Epigenetic Regulation of Alternative Splicing: How Lncrnas Tailor the Message
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Ribonucleotidyl Transferase USIP-1 Acts with SART3 to Promote U6 Snrna Recycling Stefan Ruegger¨ 1,2, Takashi S
3344–3357 Nucleic Acids Research, 2015, Vol. 43, No. 6 Published online 09 March 2015 doi: 10.1093/nar/gkv196 The ribonucleotidyl transferase USIP-1 acts with SART3 to promote U6 snRNA recycling Stefan Ruegger¨ 1,2, Takashi S. Miki1, Daniel Hess1 and Helge Großhans1,* 1Friedrich Miescher Institute for Biomedical Research, Maulbeerstrasse 66, CH-4058 Basel, Switzerland and 2University of Basel, Petersplatz 1, CH-4003 Basel, Switzerland Received October 13, 2014; Revised February 10, 2015; Accepted February 24, 2015 ABSTRACT rearrangements of U6 then lead to disruption of the U4– U6 snRNA base-pairing in favor of U6–U2 snRNA base- The spliceosome is a large molecular machine that pairing, resulting in release of U4 snRNA (3). Moreover, U6 serves to remove the intervening sequences that binding to the 5 splice site displaces the U1 snRNA leading are present in most eukaryotic pre-mRNAs. At its to its release from the spliceosome (3). core are five small nuclear ribonucleoprotein com- Following execution of the splicing step, U2, U5 and U6 plexes, the U1, U2, U4, U5 and U6 snRNPs, which snRNPs and the resected intron lariat are released and fur- undergo dynamic rearrangements during splicing. ther disassembled through mechanisms that are not well un- Their reutilization for subsequent rounds of splic- derstood (1). Reuse of the snRNPs for further rounds of ing requires reversion to their original configura- splicing thus requires regeneration of their distinct, initial tions, but little is known about this process. Here, we conformations and interactions. For the U6 snRNP,this ‘re- / show that ZK863.4/USIP-1 (U Six snRNA-Interacting cycling’ includes the reformation of a U4 U6 snRNP. -

Differential Analysis of Gene Expression and Alternative Splicing
i bioRxiv preprint doi: https://doi.org/10.1101/2020.08.23.234237; this version posted August 24, 2020. The copyright holder for this preprint i (which was not certified by peer review)“main” is the author/funder, — 2020/8/23 who has — granted 9:38 — bioRxiv page a1 license — #1 to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. i i EmpiReS: Differential Analysis of Gene Expression and Alternative Splicing Gergely Csaba∗, Evi Berchtold*,y, Armin Hadziahmetovic, Markus Gruber, Constantin Ammar and Ralf Zimmer Department of Informatics, Ludwig-Maximilians-Universitat¨ Munchen,¨ 80333, Munich, Germany ABSTRACT to translation. Different transcripts of the same gene are often expressed at the same time, possibly at different abundance, While absolute quantification is challenging in high- yielding a defined mixture of isoforms. Changes to this throughput measurements, changes of features composition are further referred to as differential alternative between conditions can often be determined with splicing (DAS). Various diseases can be linked to DAS high precision. Therefore, analysis of fold changes events (3). Most hallmarks of cancer like tumor progression is the standard method, but often, a doubly and immune escape (4, 5) can be attributed to changes in differential analysis of changes of changes is the transcript composition (6). DAS plays a prominent role required. Differential alternative splicing is an in various types of muscular dystrophy (MD) (7), splicing example of a doubly differential analysis, i.e. fold intervention by targeted exon removal is a therapeutic option changes between conditions for different isoforms in Duchenne MD (8). -

