How to Protect Yourself Against Malaria 1 Fig
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Artemether-Lumefantrine (Six-Dose Regimen) for Treating Uncomplicated Falciparum Malaria (Review)
Artemether-lumefantrine (six-dose regimen) for treating uncomplicated falciparum malaria (Review) Omari AAA, Gamble CL, Garner P This is a reprint of a Cochrane review, prepared and maintained by The Cochrane Collaboration and published in The Cochrane Library 2009, Issue 1 http://www.thecochranelibrary.com Artemether-lumefantrine (six-dose regimen) for treating uncomplicated falciparum malaria (Review) Copyright © 2009 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd. TABLE OF CONTENTS HEADER....................................... 1 ABSTRACT ...................................... 1 PLAINLANGUAGESUMMARY . 2 BACKGROUND .................................... 2 OBJECTIVES ..................................... 3 METHODS ...................................... 3 RESULTS....................................... 5 DISCUSSION ..................................... 9 AUTHORS’CONCLUSIONS . 9 ACKNOWLEDGEMENTS . 10 REFERENCES ..................................... 10 CHARACTERISTICSOFSTUDIES . 13 DATAANDANALYSES. 20 Analysis 1.1. Comparison 1 Artemether-lumefantrine vs amodiaquine, Outcome 1 Total failure by day 28. 22 Analysis 1.2. Comparison 1 Artemether-lumefantrine vs amodiaquine, Outcome 2 Total failure by day 14. 23 Analysis 1.3. Comparison 1 Artemether-lumefantrine vs amodiaquine, Outcome 3 Gametocyte carriage on day 14. 23 Analysis 2.1. Comparison 2 Artemether-lumefantrine vs chloroquine plus sulfadoxine-pyrimethamine, Outcome 1 Total failurebyday28. ................................ 24 Analysis 2.2. Comparison 2 Artemether-lumefantrine -

Psychiatric Side Effects of Mefloquine: Applications to Forensic Psychiatry
REGULAR ARTICLE Psychiatric Side Effects of Mefloquine: Applications to Forensic Psychiatry Elspeth Cameron Ritchie, MD, MPH, Jerald Block, MD, and Remington Lee Nevin, MD, MPH Mefloquine (previously marketed in the United States as Lariam®) is an antimalarial medication with potent psychotropic potential. Severe psychiatric side effects due to mefloquine intoxication are well documented, including anxiety, panic attacks, paranoia, persecutory delusions, dissociative psychosis, and anterograde amnesia. Exposure to the drug has been associated with acts of violence and suicide. In this article, we discuss the history of mefloquine use and describe plausible mechanisms of its psychotropic action. Mefloquine intoxication has not yet been successfully advanced in legal proceedings as a defense or as a mitigating factor, but it appears likely that it eventually will be. Considerations for the application of claims of mefloquine intoxication in forensic settings are discussed. J Am Acad Psychiatry Law 41:224–35, 2013 Mefloquine is a 4-quinolinemethanol antimalarial The company pursued regulatory approval and mar- first synthesized in the early 1970s1 by researchers keted the drug to civilian travelers in the United affiliated with the United States military’s Walter States under the trade name Lariam® after its initial Reed Army Institute of Research (WRAIR).2 The Food and Drug Administration (FDA) licensure in drug’s development was the culmination of a 10-year 1989.5 Owing to its efficacy, presumed safety, and drug discovery effort, during which time more than convenient dose schedule that facilitated prophylac- 300,000 compounds were screened for their antima- tic use, mefloquine was soon identified as the drug of 2 larial properties. -

