4106.Full.Pdf
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
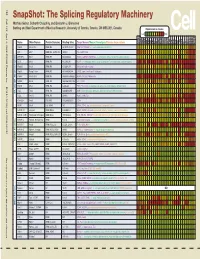
Snapshot: the Splicing Regulatory Machinery Mathieu Gabut, Sidharth Chaudhry, and Benjamin J
192 Cell SnapShot: The Splicing Regulatory Machinery Mathieu Gabut, Sidharth Chaudhry, and Benjamin J. Blencowe 133 Banting and Best Department of Medical Research, University of Toronto, Toronto, ON M5S 3E1, Canada Expression in mouse , April4, 2008©2008Elsevier Inc. Low High Name Other Names Protein Domains Binding Sites Target Genes/Mouse Phenotypes/Disease Associations Amy Ceb Hip Hyp OB Eye SC BM Bo Ht SM Epd Kd Liv Lu Pan Pla Pro Sto Spl Thy Thd Te Ut Ov E6.5 E8.5 E10.5 SRp20 Sfrs3, X16 RRM, RS GCUCCUCUUC SRp20, CT/CGRP; −/− early embryonic lethal E3.5 9G8 Sfrs7 RRM, RS, C2HC Znf (GAC)n Tau, GnRH, 9G8 ASF/SF2 Sfrs1 RRM, RS RGAAGAAC HipK3, CaMKIIδ, HIV RNAs; −/− embryonic lethal, cond. KO cardiomyopathy SC35 Sfrs2 RRM, RS UGCUGUU AChE; −/− embryonic lethal, cond. KO deficient T-cell maturation, cardiomyopathy; LS SRp30c Sfrs9 RRM, RS CUGGAUU Glucocorticoid receptor SRp38 Fusip1, Nssr RRM, RS ACAAAGACAA CREB, type II and type XI collagens SRp40 Sfrs5, HRS RRM, RS AGGAGAAGGGA HipK3, PKCβ-II, Fibronectin SRp55 Sfrs6 RRM, RS GGCAGCACCUG cTnT, CD44 DOI 10.1016/j.cell.2008.03.010 SRp75 Sfrs4 RRM, RS GAAGGA FN1, E1A, CD45; overexpression enhances chondrogenic differentiation Tra2α Tra2a RRM, RS GAAARGARR GnRH; overexpression promotes RA-induced neural differentiation SR and SR-Related Proteins Tra2β Sfrs10 RRM, RS (GAA)n HipK3, SMN, Tau SRm160 Srrm1 RS, PWI AUGAAGAGGA CD44 SWAP Sfrs8 RS, SWAP ND SWAP, CD45, Tau; possible asthma susceptibility gene hnRNP A1 Hnrnpa1 RRM, RGG UAGGGA/U HipK3, SMN2, c-H-ras; rheumatoid arthritis, systemic lupus -

Nuclear PTEN Safeguards Pre-Mrna Splicing to Link Golgi Apparatus for Its Tumor Suppressive Role
ARTICLE DOI: 10.1038/s41467-018-04760-1 OPEN Nuclear PTEN safeguards pre-mRNA splicing to link Golgi apparatus for its tumor suppressive role Shao-Ming Shen1, Yan Ji2, Cheng Zhang1, Shuang-Shu Dong2, Shuo Yang1, Zhong Xiong1, Meng-Kai Ge1, Yun Yu1, Li Xia1, Meng Guo1, Jin-Ke Cheng3, Jun-Ling Liu1,3, Jian-Xiu Yu1,3 & Guo-Qiang Chen1 Dysregulation of pre-mRNA alternative splicing (AS) is closely associated with cancers. However, the relationships between the AS and classic oncogenes/tumor suppressors are 1234567890():,; largely unknown. Here we show that the deletion of tumor suppressor PTEN alters pre-mRNA splicing in a phosphatase-independent manner, and identify 262 PTEN-regulated AS events in 293T cells by RNA sequencing, which are associated with significant worse outcome of cancer patients. Based on these findings, we report that nuclear PTEN interacts with the splicing machinery, spliceosome, to regulate its assembly and pre-mRNA splicing. We also identify a new exon 2b in GOLGA2 transcript and the exon exclusion contributes to PTEN knockdown-induced tumorigenesis by promoting dramatic Golgi extension and secretion, and PTEN depletion significantly sensitizes cancer cells to secretion inhibitors brefeldin A and golgicide A. Our results suggest that Golgi secretion inhibitors alone or in combination with PI3K/Akt kinase inhibitors may be therapeutically useful for PTEN-deficient cancers. 1 Department of Pathophysiology, Key Laboratory of Cell Differentiation and Apoptosis of Chinese Ministry of Education, Shanghai Jiao Tong University School of Medicine (SJTU-SM), Shanghai 200025, China. 2 Institute of Health Sciences, Shanghai Institutes for Biological Sciences of Chinese Academy of Sciences and SJTU-SM, Shanghai 200025, China. -

Transcriptomic Analysis of Short-Fruit 1 (Sf1)