Androgen Receptor Interacting Proteins and Coregulators Table
ANDROGEN RECEPTOR INTERACTING PROTEINS AND COREGULATORS TABLE Compiled by: Lenore K. Beitel, Ph.D. Lady Davis Institute for Medical Research 3755 Cote Ste Catherine Rd, Montreal, Quebec H3T 1E2 Canada Telephone: 514-340-8260 Fax: 514-340-7502 E-Mail: [email protected] Internet: http://androgendb.mcgill.ca Date of this version: 2010-08-03 (includes articles published as of 2009-12-31) Table Legend: Gene: Official symbol with hyperlink to NCBI Entrez Gene entry Protein: Protein name Preferred Name: NCBI Entrez Gene preferred name and alternate names Function: General protein function, categorized as in Heemers HV and Tindall DJ. Endocrine Reviews 28: 778-808, 2007. Coregulator: CoA, coactivator; coR, corepressor; -, not reported/no effect Interactn: Type of interaction. Direct, interacts directly with androgen receptor (AR); indirect, indirect interaction; -, not reported Domain: Interacts with specified AR domain. FL-AR, full-length AR; NTD, N-terminal domain; DBD, DNA-binding domain; h, hinge; LBD, ligand-binding domain; C-term, C-terminal; -, not reported References: Selected references with hyperlink to PubMed abstract. Note: Due to space limitations, all references for each AR-interacting protein/coregulator could not be cited. The reader is advised to consult PubMed for additional references. Also known as: Alternate gene names Gene Protein Preferred Name Function Coregulator Interactn Domain References Also known as AATF AATF/Che-1 apoptosis cell cycle coA direct FL-AR Leister P et al. Signal Transduction 3:17-25, 2003 DED; CHE1; antagonizing regulator Burgdorf S et al. J Biol Chem 279:17524-17534, 2004 CHE-1; AATF transcription factor ACTB actin, beta actin, cytoplasmic 1; cytoskeletal coA - - Ting HJ et al. -

Identification of a Gene Coding for a Protein Possessing Shared Tumor Epitopes Capable of Inducing HLA-A24-Restricted Cytotoxic T Lymphocytes in Cancer Patients1
[CANCER RESEARCH 59, 4056–4063, August 15, 1999] Identification of a Gene Coding for a Protein Possessing Shared Tumor Epitopes Capable of Inducing HLA-A24-restricted Cytotoxic T Lymphocytes in Cancer Patients1 Damu Yang, Masanobu Nakao, Shigeki Shichijo, Teruo Sasatomi, Hideo Takasu, Hajime Matsumoto, Kazunori Mori, Akihiro Hayashi, Hideaki Yamana, Kazuo Shirouzu, and Kyogo Itoh2 Cancer Vaccine Development Division, Kurume University Research Center for Innovative Cancer Therapy [D. Y., M. N., K. I.], and Departments of Surgery [A. H., H. Y., K. S.], Immunology [S. S., T. S., H. T., H. M., K. I.], and Otolaryngology [K. M.], Kurume University School of Medicine, Kurume, 830-0011, Japan ABSTRACT we report a gene encoding epitopes that are capable of inducing CTLs in PBMCs of patients with SCCs and adenocarcinomas. Genes encoding tumor epitopes that are capable of inducing CTLs against adenocarcinomas and squamous cell carcinomas, two major hu- man cancers histologically observed in various organs, have rarely been MATERIALS AND METHODS identified. Here, we report a new gene from cDNA of esophageal cancer cells that encodes a shared tumor antigen recognized by HLA-A2402- Generation of HLA-A2402-restricted CTLs. HLA-A2402-restricted and restricted and tumor-specific CTLs. The sequence of this gene is almost tumor-specific CTLs were established from the PBMCs of an esophageal identical to that of the KIAA0156 gene, which has been registered in cancer patient (HLA-A2402/A2601) by the standard method of mixed lym- phocyte tumor cell culture, -

