND EARTHQUAKE HAZARD Hooke’s law of elasticity Force Extension = E × Area Length Hooke’s law σn = E εn where E is material constant, the Young’s Modulus Units are force/area – N/m2 or Pa Robert Hooke (1635-1703) was a virtuoso scientist contributing to geology, σ = C ε palaeontology, biology as well as mechanics ij ijkl kl ß Constitutive equations These are relationships between forces and deformation in a continuum, which define the material behaviour. GNH7/GG09/GEOL4002 EARTHQUAKE SEISMOLOGY AND EARTHQUAKE HAZARD Shear modulus and bulk modulus Young’s or stiffness modulus: σ n = Eε n Shear or rigidity modulus: σ S = Gε S = µε s Bulk modulus (1/compressibility): Mt Shasta andesite − P = Kεv Can write the bulk modulus in terms of the Lamé parameters λ, µ: K = λ + 2µ/3 and write Hooke’s law as: σ = (λ +2µ) ε GNH7/GG09/GEOL4002 EARTHQUAKE SEISMOLOGY AND EARTHQUAKE HAZARD Young’s Modulus or stiffness modulus Young’s Modulus or stiffness modulus: σ n = Eε n Interatomic force Interatomic distance GNH7/GG09/GEOL4002 EARTHQUAKE SEISMOLOGY AND EARTHQUAKE HAZARD Shear Modulus or rigidity modulus Shear modulus or stiffness modulus: σ s = Gε s Interatomic force Interatomic distance GNH7/GG09/GEOL4002 EARTHQUAKE SEISMOLOGY AND EARTHQUAKE HAZARD Hooke’s Law σij and εkl are second-rank tensors so Cijkl is a fourth-rank tensor. For a general, anisotropic material there are 21 independent elastic moduli. In the isotropic case this tensor reduces to just two independent elastic constants, λ and µ. -

Thermodynamics Formulation of Economics Burin Gumjudpai A
Thermodynamics Formulation of Economics 306706052 NIDA E-THESIS 5710313001 thesis / recv: 09012563 15:07:41 seq: 16 Burin Gumjudpai A Thesis Submitted in Partial Fulfillment of the Requirements for the Degree of Master of Economics (Financial Economics) School of Development Economics National Institute of Development Administration 2019 Thermodynamics Formulation of Economics Burin Gumjudpai School of Development Economics Major Advisor (Associate Professor Yuthana Sethapramote, Ph.D.) The Examining Committee Approved This Thesis Submitted in Partial 306706052 Fulfillment of the Requirements for the Degree of Master of Economics (Financial Economics). Committee Chairperson NIDA E-THESIS 5710313001 thesis / recv: 09012563 15:07:41 seq: 16 (Assistant Professor Pongsak Luangaram, Ph.D.) Committee (Assistant Professor Athakrit Thepmongkol, Ph.D.) Committee (Associate Professor Yuthana Sethapramote, Ph.D.) Dean (Associate Professor Amornrat Apinunmahakul, Ph.D.) ______/______/______ ABST RACT ABSTRACT Title of Thesis Thermodynamics Formulation of Economics Author Burin Gumjudpai Degree Master of Economics (Financial Economics) Year 2019 306706052 We consider a group of information-symmetric consumers with one type of commodity in an efficient market. The commodity is fixed asset and is non-disposable. We are interested in seeing if there is some connection of thermodynamics formulation NIDA E-THESIS 5710313001 thesis / recv: 09012563 15:07:41 seq: 16 to microeconomics. We follow Carathéodory approach which requires empirical existence of equation of state (EoS) and coordinates before performing maximization of variables. In investigating EoS of various system for constructing the economics EoS, unexpectedly new insights of thermodynamics are discovered. With definition of truly endogenous function, criteria rules and diagrams are proposed in identifying the status of EoS for an empirical equation. -