www.nature.com/scientificreports OPEN Transcriptomic analysis of short- fruit 1 (sf1) reveals new insights into the variation of fruit-related Received: 15 November 2016 Accepted: 20 April 2017 traits in Cucumis sativus Published: xx xx xxxx Lina Wang, Chenxing Cao, Shuangshuang Zheng, Haiyang Zhang, Panjing Liu, Qian Ge, Jinrui Li & Zhonghai Ren Fruit size is an important quality trait in different market classes ofCucumis sativus L., an economically important vegetable cultivated worldwide, but the genetic and molecular mechanisms that control fruit size are largely unknown. In this study, we isolated a natural cucumber mutant, short fruit 1 (sf1), caused by a single recessive Mendelian factor, from the North China-type inbred line CNS2. In addition to significantly decreased fruit length, other fruit-related phenotypic variations were also observed in sf1 compared to the wild-type (WT) phenotype, indicating that sf1 might have pleiotropic effects. Microscopic imaging showed that fruit cell size in sf1 was much larger than that in WT, suggesting that the short fruit phenotype in sf1 is caused by decreased cell number. Fine mapping revealed that sf1 was localized to a 174.3 kb region on chromosome 6. Similarly, SNP association analysis of bulked segregant RNA-Seq data showed increased SNP frequency in the same region of chromosome 6. In addition, transcriptomic analysis revealed that sf1 might control fruit length through the fine-tuning of cytokinin and auxin signalling, gibberellin biosynthesis and signal transduction in cucumber fruits. Overall, our results provide important information for further study of fruit length and other fruit-related features in cucumber. Cucumber (Cucumis sativus L., 2n = 14), a member of the family Cucurbitaceae, is one of the most economically important vegetable crops cultivated throughout the world. -

Producing Cells of the Testis, Ovary and Adrenal Gland F
RESEARCH ARTICLE 4561 Development 139, 4561-4570 (2012) doi:10.1242/dev.087247 © 2012. Published by The Company of Biologists Ltd In vivo evidence for the crucial role of SF1 in steroid- producing cells of the testis, ovary and adrenal gland F. William Buaas1,*, Jennifer R. Gardiner1, Sally Clayton1, Pierre Val2 and Amanda Swain1,‡ SUMMARY Adrenal and gonadal steroids are essential for life and reproduction. The orphan nuclear receptor SF1 (NR5A1) has been shown to regulate the expression of enzymes involved in steroid production in vitro. However, the in vivo role of this transcription factor in steroidogenesis has not been elucidated. In this study, we have generated steroidogenic-specific Cre-expressing mice to lineage mark and delete Sf1 in differentiated steroid-producing cells of the testis, the ovary and the adrenal gland. Our data show that SF1 is a regulator of the expression of steroidogenic genes in all three organs. In addition, Sf1 deletion leads to a radical change in cell morphology and loss of identity. Surprisingly, sexual development and reproduction in mutant animals were not compromised owing, in part, to the presence of a small proportion of SF1-positive cells. In contrast to the testis and ovary, the mutant adult adrenal gland showed a lack of Sf1-deleted cells and our studies suggest that steroidogenic adrenal cells during foetal stages require Sf1 to give rise to the adult adrenal population. This study is the first to show the in vivo requirements of SF1 in steroidogenesis and provides novel data on the cellular consequences of the loss of this protein specifically within steroid-producing cells. -

Structural Insights Into How Prp5 Proofreads the Pre-Mrna Branch Site
