Nanoarchaeota”: Indication for a Wide Distribution in High Temperature Biotopes
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Genome of Nanoarchaeum Equitans: Insights Into Early Archaeal Evolution and Derived Parasitism
The genome of Nanoarchaeum equitans: Insights into early archaeal evolution and derived parasitism Elizabeth Waters†‡, Michael J. Hohn§, Ivan Ahel¶, David E. Graham††, Mark D. Adams‡‡, Mary Barnstead‡‡, Karen Y. Beeson‡‡, Lisa Bibbs†, Randall Bolanos‡‡, Martin Keller†, Keith Kretz†, Xiaoying Lin‡‡, Eric Mathur†, Jingwei Ni‡‡, Mircea Podar†, Toby Richardson†, Granger G. Sutton‡‡, Melvin Simon†, Dieter So¨ ll¶§§¶¶, Karl O. Stetter†§¶¶, Jay M. Short†, and Michiel Noordewier†¶¶ †Diversa Corporation, 4955 Directors Place, San Diego, CA 92121; ‡Department of Biology, San Diego State University, 5500 Campanile Drive, San Diego, CA 92182; §Lehrstuhl fu¨r Mikrobiologie und Archaeenzentrum, Universita¨t Regensburg, Universita¨tsstrasse 31, D-93053 Regensburg, Germany; ‡‡Celera Genomics Rockville, 45 West Gude Drive, Rockville, MD 20850; Departments of ¶Molecular Biophysics and Biochemistry and §§Chemistry, Yale University, New Haven, CT 06520-8114; and ʈDepartment of Biochemistry, Virginia Polytechnic Institute and State University, Blacksburg, VA 24061 Communicated by Carl R. Woese, University of Illinois at Urbana–Champaign, Urbana, IL, August 21, 2003 (received for review July 22, 2003) The hyperthermophile Nanoarchaeum equitans is an obligate sym- (6–8). Genomic DNA was either digested with restriction en- biont growing in coculture with the crenarchaeon Ignicoccus. zymes or sheared to provide clonable fragments. Two plasmid Ribosomal protein and rRNA-based phylogenies place its branching libraries were made by subcloning randomly sheared fragments point early in the archaeal lineage, representing the new archaeal of this DNA into a high-copy number vector (Ϸ2.8 kbp library) kingdom Nanoarchaeota. The N. equitans genome (490,885 base or low-copy number vector (Ϸ6.3 kbp library). DNA sequence pairs) encodes the machinery for information processing and was obtained from both ends of plasmid inserts to create repair, but lacks genes for lipid, cofactor, amino acid, or nucleotide ‘‘mate-pairs,’’ pairs of reads from single clones that should be biosyntheses. -

The Mínimum Cell
The minimum cell GENOMIC – ADVANCED GENETICS AUTHOR: I. ODEI BARREÑADA Overview The Concept Small genomes Minimal gene set Approache theories Minimal genome proyect Future insight The concept “MINIMUN CELL“ The smallest size (of genetic The smallest unit of life that can information) replicate autonomously = minimum genome Small genomes Circovirus (1.800 base pairs / 3 gens) Virus Carsonella ruddi (159 kb /182 genes) Symbiont Nanoarchaeum equitans (490 kb/ 553 genes) Parasite Mycoplasma genitalium (582 kb/ 521 genes ) Parasite Pelagibacter ubique (1,3 Mb/ 1,370 genes) Free-living Smallest genomes Circovirus (1.800 base pairs / 3 gens) Virus Carsonella ruddi (159 kb /182 genes) Symbiont Nanoarchaeum equitans (490 kb/ 553 genes) Parasite Mycoplasma genitalium (582 kb/ 521 genes ) Parasite Pelagibacter ubique (1,3 Mb/ 1,370 genes) Free-living (Giovannoni et al., 2005) “MINIMAL GENE SET” By genome comparison Ubiquitous genes: Translation Transcription Replication of DNA Variable genes: Depends of environment (Koonin, 2003) Theories for reach the minimum cell Two approaches Top – Down knock-down known organism Bottom-up --> build from synthetics DNA Minimal genome project J. Craig Venter Institute Search of essential genes Gene disruption by transposons Seq the survivors organisms Detect the disrupted gene Declare this genes as NON-ESENTIAL In M. genitalium only 382 of 521 genes are essential (Gibson et al., 2011) Build de novo M. genitalium genome Future insight - Cell design Create artificial cells for: Generation of hydrogen for fuel Capturing excess carbon dioxide in the atmosphere Drug delivery directly into the body As Enzyme therapy Artificial blood cells … And all you can think References Koonin E V. -