Hydroxychloroquine Or Chloroquine for Treating Coronavirus Disease 2019 (COVID-19) – a PROTOCOL for a Systematic Review of Individual Participant Data
Hydroxychloroquine or Chloroquine for treating Coronavirus Disease 2019 (COVID-19) – a PROTOCOL for a systematic review of Individual Participant Data Authors Fontes LE, Riera R, Miranda E, Oke J, Heneghan CJ, Aronson JK, Pacheco RL, Martimbianco ALC, Nunan D BACKGROUND In the face of the pandemic of SARS CoV2, urgent research is needed to test potential therapeutic agents against the disease. Reliable research shall inform clinical decision makers. Currently, there are several studies testing the efficacy and safety profiles of different pharmacological interventions. Among these drugs, we can cite antimalarial, antivirals, biological drugs, interferon, etc. As of 6 April 2020 there are three published reportsand 100 ongoing trials testing hydroxychloroquine/chloroquine alone or in association with other drugs for COVID-19. This prospective systematic review with Individual Participant data aims to assess the rigour of the best-available evidence for hydroxychloroquine or chloroquine as treatment for COVID-19 infection. The PICO framework is: P: adults with COVID-19 infection I: chloroquine or hydroxychloroquine (alone or in association) C: placebo, other active treatments, usual standard care without antimalarials O: efficacy and safety outcomes OBJECTIVES To assess the effects (benefits and harms) of chloroquine or hydroxychloroquine for the treatment of COVID-19 infection. METHODS Criteria for considering studies for this review Types of studies We shall include randomized controlled trials (RCTs) with a parallel design. We intend to include even small trials (<50 participants), facing the urgent need for evidence to respond to the current pandemic. Quasi-randomized, non-randomized, or observational studies will be excluded due to a higher risk of confounding and selection bias (1). -

The History and Ethics of Malaria Eradication and Control Campaigns in Tropical Africa
Malaria Redux: The History and Ethics of Malaria Eradication and Control Campaigns in Tropical Africa Center for Historical Research Ohio State University Spring 2012 Seminars: Epidemiology in World History Prof. J.L.A. Webb, Jr. Department of History Colby College DRAFT: NOT FOR CITATION 2 During the 1950s, colonial malariologists, in conjunction with experts from the World Health Organization (WHO), set up malaria eradication pilot projects across tropical Africa. They deployed new synthetic insecticides such as DLD, HCH, and DDT, and new antimalarials, such as chloroquine and pyrimethamine, in an effort to establish protocols for eradication. These efforts ‘protected’ some fourteen million Africans. Yet by the early 1960s, the experts concluded that malaria eradication was not feasible, and the pilot projects were disbanded. The projects had achieved extremely low levels of infection for years at a time, but the experts had to accept with regret that their interventions were unable to reduce malaria transmission to zero. The projects had high recurrent costs, and it was understood that they were financially unsustainable. The pilot projects were allowed to lapse. The malaria eradication pilot projects had reduced the rates of infection to levels so low that the ‘protected’ populations lost their acquired immunities to malaria during the years of the projects. In the immediate aftermath of the projects, the Africans were subject to severe malaria, which sometimes afflicted entire communities in epidemic form, until they regained their immunities. How Should We Understand the Ethics of the Early Eradication Efforts? The ethics of malaria control in the 1950s and 1960s seemed self-evident to the interventionists. -

Prevention of Malaria in Travellers (Pdf 59KB)
THEME Travel medicine Prevention of malaria in travellers BACKGROUND Malaria remains endemic in over 100 countries worldwide. Travellers to these countries may be at risk of contracting disease. Assessing risk on an individual basis can be challenging. Trish Batchelor OBJECTIVE MBBS, MPH, FRACGP, is This article identifies the traveller at high risk of contracting malaria, outlines preventive methods, including personal Medical Director, Travel Doctor- protection and chemoprophylaxis if indicated, and summarises the risks and benefits of the most commonly used TMVC, Canberra, Australian chemoprophylactic agents. Capital Territory, and Executive Board Member, International DISCUSSION Society of Travel Medicine. Appropriately assessing the risk of malaria in an individual traveller can be complex. A number of factors beyond the trish.batchelor@traveldoctor. com.au country being visited influence the level of risk. These should be identified and taken into account when discussing malaria prevention with individuals. The traveller should be actively involved in the decision making process in order Tony Gherardin to enhance compliance. High risk individuals should be identified. Personal protection methods should always be MBBS, MPH, FRACGP, is emphasised. If chemoprophylaxis is indicated, the contraindications, advantages and side effects should be discussed. National Medical Adviser, Travel Doctor-TMVC, If in doubt, referral to a specialised travel medicine clinic should be considered. Melbourne, Victoria. The malaria parasite, spread via the bite of an infected Risk for travellers female Anopheles mosquito, remains a significant health threat in over 100 countries worldwide. An The risk of contracting malaria varies significantly from estimated 350–500 million cases occur annually, with traveller to traveller, and this is the challenge of providing over a million sub-Saharan Africans, mainly children, quality advice. -
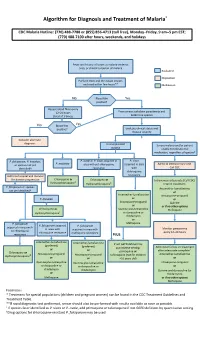
Algorithm for Diagnosis and Treatment of Malaria*
Algorithm for Diagnosis and Treatment of Malaria* CDC Malaria Hotline: (770) 488-7788 or (855) 856-4713 (toll free), Monday–Friday, 9 am–5 pm EST; (770) 488-7100 after hours, weekends, and holidays Fever and history of travel to malaria-endemic area, or clinical suspicion of malaria Evaluation Disposition Perform thick and thin blood smears and read within few hours** Medication No Blood film Yes positive? Repeat blood films every 12–24 hours From smear: calculate parasitemia and (total of 3 times) determine species No Blood film Yes positive? Evaluate clinical status and disease severity Consider alternate diagnosis Uncomplicated Severe malaria and/or patient malaria unable to tolerate oral medication, regardless of species† P. falciparum, P. knowlesi, P. ovale or P. vivax acquired in P. vivax P. malariae or species not yet area without chloroquine acquired in area Admit to intensive care unit. identified† resistance with Call CDC chloroquine Admit to hospital and monitor resistance for disease progression Chloroquine or Chloroquine or ‡ Intravenous artesunate (Call CDC) Hydroxychloroquine Hydroxychloroquine‡ Interim treatment: P. falciparum or species Artemether-lumefantrine not yet identified† or Artemether-lumefantrine Atovaquone-proguanil P. knowlesi or or Atovaquone-proguanil Quinine or or if no other options Chloroquine or Quinine plus tetracycline Mefloquine Hydroxychloroquine‡ or doxycycline or clindamycin or P. falciparum Mefloquine P. falciparum acquired P. falciparum acquired in area with in area with acquired in area with -