Efficient Hydraulic Fluids Compressibility Is Still One of the Most Critically Important Factors
WORLDWIDE R. David Whitby Efficient hydraulic fluids Compressibility is still one of the most critically important factors. ydraulic fluids transmit power as a hydraulic system con- while phosphate and vegetable-oil esters have compressibilities H verts mechanical energy into fluid energy and subsequently similar to that of water. Polyalphaolefins are more compressible to mechanical work. To achieve this conversion efficiently, hydrau- than mineral oils. Water-glycol fluids are intermediate between lic fluids need to be relatively incompressible. Hydraulic fluids mineral oils and water. must also minimize wear, reduce friction, provide cooling and pre- Mineral oils are relatively incompressible, but volume reduc- vent rust and corrosion, be compatible with system components tions can be approximately 0.5% for pressures ranging from 6,900 and help keep the system free of deposits. kPa (1,000 psi) up to 27,600 kPa (4,000 psi). Compressibility in- Compressibility measures the rela- creases with pressure and temperature tive change in volume of a fluid or solid and has significant effects on high-pres- as a response to a change in pressure and sure fluid systems. Hydraulic oils typi- is the reciprocal of the volume-elastic cally contain 6% to 10% of dissolved air, modulus or bulk modulus of elasticity. which has no measurable effect on bulk Bulk modulus defines the pressure in- modulus provided it stays in solution. crease needed to cause a given relative Bulk modulus is an inherent property decrease in volume. of the hydraulic fluid and, therefore, is The bulk modulus of a fluid is nonlin- an inherent inefficiency of the hydraulic ear. -

Velocity, Density, Modulus of Hydrocarbon Fluids
Velocity, Density and Modulus of Hydrocarbon Fluids --Data Measurement De-hua Han*, HARC; M. Batzle, CSM Summary measured with pressure up to 55.2 Mpa (8000 Psi) and temperature up to 100 °C. Density and ultrasonic velocity of numerous hydrocarbon fluids (oil, oil based mud filtrate, hydrocarbon gases and Velocity of Dead Oil miscible CO2-oil) were measured at in situ conditions of pressure up to 50 Mpa and temperatures up to100 °C. Initial measurements were on the gas-free or ‘dead’ oils at Dynamic moduli are derived from velocities and densities. pressure and temperature (see Fig. 1). We used the Newly measured data refine correlations of velocity and following model to fit data: density to API gravity, Gas Oil ratio (GOR), Gas gravity and in situ pressure and temperature. Gas in solution is Vp (m/s) = A – B * T + C * P + D * T * P (1) largely responsible for reducing the bulk modulus of the live oil. Phase changes, such as exsolving gas during Here A is a pseudo velocity at 0 °C and room pressure (0 production, can dramatically lower velocities and modulus, Mpa, gauge), B is temperature gradient, C is pressure but is dependent on pressure conditions. Distinguish gas gradient and D is coefficient of coupled temperature and from liquid phase may not be possible at a high pressure. pressure effects. Fluids are often supercritical. With increasing pressure, a gas-like fluid can begin to behave like a liquid Dead & live oil of #1 B.P. = 1900 psi at 60 C Introduction 1800 1600 Hydrocarbon fluids are the primary targets of the seismic exploration. -

FORMULAS for CALCULATING the SPEED of SOUND Revision G
FORMULAS FOR CALCULATING THE SPEED OF SOUND Revision G By Tom Irvine Email: [email protected] July 13, 2000 Introduction A sound wave is a longitudinal wave, which alternately pushes and pulls the material through which it propagates. The amplitude disturbance is thus parallel to the direction of propagation. Sound waves can propagate through the air, water, Earth, wood, metal rods, stretched strings, and any other physical substance. The purpose of this tutorial is to give formulas for calculating the speed of sound. Separate formulas are derived for a gas, liquid, and solid. General Formula for Fluids and Gases The speed of sound c is given by B c = (1) r o where B is the adiabatic bulk modulus, ro is the equilibrium mass density. Equation (1) is taken from equation (5.13) in Reference 1. The characteristics of the substance determine the appropriate formula for the bulk modulus. Gas or Fluid The bulk modulus is essentially a measure of stress divided by strain. The adiabatic bulk modulus B is defined in terms of hydrostatic pressure P and volume V as DP B = (2) - DV / V Equation (2) is taken from Table 2.1 in Reference 2. 1 An adiabatic process is one in which no energy transfer as heat occurs across the boundaries of the system. An alternate adiabatic bulk modulus equation is given in equation (5.5) in Reference 1. æ ¶P ö B = ro ç ÷ (3) è ¶r ø r o Note that æ ¶P ö P ç ÷ = g (4) è ¶r ø r where g is the ratio of specific heats. -