The Mysterious Orphans of Mycoplasmataceae
The mysterious orphans of Mycoplasmataceae Tatiana V. Tatarinova1,2*, Inna Lysnyansky3, Yuri V. Nikolsky4,5,6, and Alexander Bolshoy7* 1 Children’s Hospital Los Angeles, Keck School of Medicine, University of Southern California, Los Angeles, 90027, California, USA 2 Spatial Science Institute, University of Southern California, Los Angeles, 90089, California, USA 3 Mycoplasma Unit, Division of Avian and Aquatic Diseases, Kimron Veterinary Institute, POB 12, Beit Dagan, 50250, Israel 4 School of Systems Biology, George Mason University, 10900 University Blvd, MSN 5B3, Manassas, VA 20110, USA 5 Biomedical Cluster, Skolkovo Foundation, 4 Lugovaya str., Skolkovo Innovation Centre, Mozhajskij region, Moscow, 143026, Russian Federation 6 Vavilov Institute of General Genetics, Moscow, Russian Federation 7 Department of Evolutionary and Environmental Biology and Institute of Evolution, University of Haifa, Israel 1,2 [email protected] 3 [email protected] 4-6 [email protected] 7 [email protected] 1 Abstract Background: The length of a protein sequence is largely determined by its function, i.e. each functional group is associated with an optimal size. However, comparative genomics revealed that proteins’ length may be affected by additional factors. In 2002 it was shown that in bacterium Escherichia coli and the archaeon Archaeoglobus fulgidus, protein sequences with no homologs are, on average, shorter than those with homologs [1]. Most experts now agree that the length distributions are distinctly different between protein sequences with and without homologs in bacterial and archaeal genomes. In this study, we examine this postulate by a comprehensive analysis of all annotated prokaryotic genomes and focusing on certain exceptions. -

University of Oklahoma Graduate College Development of High
UNIVERSITY OF OKLAHOMA GRADUATE COLLEGE DEVELOPMENT OF HIGH-THROUGHPUT EXPERIMENTAL AND COMPUTATIONAL TECHNOLOGIES FOR ANALYZING MICROBIAL FUNCTIONS AND INTERACTIONS IN ENVIRONMENTAL METAGENOMES A DISSERTATION SUBMITTED TO THE GRADUATE FACULTY in partial fulfillment of the requirements for the Degree of DOCTOR OF PHILOSOPHY By ZHOU SHI Norman, Oklahoma 2017 DEVELOPMENT OF HIGH-THROUGHPUT EXPERIMENTAL AND COMPUTATIONAL TECHNOLOGIES FOR ANALYZING MICROBIAL FUNCTIONS AND INTERACTIONS IN ENVIRONMENTAL METAGENOMES A DISSERTATION APPROVED FOR THE DEPARTMENT OF MICROBIOLOGY AND PLANT BIOLOGY BY ______________________________ Dr. Lee Krumholz, Chair ______________________________ Dr. Meijun Zhu ______________________________ Dr. Sridhar Radhakrishnan ______________________________ Dr. Yiqi Luo ______________________________ Dr. Jizhong Zhou © Copyright by ZHOU SHI 2017 All Rights Reserved. Dedication I dedicate this dissertation to my wife, Mengting Yuan, whom I met and married during the time period of this work and whose love, faith and support is the greatest gift that I can ever imagine in my life; to my son, Andrew Shi, who is always curious, energetic and wonderful, and is my lasting source of enlightenment and true happiness; to my parents, Yiping Shi and Aiping Zeng, whose selfless love and care transcended the distance and time, and carried me whenever I need; to my aunt, Cindy Shi, whose unwavering support and encouragement instilled in me the spirit of striving for excellence; to my friend, Su Xu, who boarded on the same plane with me to the U.S. seven years ago and then has witnessed all parts of this work as my best friend, and Dian Zou, who passed away in an accident during the time period of this work and is a friend I will forever miss. -