Chemoprophylaxis of Tropical Infectious Diseases
Pharmaceuticals 2010, 3, 1561-1575; doi:10.3390/ph3051561 OPEN ACCESS pharmaceuticals ISSN 1424-8247 www.mdpi.com/journal/pharmaceuticals Review Chemoprophylaxis of Tropical Infectious Diseases William J. H. McBride School of Medicine and Dentistry, James Cook University, Cairns Base Hospital campus, The Esplanade, Cairns, Queensland 4870, Australia; E-Mail: [email protected]; Tel.: +617-40506530; Fax: +617-40506831 Received: 08 April 2010; in revised form: 28 April 2010 / Accepted: 10 May 2010 / Published: 18 May 2010 Abstract: Travelers to tropical countries are at risk for a variety of infectious diseases. In some cases effective vaccinations are available, but for other infections chemoprophylaxis can be offered. Malaria prevention has become increasingly complex as Plasmodium species become resistant to available drugs. In certain high risk settings, antibiotics can be used to prevent leptospirosis, scrub typhus and other infections. Post-exposure prophylaxis is appropriate for selected virulent infections. In this article the evidence for chemoprophylaxis will be reviewed. Keywords: chemoprophylaxis; malaria; leptospirosis; scrub typhus; diarrhea 1. Introduction Chemoprophylaxis is the administration of drug to prevent the development of a disease. This review will focus on the use of medications to prevent tropical infectious diseases. The use of chemo- prophylactic agents is based on knowledge of the epidemiology and clinical implications of the infectious diseases from which protection is sought. Generally, chemoprophylaxis is taken for diseases that are common, or where the clinical impact of infection is high. Drugs may be taken before exposure (pre-exposure prophylaxis) or after potential exposure to an infectious agent (post-exposure prophylaxis). In addition to the severity and frequency of the disease, the tolerability, toxicity and ecological implications of the medications being used are important considerations in whether drugs are prescribed and taken. -