RNA Components of the Spliceosome Regulate Tissue- and Cancer-Specific Alternative Splicing
Downloaded from genome.cshlp.org on September 29, 2021 - Published by Cold Spring Harbor Laboratory Press Research RNA components of the spliceosome regulate tissue- and cancer-specific alternative splicing Heidi Dvinge,1,2,4 Jamie Guenthoer,3 Peggy L. Porter,3 and Robert K. Bradley1,2 1Computational Biology Program, Public Health Sciences Division, Fred Hutchinson Cancer Research Center, Seattle, Washington 98109, USA; 2Basic Sciences Division, Fred Hutchinson Cancer Research Center, Seattle, Washington 98109, USA; 3Human Biology Division, Fred Hutchinson Cancer Research Center, Seattle, Washington 98109, USA Alternative splicing of pre-mRNAs plays a pivotal role during the establishment and maintenance of human cell types. Characterizing the trans-acting regulatory proteins that control alternative splicing has therefore been the focus of much research. Recent work has established that even core protein components of the spliceosome, which are required for splicing to proceed, can nonetheless contribute to splicing regulation by modulating splice site choice. We here show that the RNA components of the spliceosome likewise influence alternative splicing decisions. Although these small nuclear RNAs (snRNAs), termed U1, U2, U4, U5, and U6 snRNA, are present in equal stoichiometry within the spliceosome, we found that their relative levels vary by an order of magnitude during development, across tissues, and across cancer samples. Physiologically relevant perturbation of individual snRNAs drove widespread gene-specific differences in alternative splic- ing but not transcriptome-wide splicing failure. Genes that were particularly sensitive to variations in snRNA abundance in a breast cancer cell line model were likewise preferentially misspliced within a clinically diverse cohort of invasive breast ductal carcinomas. -
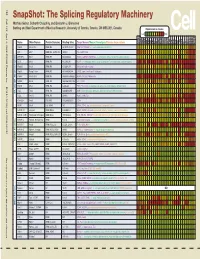
Snapshot: the Splicing Regulatory Machinery Mathieu Gabut, Sidharth Chaudhry, and Benjamin J
192 Cell SnapShot: The Splicing Regulatory Machinery Mathieu Gabut, Sidharth Chaudhry, and Benjamin J. Blencowe 133 Banting and Best Department of Medical Research, University of Toronto, Toronto, ON M5S 3E1, Canada Expression in mouse , April4, 2008©2008Elsevier Inc. Low High Name Other Names Protein Domains Binding Sites Target Genes/Mouse Phenotypes/Disease Associations Amy Ceb Hip Hyp OB Eye SC BM Bo Ht SM Epd Kd Liv Lu Pan Pla Pro Sto Spl Thy Thd Te Ut Ov E6.5 E8.5 E10.5 SRp20 Sfrs3, X16 RRM, RS GCUCCUCUUC SRp20, CT/CGRP; −/− early embryonic lethal E3.5 9G8 Sfrs7 RRM, RS, C2HC Znf (GAC)n Tau, GnRH, 9G8 ASF/SF2 Sfrs1 RRM, RS RGAAGAAC HipK3, CaMKIIδ, HIV RNAs; −/− embryonic lethal, cond. KO cardiomyopathy SC35 Sfrs2 RRM, RS UGCUGUU AChE; −/− embryonic lethal, cond. KO deficient T-cell maturation, cardiomyopathy; LS SRp30c Sfrs9 RRM, RS CUGGAUU Glucocorticoid receptor SRp38 Fusip1, Nssr RRM, RS ACAAAGACAA CREB, type II and type XI collagens SRp40 Sfrs5, HRS RRM, RS AGGAGAAGGGA HipK3, PKCβ-II, Fibronectin SRp55 Sfrs6 RRM, RS GGCAGCACCUG cTnT, CD44 DOI 10.1016/j.cell.2008.03.010 SRp75 Sfrs4 RRM, RS GAAGGA FN1, E1A, CD45; overexpression enhances chondrogenic differentiation Tra2α Tra2a RRM, RS GAAARGARR GnRH; overexpression promotes RA-induced neural differentiation SR and SR-Related Proteins Tra2β Sfrs10 RRM, RS (GAA)n HipK3, SMN, Tau SRm160 Srrm1 RS, PWI AUGAAGAGGA CD44 SWAP Sfrs8 RS, SWAP ND SWAP, CD45, Tau; possible asthma susceptibility gene hnRNP A1 Hnrnpa1 RRM, RGG UAGGGA/U HipK3, SMN2, c-H-ras; rheumatoid arthritis, systemic lupus -