Cultivating the Macroalgal Holobiont: Effects of Integrated Multi-Trophic Aquaculture on the Microbiome of Ulva Rigida (Chlorophyta)
fmars-07-00052 February 10, 2020 Time: 15:0 # 1 ORIGINAL RESEARCH published: 12 February 2020 doi: 10.3389/fmars.2020.00052 Cultivating the Macroalgal Holobiont: Effects of Integrated Multi-Trophic Aquaculture on the Microbiome of Ulva rigida (Chlorophyta) Gianmaria Califano1, Michiel Kwantes1, Maria Helena Abreu2, Rodrigo Costa3,4,5 and Thomas Wichard1,6* 1 Institute for Inorganic and Analytical Chemistry, Friedrich Schiller University Jena, Jena, Germany, 2 ALGAplus Lda, Ílhavo, Portugal, 3 Institute for Bioengineering and Biosciences, Instituto Superior Técnico, Universidade de Lisboa, Lisbon, Portugal, 4 Centre of Marine Sciences, University of Algarve, Faro, Portugal, 5 Lawrence Berkeley National Laboratory, U.S. Department of Energy Joint Genome Institute, University of California, Berkeley, Berkeley, CA, United States, 6 Jena School for Microbial Communication, Jena, Germany Ulva is a ubiquitous macroalgal genus of commercial interest. Integrated Multi-Trophic Aquaculture (IMTA) systems promise large-scale production of macroalgae due to Edited by: their high productivity and environmental sustainability. Complex host–microbiome Bernardo Antonio Perez Da Gama, interactions play a decisive role in macroalgal development, especially in Ulva spp. Universidade Federal Fluminense, due to algal growth- and morphogenesis-promoting factors released by associated Brazil bacteria. However, our current understanding of the microbial community assembly and Reviewed by: Alejandro H. Buschmann, structure in cultivated macroalgae is scant. We aimed to determine (i) to what extent University of Los Lagos, Chile IMTA settings influence the microbiome associated with U. rigida and its rearing water, (ii) Henrique Fragoso Santos, to explore the dynamics of beneficial microbes to algal growth and development under Universidade Federal Fluminense, Brazil IMTA settings, and (iii) to improve current knowledge of host–microbiome interactions. -

Insights Into Archaeal Evolution and Symbiosis from the Genomes of a Nanoarchaeon and Its Inferred Crenarchaeal Host from Obsidian Pool, Yellowstone National Park
University of Tennessee, Knoxville TRACE: Tennessee Research and Creative Exchange Microbiology Publications and Other Works Microbiology 4-22-2013 Insights into archaeal evolution and symbiosis from the genomes of a nanoarchaeon and its inferred crenarchaeal host from Obsidian Pool, Yellowstone National Park Mircea Podar University of Tennessee - Knoxville, [email protected] Kira S. Makarova National Institutes of Health David E. Graham University of Tennessee - Knoxville, [email protected] Yuri I. Wolf National Institutes of Health Eugene V. Koonin National Institutes of Health See next page for additional authors Follow this and additional works at: https://trace.tennessee.edu/utk_micrpubs Part of the Microbiology Commons Recommended Citation Biology Direct 2013, 8:9 doi:10.1186/1745-6150-8-9 This Article is brought to you for free and open access by the Microbiology at TRACE: Tennessee Research and Creative Exchange. It has been accepted for inclusion in Microbiology Publications and Other Works by an authorized administrator of TRACE: Tennessee Research and Creative Exchange. For more information, please contact [email protected]. Authors Mircea Podar, Kira S. Makarova, David E. Graham, Yuri I. Wolf, Eugene V. Koonin, and Anna-Louise Reysenbach This article is available at TRACE: Tennessee Research and Creative Exchange: https://trace.tennessee.edu/ utk_micrpubs/44 Podar et al. Biology Direct 2013, 8:9 http://www.biology-direct.com/content/8/1/9 RESEARCH Open Access Insights into archaeal evolution and symbiosis from the genomes of a nanoarchaeon and its inferred crenarchaeal host from Obsidian Pool, Yellowstone National Park Mircea Podar1,2*, Kira S Makarova3, David E Graham1,2, Yuri I Wolf3, Eugene V Koonin3 and Anna-Louise Reysenbach4 Abstract Background: A single cultured marine organism, Nanoarchaeum equitans, represents the Nanoarchaeota branch of symbiotic Archaea, with a highly reduced genome and unusual features such as multiple split genes. -