The First Law of Thermodynamics Continued Pre-Reading: §19.5 Where We Are
Lecture 7 The first law of thermodynamics continued Pre-reading: §19.5 Where we are The pressure p, volume V, and temperature T are related by an equation of state. For an ideal gas, pV = nRT = NkT For an ideal gas, the temperature T is is a direct measure of the average kinetic energy of its 3 3 molecules: KE = nRT = NkT tr 2 2 2 3kT 3RT and vrms = (v )av = = r m r M p Where we are We define the internal energy of a system: UKEPE=+∑∑ interaction Random chaotic between atoms motion & molecules For an ideal gas, f UNkT= 2 i.e. the internal energy depends only on its temperature Where we are By considering adding heat to a fixed volume of an ideal gas, we showed f f Q = Nk∆T = nR∆T 2 2 and so, from the definition of heat capacity Q = nC∆T f we have that C = R for any ideal gas. V 2 Change in internal energy: ∆U = nCV ∆T Heat capacity of an ideal gas Now consider adding heat to an ideal gas at constant pressure. By definition, Q = nCp∆T and W = p∆V = nR∆T So from ∆U = Q W − we get nCV ∆T = nCp∆T nR∆T − or Cp = CV + R It takes greater heat input to raise the temperature of a gas a given amount at constant pressure than constant volume YF §19.4 Ratio of heat capacities Look at the ratio of these heat capacities: we have f C = R V 2 and f + 2 C = C + R = R p V 2 so C p γ = > 1 CV 3 For a monatomic gas, CV = R 3 5 2 so Cp = R + R = R 2 2 C 5 R 5 and γ = p = 2 = =1.67 C 3 R 3 YF §19.4 V 2 Problem An ideal gas is enclosed in a cylinder which has a movable piston. -

Session 15 Thermodynamics
Session 15 Thermodynamics Cheryl Hurkett Physics Innovations Centre for Excellence in Learning and Teaching CETL (Leicester) Department of Physics and Astronomy University of Leicester 1 Contents Welcome .................................................................................................................................. 4 Session Authors .................................................................................................................. 4 Learning Objectives .............................................................................................................. 5 The Problem ........................................................................................................................... 6 Issues .................................................................................................................................... 6 Energy, Heat and Work ........................................................................................................ 7 The First Law of thermodynamics................................................................................... 8 Work..................................................................................................................................... 9 Heat .................................................................................................................................... 11 Principal Specific heats of a gas ..................................................................................... 12 Summary .......................................................................................................................... -

1 Microphysics
ASTR 501 Stellar Physics 1 1 Microphysics • equation of state, density: ρ(P, T, X) • radiative absorption coefficient, opacity: κ(P, T, X) • rate of energy production: (P, T, X) (nuclear physics) Nuclear physics also responsible for composition changes dXj = Fˆj · Xj (1) dt burn while the total composition change also includes a mixing term: dX ∂ dX i = 4πr2ρ D i (2) dt mix ∂m dm 1.1 Equation of state For the fluid to have an EOS it must be collisional: lregion λ (3) where λ is the mean free path. P from equation of state, consider three cases: ASTR 501 Stellar Physics 2 (a) ideal gas equation of state: p = R ρT (b) barotropic equation of state: pressure p depends only on ρ, as for example • isothermal: p ∼ ρ • adiabatic: p = Kργ Plus non-ideal effects, e.g. electron degeneracy. 1.2 Ideal gas equation of state Assumption: internal energy entirely in kinetic energy according to kinetic theory: = CVT , where CV is the specific heat at constant volume. This can be seen from the first law of thermodynamics dQ = d + pdV , where V is the specific volume, d if we express d = dT dT . d Similarly, the specific heat at constant pressure Cp = dT + R where R is the gas constant. This follows from the ideal gas equation of state. [†13] It also follows that Cp − CV = R [†14], and the ratio of specific heats is defined as C γ = p . (4) CV ASTR 501 Stellar Physics 3 α CV can be related to R as CV = 2 R , where α is the number of degrees of freedom 1 per particle, since the energy per particle per degree of freedom is 2kT where k is 2+α the Boltzmann constant, and R = nk. -

Course: US01CPHY01 UNIT – 1 ELASTICITY – I Introduction
Course: US01CPHY01 UNIT – 1 ELASTICITY – I Introduction: If the distance between any two points in a body remains invariable, the body is said to be a rigid body. In practice it is not possible to have a perfectly rigid body. The deformations are (i) There may be change in length (ii) There is a change of volume but no change in shape (iii) There is a change in shape with no change in volume All bodies get deformed under the action of force. The size and shape of the body will change on application of force. There is a tendency of body to recover its original size and shape on removal of this force. Elasticity: The property of a material body to regain its original condition on the removal of deforming forces, is called elasticity. Quartz fibre is considered to be the perfectly elastic body. Plasticity: The bodies which do not show any tendency to recover their original condition on the removal of deforming forces are called plasticity. Putty is considered to be the perfectly plastic body. Load: The load is the combination of external forces acting on a body and its effect is to change the form or the dimensions of the body. Any kind of deforming force is known as Load. When a body is subjected to a force or a system of forces it undergoes a change in size or shape or both. Elastic bodies offer appreciable resistance to the deforming forces. As a result, work has to be done to deform them. This amount of work is stored in body as elastic potential energy. -