(Artemether / Lumefantrine 20Mg/120 Mg) Tablets
SUMMARY OF PRODUCT CHARACTERISTICS Page 1 of 13 1. NAME OF THE MEDICINAL PRODUCT Lumartem (Artemether / Lumefantrine 20mg/120 mg) Tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains: 20 mg artemether and 120 mg lumefantrine. For a full list of excipients see section 6.1. 3. PHARMACEUTICAL FORM Yellow coloured, circular, uncoated, flat faced, bevelled edged, matt finished tablets with a break line on one side and plain on the other side. The scoreline is only to facilitate breaking for ease of swallowing and not to divide into equal doses. 4. CLINICAL PARTICULARS 4.1 Therapeutic indication Artemether 20 mg and Lumefantrine 120 mg Tablets is indicated for the treatment of uncomplicated cases of malaria due to Plasmodium falciparum in adults, children and infants of 5 kg and above. The most recent official guidelines on the appropriate use of antimalarial agents and local information on the prevalence of resistance to antimalarial drugs must be taken into consideration for deciding on the appropriateness of therapy with Artemether 20 mg and Lumefantrine 120 mg Tablets. Official guidance will normally include WHO (http://whqlibdoc.who.int/publications/2010/9789241547925_eng.pdf) and local health authorities’ guidelines (see also sections 4.4 and 5.1). 4.2 Posology and method of administration Tablets for oral administration. Page 2 of 13 Number of Artemether 20 mg and Lumefantrine 120 mg Tablets for treatment according to weight bands st nd rd Weight range 1 day of 2 day of 3 day treatment treatment of treatment ≥ 5kg -

Drug Resistance in Malaria Eugene Mark Department of Biochemistry University of Ghana
http://www.inosr.net/inosr-scientific-research/ INOSR Scientific Research 4(1): 1-12, 2018. Eugene ©INOSR PUBLICATIONS International Network Organization for Scientific Research ISSN: 2705-1706 Drug Resistance in Malaria Eugene Mark Department of Biochemistry University of Ghana ABSTRACT Drug resistant malaria is primarily caused Established and strong drug pressure by Plasmodium falciparum, a species combined with low antiparasitic immunity highly prevalent in tropical. It causes probably explains the multidrug-resistance severe fever or anaemia that leads to more encountered in the forests of South-east than a million deaths each year. The Asia and South America. In Africa, emergence of chloroquine resistance has frequent genetic recombination in been associated with a dramatic increase Plasmodium originate from a high level of in malaria mortality among inhabitants of malaria transmission, and falciparum some endemic regions. The mechanisms of chloroquine-resistant prevalence seems to resistance for amino-alcohols (quinine, stabilize at the same level as chloroquine- mefloquine and halofantrine) are still sensitive malaria. Nevertheless, resistance unclear. Epidemiological studies have levels may differ according to place and established that the frequency of time. In vivo and in vitro tests do not chloroquine resistant mutants varies provide an adequate accurate map of among isolated parasite populations, while resistance. Biochemical tools at a low cost resistance to antifolates is highly prevalent are urgently needed for prospective in most malarial endemic countries. monitoring of resistance. Keywords: Drug, Resistance, Malaria. INTRODUCTION Malaria is a mosquito-borne infectious reproduce [3]. Five species of Plasmodium disease that affects humans and other can infect and be spread by humans. -

Hydroxychloroquine: a Physiologically-Based Pharmacokinetic Model in the Context of Cancer-Related Autophagy Modulation S
Supplemental material to this article can be found at: http://jpet.aspetjournals.org/content/suppl/2018/02/08/jpet.117.245639.DC1 1521-0103/365/3/447–459$35.00 https://doi.org/10.1124/jpet.117.245639 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS J Pharmacol Exp Ther 365:447–459, June 2018 Copyright ª 2018 by The American Society for Pharmacology and Experimental Therapeutics Hydroxychloroquine: A Physiologically-Based Pharmacokinetic Model in the Context of Cancer-Related Autophagy Modulation s Keagan P. Collins, Kristen M. Jackson, and Daniel L. Gustafson School of Biomedical Engineering (K.P.C., K.M.J., D.L.G.) and Department of Clinical Sciences (D.L.G.), Colorado State University, Fort Collins, Colorado; and University of Colorado Cancer Center, Aurora, Colorado (D.L.G.) Received October 10, 2017; accepted February 6, 2018 ABSTRACT Downloaded from Hydroxychloroquine (HCQ) is a lysosomotropic autophagy in- adapted to simulate human HCQ exposure in whole blood and hibitor being used in over 50 clinical trials either alone or in urine through allometric scaling and species-specific parameter combination with chemotherapy. Pharmacokinetic (PK) and modification. The human model accurately simulated average pharmacodynamic (PD) studies with HCQ have shown that drug steady-state concentrations (Css) of those observed in five exposure in the blood does not correlate with autophagy different HCQ combination clinical trials across seven different inhibition in either peripheral blood mononuclear cells or tumor doses, which was then expanded by comparison of the Css tissue. To better explain this PK/PD disconnect, a PBPK was distribution in a virtual human population at this range of doses. -