Archaea and the Origin of Eukaryotes
REVIEWS Archaea and the origin of eukaryotes Laura Eme, Anja Spang, Jonathan Lombard, Courtney W. Stairs and Thijs J. G. Ettema Abstract | Woese and Fox’s 1977 paper on the discovery of the Archaea triggered a revolution in the field of evolutionary biology by showing that life was divided into not only prokaryotes and eukaryotes. Rather, they revealed that prokaryotes comprise two distinct types of organisms, the Bacteria and the Archaea. In subsequent years, molecular phylogenetic analyses indicated that eukaryotes and the Archaea represent sister groups in the tree of life. During the genomic era, it became evident that eukaryotic cells possess a mixture of archaeal and bacterial features in addition to eukaryotic-specific features. Although it has been generally accepted for some time that mitochondria descend from endosymbiotic alphaproteobacteria, the precise evolutionary relationship between eukaryotes and archaea has continued to be a subject of debate. In this Review, we outline a brief history of the changing shape of the tree of life and examine how the recent discovery of a myriad of diverse archaeal lineages has changed our understanding of the evolutionary relationships between the three domains of life and the origin of eukaryotes. Furthermore, we revisit central questions regarding the process of eukaryogenesis and discuss what can currently be inferred about the evolutionary transition from the first to the last eukaryotic common ancestor. Sister groups Two descendants that split The pioneering work by Carl Woese and colleagues In this Review, we discuss how culture- independent from the same node; the revealed that all cellular life could be divided into three genomics has transformed our understanding of descendants are each other’s major evolutionary lines (also called domains): the archaeal diversity and how this has influenced our closest relative. -

1 Characterization of Sulfur Metabolizing Microbes in a Cold Saline Microbial Mat of the Canadian High Arctic Raven Comery Mast
Characterization of sulfur metabolizing microbes in a cold saline microbial mat of the Canadian High Arctic Raven Comery Master of Science Department of Natural Resource Sciences Unit: Microbiology McGill University, Montreal July 2015 A thesis submitted to McGill University in partial fulfillment of the requirements of the degree of Master in Science © Raven Comery 2015 1 Abstract/Résumé The Gypsum Hill (GH) spring system is located on Axel Heiberg Island of the High Arctic, perennially discharging cold hypersaline water rich in sulfur compounds. Microbial mats are found adjacent to channels of the GH springs. This thesis is the first detailed analysis of the Gypsum Hill spring microbial mats and their microbial diversity. Physicochemical analyses of the water saturating the GH spring microbial mat show that in summer it is cold (9°C), hypersaline (5.6%), and contains sulfide (0-10 ppm) and thiosulfate (>50 ppm). Pyrosequencing analyses were carried out on both 16S rRNA transcripts (i.e. cDNA) and genes (i.e. DNA) to investigate the mat’s community composition, diversity, and putatively active members. In order to investigate the sulfate reducing community in detail, the sulfite reductase gene and its transcript were also sequenced. Finally, enrichment cultures for sulfate/sulfur reducing bacteria were set up and monitored for sulfide production at cold temperatures. Overall, sulfur metabolism was found to be an important component of the GH microbial mat system, particularly the active fraction, as 49% of DNA and 77% of cDNA from bacterial 16S rRNA gene libraries were classified as taxa capable of the reduction or oxidation of sulfur compounds. -