Article Structural insights into how Prp5 proofreads the pre-mRNA branch site https://doi.org/10.1038/s41586-021-03789-5 Zhenwei Zhang1, Norbert Rigo2, Olexandr Dybkov2, Jean-Baptiste Fourmann2, Cindy L. Will2, Vinay Kumar2, Henning Urlaub3,4, Holger Stark1 ✉ & Reinhard Lührmann2 ✉ Received: 10 December 2020 Accepted: 30 June 2021 During the splicing of introns from precursor messenger RNAs (pre-mRNAs), the U2 Published online: 4 August 2021 small nuclear ribonucleoprotein (snRNP) must undergo stable integration into the Open access spliceosomal A complex—a poorly understood, multistep process that is facilitated by Check for updates the DEAD-box helicase Prp5 (refs. 1–4). During this process, the U2 small nuclear RNA (snRNA) forms an RNA duplex with the pre-mRNA branch site (the U2–BS helix), which is proofread by Prp5 at this stage through an unclear mechanism5. Here, by deleting the branch-site adenosine (BS-A) or mutating the branch-site sequence of an actin pre-mRNA, we stall the assembly of spliceosomes in extracts from the yeast Saccharomyces cerevisiae directly before the A complex is formed. We then determine the three-dimensional structure of this newly identifed assembly intermediate by cryo-electron microscopy. Our structure indicates that the U2–BS helix has formed in this pre-A complex, but is not yet clamped by the HEAT domain of the Hsh155 protein (Hsh155HEAT), which exhibits an open conformation. The structure further reveals a large-scale remodelling/repositioning of the U1 and U2 snRNPs during the formation of the A complex that is required to allow subsequent binding of the U4/U6.U5 tri-snRNP, but that this repositioning is blocked in the pre-A complex by the presence of Prp5. -
Possible Misdiagnosis of 46,XX Testicular Disorders of Sex
Int. J. Med. Sci. 2020, Vol. 17 1136 Ivyspring International Publisher International Journal of Medical Sciences 2020; 17(9): 1136-1141. doi: 10.7150/ijms.46058 Research Paper Possible misdiagnosis of 46,XX testicular disorders of sex development in infertile males Tong Chen1,2†, Linlin Tian1,3†, Xianlong Wang1, Demin Fan4, Gang Ma1, Rong Tang1, Xujun Xuan1,5 1. Center for Reproductive Medicine, Shandong University, National Research Center for Assisted Reproductive Technology and Reproductive Genetics; The Key Laboratory for Reproductive Endocrinology of Ministry of Education, Jinan, Shandong 250021, P.R. China 2. Department of Pediatric Surgery, Shanghai Children's Hospital, Shanghai Jiao Tong University, Shanghai, 200062, P.R. China 3. Department of microbiology, Faculty of Basic Medical Sciences, Guilin Medical University, Guilin, Guangxi 541004, P.R. China 4. Department of Urology, Shandong Provincial Qianfoshan Hospital, Jinan, Shandong 250002, P.R. China 5. Department of Andrology, The Seventh Affiliated Hospital, Sun Yat-sen University, Shenzhen, 518107, P.R. China. †Tong Chen and Linlin Tian contributed equally to this study. Corresponding author: Xujun Xuan, Department of Andrology, The Seventh Affiliated Hospital, Sun Yat-sen University, Shenzhen, 518107, P.R. China. E-mail: [email protected] © The author(s). This is an open access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/). See http://ivyspring.com/terms for full terms and conditions. Received: 2020.03.16; Accepted: 2020.04.23; Published: 2020.05.11 Abstract Objectives: The 46,XX disorders of sex development (DSD) is a rare genetic cause of male infertility and possible misdiagnosis of this condition has never been reported. -

Identification of Tat-SF1 Cellular Targets by Exon Array Analysis Reveals Dual Roles in Transcription and Splicing