Heat Capacity Ratio of Gases
Heat Capacity Ratio of Gases Carson Hasselbrink [email protected] Office Hours: Mon 10-11am, Beaupre 360 1 Purpose • To determine the heat capacity ratio for a monatomic and a diatomic gas. • To understand and mathematically model reversible & irreversible adiabatic processes for ideal gases. • To practice error propagation for complex functions. 2 Key Physical Concepts • Heat capacity is the amount of heat required to raise the temperature of an object or substance one degree 풅풒 푪 = 풅푻 • Heat Capacity Ratio is the ratio of specific heats at constant pressure and constant volume 퐶푝 훾 = 퐶푣 휕퐻 휕퐸 Where 퐶 = ( ) and 퐶 = ( ) 푝 휕푇 푝 푣 휕푇 푣 • An adiabatic process occurs when no heat is exchanged between the system and the surroundings 3 Theory: Heat Capacity 휕퐸 • Heat Capacity (Const. Volume): 퐶 = ( ) /푛 푣,푚 휕푇 푣 3푅푇 3푅 – Monatomic: 퐸 = , so 퐶 = 푚표푛푎푡표푚푖푐 2 푣,푚 2 5푅푇 5푅 – Diatomic: 퐸 = , so 퐶 = 푑푖푎푡표푚푖푐 2 푣,푚 2 • Heat Capacity Ratio: 퐶푝,푚 = 퐶푣,푚 + 푅 퐶푝,푚 푅 훾 = = 1 + 퐶푣,푚 퐶푣,푚 푃 [ln 1 ] 푃2 – Reversible: 훾 = 푃 [ln 1 ] 푃3 푃 [ 1 −1] 푃2 – Irreversible: 훾 = 푃 [ 1 −1] 푃3 • Diatomic heat capacity > Monatomic Heat Capacity 4 Theory: Determination of Heat Capacity Ratio • We will subject a gas to an adiabatic expansion and then allow the gas to return to its original temperature via an isochoric process, during which time it will cool. • This expansion and warming can be modeled in two different ways. 5 Reversible Expansion (Textbook) • Assume that pressure in carboy (P1) and exterior pressure (P2) are always close enough that entire process is always in equilibrium • Since system is in equilibrium, each step must be reversible 6 Irreversible Expansion (Lab Syllabus) • Assume that pressure in carboy (P1) and exterior pressure (P2) are not close enough; there is sudden deviation in pressure; the system is not in equilibrium • Since system is not in equilibrium, the process becomes irreversible. -

The Mechanical Properties and Elastic Anisotropies of Cubic Ni3al from First Principles Calculations
crystals Article The Mechanical Properties and Elastic Anisotropies of Cubic Ni3Al from First Principles Calculations Xinghe Luan 1,2, Hongbo Qin 1,2,* ID , Fengmei Liu 1,*, Zongbei Dai 1, Yaoyong Yi 1 and Qi Li 1 1 Guangdong Provincial Key Laboratory of Advanced Welding Technology, Guangdong Welding Institute (China-Ukraine E.O. Paton Institute of Welding), Guangzhou 541630, China; [email protected] (X.L.); [email protected] (Z.D.); [email protected] (Y.Y.); [email protected] (Q.L.) 2 Key Laboratory of Guangxi Manufacturing System and Advanced Manufacturing Technology, Guilin University of Electronic Technology, Guilin 541004, China * Correspondence: [email protected] (H.Q.); [email protected] (F.L.) Received: 21 June 2018; Accepted: 22 July 2018; Published: 25 July 2018 Abstract: Ni3Al-based superalloys have excellent mechanical properties which have been widely used in civilian and military fields. In this study, the mechanical properties of the face-centred cubic structure Ni3Al were investigated by a first principles study based on density functional theory (DFT), and the generalized gradient approximation (GGA) was used as the exchange-correlation function. The bulk modulus, Young’s modulus, shear modulus and Poisson’s ratio of Ni3Al polycrystal were calculated by Voigt-Reuss approximation method, which are in good agreement with the existing experimental values. Moreover, directional dependences of bulk modulus, Young’s modulus, shear modulus and Poisson’s ratio of Ni3Al single crystal were explored. In addition, the thermodynamic properties (e.g., Debye temperature) of Ni3Al were investigated based on the calculated elastic constants, indicating an improved accuracy in this study, verified with a small deviation from the previous experimental value.