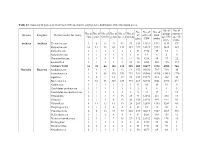
Domain Kingdom Phylum (Order for Virus) No. of Class No. of Order No. of Family No. of Genus No. of Species No. of Strains No. O
Table S2. Summary of probes on GeoChip 5.0M based on the phylogenetic distribution of the functional genes. No. of No. of No. No. of No. of No. of No. of No. of No. of No. of No. of group- sequence Domain Kingdom Phylum (order for virus) of covered total class order family genus species strains specific -specific gene CDS probe probe probe Archaea Archaea Crenarchaeota 2 6 9 22 43 57 142 13921 1312 1133 179 Euryarchaeota 10 12 23 69 133 187 229 33323 3992 3625 367 Korarchaeota 1 1 1 1 1 1 33 3718 35 33 2 Nanoarchaeota 1 1 1 1 1 1 6 12 6 6 0 Thaumarchaeota 1 4 5 7 9 14 46 5218 95 73 22 unclassified 1 1 1 1 1 25 46 1885 288 115 173 Archaea Total 16 25 40 101 188 285 502 58077 5728 4985 743 Bacteria Bacteria Acidobacteria 5 4 4 8 13 18 155 20180 745 715 30 Actinobacteria 1 8 48 120 351 753 381 80060 15394 13618 1776 Aquificae 1 2 4 12 17 22 127 19379 533 498 35 Bacteroidetes 7 10 23 107 239 394 267 58970 9661 8790 871 Caldiserica 1 1 1 1 1 1 13 38 19 19 0 Candidatus poribacteria 1 1 1 1 1 1 2 5 4 1 3 Candidatus saccharibacteria 1 1 1 1 1 3 9 22 17 4 13 Chlamydiae 2 1 4 7 15 23 74 12652 241 230 11 Chlorobi 1 1 1 6 15 20 138 21932 995 944 51 Chloroflexi 8 11 12 14 19 28 209 32839 1353 1289 64 Chrysiogenetes 1 1 1 2 2 2 33 81 41 40 1 Cyanobacteria 2 8 12 57 97 203 237 46219 4727 4209 518 Deferribacteres 1 1 1 4 4 4 81 2583 165 154 11 Deinococcus-thermus 2 3 4 7 18 27 172 21352 1026 978 48 Dictyoglomi 1 1 1 1 2 2 51 9447 99 98 1 Elusimicrobia 2 2 2 2 2 2 34 5713 50 49 1 Fibrobacteres 2 1 1 1 1 2 50 6671 69 64 5 Firmicutes 6 10 42 176 593 1556 -

Concerted Gene Recruitment in Early Plant Evolution Jinling Huang* and J Peter Gogarten†
Open Access Research2008HuangVolume and 9, Issue Gogarten 7, Article R109 Concerted gene recruitment in early plant evolution Jinling Huang* and J Peter Gogarten† Addresses: *Department of Biology, Howell Science Complex, East Carolina University, Greenville, NC 27858, USA. †Department of Molecular and Cell Biology, University of Connecticut, 91 North Eagleville Road, Storrs, CT 06269, USA. Correspondence: Jinling Huang. Email: [email protected] Published: 8 July 2008 Received: 30 April 2008 Revised: 24 June 2008 Genome Biology 2008, 9:R109 (doi:10.1186/gb-2008-9-7-r109) Accepted: 8 July 2008 The electronic version of this article is the complete one and can be found online at http://genomebiology.com/2008/9/7/R109 © 2008 Huang and Gogarten; licensee BioMed Central Ltd. This is an open access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Eukaryotic<p>Analysesto the split ofhorizontal ofred the algae red gene andalgal transfergreen <it>Cyanidioschyzon</it> plants.</p> genome identified 37 genes that were acquired from non-organellar sources prior Abstract Background: Horizontal gene transfer occurs frequently in prokaryotes and unicellular eukaryotes. Anciently acquired genes, if retained among descendants, might significantly affect the long-term evolution of the recipient lineage. However, no systematic studies on the scope of anciently acquired genes and their impact on macroevolution are currently available in eukaryotes. Results: Analyses of the genome of the red alga Cyanidioschyzon identified 37 genes that were acquired from non-organellar sources prior to the split of red algae and green plants. -