Current Antimalarial Therapies and Advances in the Development of Semi-Synthetic Artemisinin Derivatives
Anais da Academia Brasileira de Ciências (2018) 90(1 Suppl. 2): 1251-1271 (Annals of the Brazilian Academy of Sciences) Printed version ISSN 0001-3765 / Online version ISSN 1678-2690 http://dx.doi.org/10.1590/0001-3765201820170830 www.scielo.br/aabc | www.fb.com/aabcjournal Current Antimalarial Therapies and Advances in the Development of Semi-Synthetic Artemisinin Derivatives LUIZ C.S. PINHEIRO1, LÍVIA M. FEITOSA1,2, FLÁVIA F. DA SILVEIRA1,2 and NUBIA BOECHAT1 1Fundação Oswaldo Cruz, Instituto de Tecnologia em Fármacos Farmanguinhos, Fiocruz, Departamento de Síntese de Fármacos, Rua Sizenando Nabuco, 100, Manguinhos, 21041-250 Rio de Janeiro, RJ, Brazil 2Universidade Federal do Rio de Janeiro, Programa de Pós-Graduação em Química, Avenida Athos da Silveira Ramos, 149, Cidade Universitária, 21941-909 Rio de Janeiro, RJ, Brazil Manuscript received on October 17, 2017; accepted for publication on December 18, 2017 ABSTRACT According to the World Health Organization, malaria remains one of the biggest public health problems in the world. The development of resistance is a current concern, mainly because the number of safe drugs for this disease is limited. Artemisinin-based combination therapy is recommended by the World Health Organization to prevent or delay the onset of resistance. Thus, the need to obtain new drugs makes artemisinin the most widely used scaffold to obtain synthetic compounds. This review describes the drugs based on artemisinin and its derivatives, including hybrid derivatives and dimers, trimers and tetramers that contain an endoperoxide bridge. This class of compounds is of extreme importance for the discovery of new drugs to treat malaria. Key words: malaria, Plasmodium falciparum, artemisinin, hybrid. -

PHARMACOLOGY of NEWER ANTIMALARIAL DRUGS: REVIEW ARTICLE Bhuvaneshwari1, Souri S
REVIEW ARTICLE PHARMACOLOGY OF NEWER ANTIMALARIAL DRUGS: REVIEW ARTICLE Bhuvaneshwari1, Souri S. Kondaveti2 HOW TO CITE THIS ARTICLE: Bhuvaneshwari, Souri S. Kondaveti. ‖Pharmacology of Newer Antimalarial Drugs: Review Article‖. Journal of Evidence based Medicine and Healthcare; Volume 2, Issue 4, January 26, 2015; Page: 431-439. ABSTRACT: Malaria is currently is a major health problem, which has been attributed to wide spread resistance of the anopheles mosquito to the economical insecticides and increasing prevalence of drug resistance to plasmodium falciparum. Newer drugs are needed as there is a continual threat of emergence of resistance to both artemisins and the partner medicines. Newer artemisinin compounds like Artemisone, Artemisnic acid, Sodium artelinate, Arteflene, Synthetic peroxides like arterolane which is a synthetic trioxolane cognener of artemisins, OZ439 a second generation synthetic peroxide are under studies. Newer artemisinin combinations include Arterolane(150mg) + Piperaquine (750mg), DHA (120mg) + Piperaquine(960mg) (1:8), Artesunate + Pyronardine (1:3), Artesunate + Chlorproguanil + Dapsone, Artemisinin (125mg) + Napthoquine (50mg) single dose and Artesunate + Ferroquine.Newer drugs under development including Transmission blocking compounds like Bulaquine, Etaquine, Tafenoquine, which are primaquine congeners, Spiroindalone, Trioxaquine DU 1302, Epoxamicin, Quinolone 3 Di aryl ether. Newer drugs targeting blood & liver stages which include Ferroquine, Albitiazolium – (SAR – 97276). Older drugs with new use in malaria like beta blockers, calcium channel blockers, protease inhibitors, Dihydroorotate dehydrogenase inhibitors, methotrexate, Sevuparin sodium, auranofin, are under preclinical studies which also target blood and liver stages. Antibiotics like Fosmidomycin and Azithromycin in combination with Artesunate, Chloroquine, Clindamycin are also undergoing trials for treatment of malaria. Vaccines - RTS, S– the most effective malarial vaccine tested to date.