Identification of Tat-SF1 cellular targets by exon array analysis reveals dual roles in transcription and splicing HEATHER B. MILLER,1,2,7 TIMOTHY J. ROBINSON,1,2,3,4 RALUCA GORDAˆ N,5,8 ALEXANDER J. HARTEMINK,5 and MARIANO A. GARCIA-BLANCO1,2,6 1Department of Molecular Genetics and Microbiology, Duke University Medical Center, Durham, North Carolina 27710, USA 2Center for RNA Biology, Duke University Medical Center, Durham, North Carolina 27710, USA 3Medical Scientist Training Program, Duke University Medical Center, Durham, North Carolina 27710, USA 4Program in Molecular Cancer Biology, Duke University Medical Center, Durham, North Carolina 27710, USA 5Department of Computer Science, Duke University, Durham, North Carolina 27708, USA 6Department of Medicine, Duke University Medical Center, Durham, North Carolina 27710, USA ABSTRACT Tat specific factor 1 (Tat-SF1) interacts with components of both the transcription and splicing machineries and has been classified as a transcription-splicing factor. Although its function as an HIV-1 dependency factor has been investigated, relatively little is known about the cellular functions of Tat-SF1. To identify target genes of Tat-SF1, we utilized a combination of RNAi and exon-specific microarrays. These arrays, which survey genome-wide changes in transcript and individual exon levels, revealed 450 genes with transcript level changes upon Tat-SF1 depletion. Strikingly, 98% of these target genes were down-regulated upon depletion, indicating that Tat-SF1 generally activates gene expression. We also identified 89 genes that showed differential exon level changes after Tat-SF1 depletion. The 89 genes showed evidence of many different types of alternative exon use consistent with the regulation of transcription initiation sites and RNA processing. -

The RNA Splicing Response to DNA Damage
Biomolecules 2015, 5, 2935-2977; doi:10.3390/biom5042935 OPEN ACCESS biomolecules ISSN 2218-273X www.mdpi.com/journal/biomolecules/ Review The RNA Splicing Response to DNA Damage Lulzim Shkreta and Benoit Chabot * Département de Microbiologie et d’Infectiologie, Faculté de Médecine et des Sciences de la Santé, Université de Sherbrooke, Sherbrooke, QC J1E 4K8, Canada; E-Mail: [email protected] * Author to whom correspondence should be addressed; E-Mail: [email protected]; Tel.: +1-819-821-8000 (ext. 75321); Fax: +1-819-820-6831. Academic Editors: Wolf-Dietrich Heyer, Thomas Helleday and Fumio Hanaoka Received: 12 August 2015 / Accepted: 16 October 2015 / Published: 29 October 2015 Abstract: The number of factors known to participate in the DNA damage response (DDR) has expanded considerably in recent years to include splicing and alternative splicing factors. While the binding of splicing proteins and ribonucleoprotein complexes to nascent transcripts prevents genomic instability by deterring the formation of RNA/DNA duplexes, splicing factors are also recruited to, or removed from, sites of DNA damage. The first steps of the DDR promote the post-translational modification of splicing factors to affect their localization and activity, while more downstream DDR events alter their expression. Although descriptions of molecular mechanisms remain limited, an emerging trend is that DNA damage disrupts the coupling of constitutive and alternative splicing with the transcription of genes involved in DNA repair, cell-cycle control and apoptosis. A better understanding of how changes in splice site selection are integrated into the DDR may provide new avenues to combat cancer and delay aging. -

The Long Non-Coding RNA Gomafu Is Acutely Regulated in Response to Neuronal Activation and Involved in Schizophrenia-Associated Alternative Splicing