Supplementary Materials
Supplementary materials Supplementary Table S1: MGNC compound library Ingredien Molecule Caco- Mol ID MW AlogP OB (%) BBB DL FASA- HL t Name Name 2 shengdi MOL012254 campesterol 400.8 7.63 37.58 1.34 0.98 0.7 0.21 20.2 shengdi MOL000519 coniferin 314.4 3.16 31.11 0.42 -0.2 0.3 0.27 74.6 beta- shengdi MOL000359 414.8 8.08 36.91 1.32 0.99 0.8 0.23 20.2 sitosterol pachymic shengdi MOL000289 528.9 6.54 33.63 0.1 -0.6 0.8 0 9.27 acid Poricoic acid shengdi MOL000291 484.7 5.64 30.52 -0.08 -0.9 0.8 0 8.67 B Chrysanthem shengdi MOL004492 585 8.24 38.72 0.51 -1 0.6 0.3 17.5 axanthin 20- shengdi MOL011455 Hexadecano 418.6 1.91 32.7 -0.24 -0.4 0.7 0.29 104 ylingenol huanglian MOL001454 berberine 336.4 3.45 36.86 1.24 0.57 0.8 0.19 6.57 huanglian MOL013352 Obacunone 454.6 2.68 43.29 0.01 -0.4 0.8 0.31 -13 huanglian MOL002894 berberrubine 322.4 3.2 35.74 1.07 0.17 0.7 0.24 6.46 huanglian MOL002897 epiberberine 336.4 3.45 43.09 1.17 0.4 0.8 0.19 6.1 huanglian MOL002903 (R)-Canadine 339.4 3.4 55.37 1.04 0.57 0.8 0.2 6.41 huanglian MOL002904 Berlambine 351.4 2.49 36.68 0.97 0.17 0.8 0.28 7.33 Corchorosid huanglian MOL002907 404.6 1.34 105 -0.91 -1.3 0.8 0.29 6.68 e A_qt Magnogrand huanglian MOL000622 266.4 1.18 63.71 0.02 -0.2 0.2 0.3 3.17 iolide huanglian MOL000762 Palmidin A 510.5 4.52 35.36 -0.38 -1.5 0.7 0.39 33.2 huanglian MOL000785 palmatine 352.4 3.65 64.6 1.33 0.37 0.7 0.13 2.25 huanglian MOL000098 quercetin 302.3 1.5 46.43 0.05 -0.8 0.3 0.38 14.4 huanglian MOL001458 coptisine 320.3 3.25 30.67 1.21 0.32 0.9 0.26 9.33 huanglian MOL002668 Worenine -

Mechanistic Involvement of Long Non-Coding Rnas in Oncotherapeutics Resistance in Triple-Negative Breast Cancer
cells Review Mechanistic Involvement of Long Non-Coding RNAs in Oncotherapeutics Resistance in Triple-Negative Breast Cancer Samarth Kansara 1, Vijay Pandey 2,3, Peter E. Lobie 2,3, Gautam Sethi 4,*, Manoj Garg 5 and Amit Kumar Pandey 1,* 1 Amity Institute of Biotechnology, Amity University Haryana, Panchgaon, Manesar, Haryana 122413, India; [email protected] 2 Tsinghua-Berkeley Shenzhen Institute, Tsinghua University, Shenzhen 518005, China; [email protected] (V.P.); [email protected] (P.E.L.) 3 Shenzhen Bay Laboratory, Shenzhen 518055, China 4 Department of Pharmacology, Yong Loo Lin School of Medicine, National University of Singapore, Singapore 117600, Singapore 5 Amity Institute of Molecular Medicine and Stem Cell Research (AIMMSCR), Amity University, Sector-125, Noida 201313, India; [email protected] * Correspondence: [email protected] (G.S.); [email protected] (A.K.P.) Received: 28 May 2020; Accepted: 19 June 2020; Published: 21 June 2020 Abstract: Triple-negative breast cancer (TNBC) is one of the most lethal forms of breast cancer (BC), with a significant disease burden worldwide. Chemoresistance and lack of targeted therapeutics are major hindrances to effective treatments in the clinic and are crucial causes of a worse prognosis and high rate of relapse/recurrence in patients diagnosed with TNBC. In the last decade, long non-coding RNAs (lncRNAs) have been found to perform a pivotal role in most cellular functions. The aberrant functional expression of lncRNAs plays an ever-increasing role in the progression of diverse malignancies, including TNBC. Therefore, lncRNAs have been recently studied as predictors and modifiers of chemoresistance. -

Intronic Mir-26B Controls Neuronal Differentiation by Repressing Its Host Transcript, Ctdsp2