Expansion of Thaumarchaeota Habitat Range Is Correlated with Horizontal Transfer of Atpase Operons
Lawrence Berkeley National Laboratory Recent Work Title Expansion of Thaumarchaeota habitat range is correlated with horizontal transfer of ATPase operons. Permalink https://escholarship.org/uc/item/19h9369p Journal The ISME journal, 13(12) ISSN 1751-7362 Authors Wang, Baozhan Qin, Wei Ren, Yi et al. Publication Date 2019-12-01 DOI 10.1038/s41396-019-0493-x Peer reviewed eScholarship.org Powered by the California Digital Library University of California The ISME Journal (2019) 13:3067–3079 https://doi.org/10.1038/s41396-019-0493-x ARTICLE Expansion of Thaumarchaeota habitat range is correlated with horizontal transfer of ATPase operons 1,2 3 4 1 2 2 5,6 Baozhan Wang ● Wei Qin ● Yi Ren ● Xue Zhou ● Man-Young Jung ● Ping Han ● Emiley A. Eloe-Fadrosh ● 7 8 1 9 9 7 4 10 10 Meng Li ● Yue Zheng ● Lu Lu ● Xin Yan ● Junbin Ji ● Yang Liu ● Linmeng Liu ● Cheryl Heiner ● Richard Hall ● 11 2 12 5 13 Willm Martens-Habbena ● Craig W. Herbold ● Sung-keun Rhee ● Douglas H. Bartlett ● Li Huang ● 3 2,14 15 1 Anitra E. Ingalls ● Michael Wagner ● David A. Stahl ● Zhongjun Jia Received: 15 April 2019 / Revised: 1 July 2019 / Accepted: 29 July 2019 / Published online: 28 August 2019 © The Author(s) 2019. This article is published with open access Abstract Thaumarchaeota are responsible for a significant fraction of ammonia oxidation in the oceans and in soils that range from alkaline to acidic. However, the adaptive mechanisms underpinning their habitat expansion remain poorly understood. Here we show that expansion into acidic soils and the high pressures of the hadopelagic zone of the oceans is tightly linked to the acquisition of a variant of the energy-yielding ATPases via horizontal transfer. -

A Comparative Analysis of the Moose Rumen Microbiota and the Pursuit of Improving Fibrolytic Systems
University of Vermont ScholarWorks @ UVM Graduate College Dissertations and Theses Dissertations and Theses 2015 A Comparative Analysis Of The oM ose Rumen Microbiota And The Pursuit Of Improving Fibrolytic Systems. Suzanne Ishaq Pellegrini University of Vermont Follow this and additional works at: https://scholarworks.uvm.edu/graddis Part of the Animal Sciences Commons, Microbiology Commons, and the Molecular Biology Commons Recommended Citation Pellegrini, Suzanne Ishaq, "A Comparative Analysis Of The oosM e Rumen Microbiota And The urP suit Of Improving Fibrolytic Systems." (2015). Graduate College Dissertations and Theses. 365. https://scholarworks.uvm.edu/graddis/365 This Dissertation is brought to you for free and open access by the Dissertations and Theses at ScholarWorks @ UVM. It has been accepted for inclusion in Graduate College Dissertations and Theses by an authorized administrator of ScholarWorks @ UVM. For more information, please contact [email protected]. A COMPARATIVE ANALYSIS OF THE MOOSE RUMEN MICROBIOTA AND THE PURSUIT OF IMPROVING FIBROLYTIC SYSTEMS. A Dissertation Presented by Suzanne Ishaq Pellegrini to The Faculty of the Graduate College of The University of Vermont In Partial Fulfillment of the Requirements For the Degree of Doctor of Philosophy Specializing in Animal, Nutrition and Food Science May, 2015 Defense Date: March 19, 2015 Dissertation Examination Committee: André-Denis G. Wright, Ph.D., Advisor Indra N. Sarkar, Ph.D., MLIS, Chairperson John W. Barlow, Ph.D., D.V.M. Douglas I. Johnson, Ph.D. Stephanie D. McKay, Ph.D. Cynthia J. Forehand, Ph.D., Dean of the Graduate College ABSTRACT The goal of the work presented herein was to further our understanding of the rumen microbiota and microbiome of wild moose, and to use that understanding to improve other processes.