Molecular Psychiatry (2014) 19, 486–494 & 2014 Macmillan Publishers Limited All rights reserved 1359-4184/14 www.nature.com/mp ORIGINAL ARTICLE The long non-coding RNA Gomafu is acutely regulated in response to neuronal activation and involved in schizophrenia-associated alternative splicing G Barry1, JA Briggs2, DP Vanichkina1, EM Poth3, NJ Beveridge4,5, VS Ratnu6, SP Nayler2, K Nones7,JHu8, TW Bredy6, S Nakagawa9, F Rigo10, RJ Taft1, MJ Cairns4,5, S Blackshaw3, EJ Wolvetang2 and JS Mattick1,11,12 Schizophrenia (SZ) is a complex disease characterized by impaired neuronal functioning. Although defective alternative splicing has been linked to SZ, the molecular mechanisms responsible are unknown. Additionally, there is limited understanding of the early transcriptomic responses to neuronal activation. Here, we profile these transcriptomic responses and show that long non-coding RNAs (lncRNAs) are dynamically regulated by neuronal activation, including acute downregulation of the lncRNA Gomafu, previously implicated in brain and retinal development. Moreover, we demonstrate that Gomafu binds directly to the splicing factors QKI and SRSF1 (serine/arginine-rich splicing factor 1) and dysregulation of Gomafu leads to alternative splicing patterns that resemble those observed in SZ for the archetypal SZ-associated genes DISC1 and ERBB4. Finally, we show that Gomafu is downregulated in post-mortem cortical gray matter from the superior temporal gyrus in SZ. These results functionally link activity-regulated lncRNAs and alternative splicing in neuronal function and suggest that their dysregulation may contribute to neurological disorders. Molecular Psychiatry (2014) 19, 486–494; doi:10.1038/mp.2013.45; published online 30 April 2013 Keywords: alternative splicing; Gomafu; neuronal activation; quaking homolog; schizophrenia INTRODUCTION LncRNAs are arbitrarily defined as transcripts longer than 200 bp 10 Schizophrenia (SZ) disorder, a debilitating mental disorder that possess no protein-coding capacity. -

The Emerging Roles of the RNA Binding Protein QKI in Cardiovascular Development and Function
fcell-09-668659 June 14, 2021 Time: 13:17 # 1 MINI REVIEW published: 16 June 2021 doi: 10.3389/fcell.2021.668659 The Emerging Roles of the RNA Binding Protein QKI in Cardiovascular Development and Function Xinyun Chen1,2†, Jianwen Yin3†, Dayan Cao1, Deyong Xiao1, Zhongjun Zhou4, Ying Liu1* and Weinian Shou1* 1 Department of Pediatrics, Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN, United States, 2 Guangdong Key Laboratory for Genome Stability and Human Disease Prevention, Department of Biochemistry and Molecular Biology, School of Basic Medical Sciences, Shenzhen University, Shenzhen, China, 3 Department of Foot, Ankle and Hand Surgery, Shenzhen Second People’s Hospital, First Affiliated Hospital of Shenzhen University, Shenzhen, China, 4 Faculty of Medicine, School of Biomedical Sciences, The University of Hong Kong, Hong Kong Edited by: Janine M. Van Gils, Leiden University Medical Center, RNA binding proteins (RBPs) have a broad biological and physiological function and Netherlands are critical in regulating pre-mRNA posttranscriptional processing, intracellular migration, Reviewed by: Jianbo Wang, and mRNA stability. QKI, also known as Quaking, is a member of the signal transduction University of Alabama at Birmingham, and activation of RNA (STAR) family, which also belongs to the heterogeneous nuclear United States ribonucleoprotein K- (hnRNP K-) homology domain protein family. There are three Wuqiang Zhu, Mayo Clinic Arizona, United States major alternatively spliced isoforms, QKI-5, QKI-6, and QKI-7, differing in carboxy- *Correspondence: terminal domains. They share a common RNA binding property, but each isoform can Ying Liu regulate pre-mRNA splicing, transportation or stability differently in a unique cell type- [email protected] Weinian Shou specific manner. -