Downloaded from genesdev.cshlp.org on September 30, 2021 - Published by Cold Spring Harbor Laboratory Press RESEARCH COMMUNICATION CTDSPL. As part of the REST complex, these enzymes Intronic miR-26b controls dephosphorylate the C-terminal domain (CTD) of RNA neuronal differentiation polymerase II (PolII) and thereby inhibit the expression of genes that have an RE1 signature sequence (Yeo et al. by repressing its host 2005; Visvanathan et al. 2007). During neural fate com- ctdsp2 mitment and terminal differentiation, the REST/CTDSP transcript, pathway is gradually inactivated to allow the expression Holger Dill,1 Bastian Linder,1 Alexander Fehr,2 of RE1-containing genes (Ballas et al. 2005; Yeo et al. 3 2005; Visvanathan et al. 2007). Neuronal gene activation and Utz Fischer in mice and humans is known to depend on the action of Department of Biochemistry, Theodor Boveri-Institute, miR-124, which prevents expression of CTDSPs (Lim University of Wu¨ rzburg, D-97074 Wu¨ rzburg, Germany et al. 2005; Conaco et al. 2006; Visvanathan et al. 2007). However, as shown in mice, the neuron-specific miR-124 Differentiation of neural stem cells (NSCs) to neurons is itself repressed by the REST/CTDSP pathway, since all of its loci contain an RE1 (Conaco et al. 2006). As mRNAs requires the activation of genes controlled by the re- often are under the control of a collection of microRNAs pressor element 1 (RE1) silencing transcription factor (miRNAs) (Krek et al. 2005), we therefore hypothesized (REST)/neuron-restrictive silencer factor (NRSF) protein that additional miRNAs might be involved in the regu- complex. Important components of REST/NRSF are phos- lation of CTDSP activity. -

Splicing Graphs and RNA-Seq Data
Splicing graphs and RNA-seq data Herv´ePag`es Daniel Bindreither Marc Carlson Martin Morgan Last modified: January 2019; Compiled: May 19, 2021 Contents 1 Introduction 1 2 Splicing graphs 2 2.1 Definitions and example . .2 2.2 Details about nodes and edges . .2 2.3 Uninformative nodes . .5 3 Computing splicing graphs from annotations 6 3.1 Choosing and loading a gene model . .6 3.2 Generating a SplicingGraphs object . .6 3.3 Basic manipulation of a SplicingGraphs object . .8 3.4 Extracting and plotting graphs from a SplicingGraphs object . 12 4 Splicing graph bubbles 15 4.1 Some definitions . 15 4.2 Computing the bubbles of a splicing graph . 17 4.3 AScodes ............................................... 18 4.4 Tabulating all the AS codes for Human chromosome 14 . 19 5 Counting reads 20 5.1 Assigning reads to the edges of a SplicingGraphs object . 22 5.2 How does read assignment work? . 23 5.3 Counting and summarizing the assigned reads . 25 6 Session Information 26 1 Introduction The SplicingGraphs package allows the user to create and manipulate splicing graphs [1] based on annotations for a given organism. Annotations must describe a gene model, that is, they need to contain the following information: The exact exon/intron structure (i.e., genomic coordinates) for the known transcripts. The grouping of transcripts by gene. Annotations need to be provided as a TxDb or GRangesList object, which is how a gene model is typically represented in Bioconductor. 1 The SplicingGraphs package defines the SplicingGraphs container for storing the splicing graphs together with the gene model that they are based on. -

Thyroid Hormone Negatively Regulates Tumorigenesis Through Suppression of BC200