A Fast, Non-Radioactive Method to Identify in Vivo Targets of RNA-Binding Proteins
bioRxiv preprint doi: https://doi.org/10.1101/158410; this version posted July 1, 2017. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. uvCLAP: a fast, non-radioactive method to identify in vivo targets of RNA-binding proteins Daniel Maticzka1,4, Ibrahim Avsar Ilik2,4, Tugce Aktas2,4, Rolf Backofen1,3,# and Asifa Akhtar2,#* 1 Bioinformatics Group, Department of Computer Science, University of Freiburg, Freiburg, Germany 2 Max Planck Institute of Immunobiology and Epigenetics, 79108 Freiburg im Breisgau, Germany 3 Centre for Biological Signalling Studies (BIOSS), University of Freiburg, Freiburg, Germany 4 These authors contributed equally to this work # Corresponding author [email protected] Phone: +49 761 203 7461 Fax: +49 761 203 7462 # Corresponding author, * Lead contact [email protected] Phone: +49 (0)7615108565 Fax: +49 (0)7615108566 1 bioRxiv preprint doi: https://doi.org/10.1101/158410; this version posted July 1, 2017. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Abstract RNA-binding proteins (RBPs) play important and essential roles in eukaryotic gene expression regulating splicing, localization, translation and stability of mRNAs. Understanding the exact contribution of RBPs to gene regulation is crucial as many RBPs are frequently mis-regulated in several neurological diseases and certain cancers. While recently developed techniques provide binding sites of RBPs, they are labor-intensive and generally rely on radioactive labeling of RNA. With more than 1,000 RBPs in a human cell, it is imperative to develop easy, robust, reproducible and high-throughput methods to determine in vivo targets of RBPs. -

Alternatively Spliced Genes
1 Alternatively Spliced Genes Jane Y. Wu1,2,LiyaYuan1 and Necat Havlioglu1 1Washington University School of Medicine, St. Louis, MO, USA 2John F. Kennedy Center for Research on Human Development, Vanderbilt University Medical Center, Nashville, TN, USA 1 Pre-mRNA Splicing and Splicing Machinery 3 1.1 Splicing Machinery: Spliceosome 3 1.2 Splicing Signals 4 1.3 Spliceosomal UsnRNP Biogenesis 12 1.4 Spliceosome Assembly 13 1.5 Biochemical Mechanisms of pre-mRNA Splicing 17 2 Alternative pre-mRNA Splicing 17 2.1 Alternative Splicing and its Role in Regulating Gene Activities and Generating Genetic Diversity 17 2.1.1 Different Patterns of Alternative Splicing 17 2.1.2 Alternative Splicing and Genetic Diversity 18 2.2 Mechanisms Underlying Alternative Splicing Regulation 19 2.2.1 Splicing Signals and Splicing Regulatory Elements 20 2.2.2 Trans-acting Splicing Regulators 23 2.3 Tissue-specific and Developmentally Regulated Alternative Splicing 26 2.4 Regulation of Alternative Splicing in Response to Extracellular Stimuli 27 3 Pre-mRNA Splicing and Human Diseases 28 3.1 Splicing Defects in Human Diseases 28 3.2 Molecular Mechanisms Underlying Splicing Defects Associated with Disease 33 4 Perspectives on Diagnosis and Treatment of Diseases Caused by pre-mRNA Splicing Defects 36 4.1 Diagnosis of Human Diseases Caused by Splicing Defects 36 2 Alternatively Spliced Genes 4.2 Potential Therapeutic Approaches 37 4.2.1 Oligonucleotide-based Approaches: Antisense, RNAi, and Chimeric Molecules 37 4.2.2 Ribozymes 37 4.2.3 SMaRT 38 4.2.4 Chemical Compounds 38 5 Concluding Remarks 38 Acknowledgment 39 Bibliography 39 Books and Reviews 39 Keywords Pre-mRNA Nascent transcripts that are precursors of mature messenger RNAs.