25 12 Endocrine-Related Y-H Lin et al. BC200 is an oncogenic driver 25:12 967–979 Cancer in HCC RESEARCH Thyroid hormone negatively regulates tumorigenesis through suppression of BC200 Yang-Hsiang Lin1,2, Meng-Han Wu1, Ya-Hui Huang2, Chau-Ting Yeh2, Hsiang-Cheng Chi1,3, Chung-Ying Tsai4, Wen-Yu Chuang5, Chia-Jung Yu6,7,8,9, I-Hsiao Chung1, Ching-Ying Chen1 and Kwang-Huei Lin1,2,7,10 1Department of Biochemistry, College of Medicine, Chang Gung University, Taoyuan, Taiwan 2Liver Research Center, Chang Gung Memorial Hospital, Linkou, Taoyuan, Taiwan 3Radiation Biology Research Center, Institute for Radiological Research, Chang Gung University/Chang Gung Memorial Hospital, Linkou, Taoyuan, Taiwan 4Kidney Research Center and Department of Nephrology, Chang Gung Memorial Hospital, Taoyuan, Taiwan 5Department of Pathology, Chang Gung Memorial Hospital and Chang Gung University College of Medicine, Taoyuan, Taiwan 6Molecular Medicine Research Center, Chang Gung University, Taoyuan, Taiwan 7Graduate Institute of Biomedical Sciences, College of Medicine, Chang Gung University, Taoyuan, Taiwan 8Department of Cell and Molecular Biology, College of Medicine, Chang Gung University, Taoyuan, Taiwan 9Department of Thoracic Medicine, Chang Gung Memorial Hospital, Linkou, Taoyuan, Taiwan 10Research Center for Chinese Herbal Medicine, College of Human Ecology, Chang Gung University of Science and Technology, Taoyuan, Taiwan Correspondence should be addressed to K-H Lin: [email protected] Abstract Thyroid hormone (T3) and its receptor (TR) are involved in cancer progression. While Key Words deregulation of long non-coding RNA (lncRNA) expression has been detected in f thyroid hormone many tumor types, the mechanisms underlying specific involvement of lncRNAs in f non-coding RNA tumorigenicity remain unclear. -

Synthesis of Orthogonal Transcription- Translation Networks
Synthesis of orthogonal transcription- translation networks Wenlin An and Jason W. Chin1 Medical Research Council Laboratory of Molecular Biology, Hills Road, Cambridge CB2 0QH, United Kingdom Edited by Susan Gottesman, National Institutes of Health, Bethesda, MD, and approved April 2, 2009 (received for review January 11, 2009) Orthogonal, parallel and independent, systems are one key foun- O-ribosomes for the translation of an O-mRNA arises from the dation for synthetic biology. The synthesis of orthogonal systems altered 16S rRNA in the O-ribosome that recognizes an altered that are uncoupled from evolutionary constraints, and selectively Shine-Dalgarno sequence in the leader sequence of the O-mRNA abstracted from cellular regulation, is an emerging approach to in the initiation phase of translation (12). In previous work O- making biology more amenable to engineering. Here, we combine ribosomes have been evolved that decode genetic information in orthogonal transcription by T7 RNA polymerase and translation by orthogonal mRNAs in new ways. In combination with orthogonal orthogonal ribosomes (O-ribosomes), creating an orthogonal gene aminoacyl-tRNA synthetases and tRNAs that recognize unnatural expression pathway in Escherichia coli. We design and implement amino acids the evolved O-ribosomes have allowed us to begin to compact, orthogonal gene expression networks. In particular we undo the ‘‘frozen accident’’ of the natural genetic code and direct focus on creating transcription–translation feed-forward loops the efficient incorporation of unnatural amino acids into proteins (FFLs). The transcription–translation FFLs reported cannot be cre- encoded on O-mRNAs (13, 14). O-ribosomes have also been used ated by using the cells’ gene expression machinery and introduce to create new translational Boolean logic functions that would not information-processing delays on the order of hours into gene be possible to create by using the essential cellular ribosome (15) expression.