Lysosomal Membrane Glycoproteins Bind Cholesterol and Contribute to Lysosomal Cholesterol Export
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Review the Significance of Interleukin-6
Review The significance of interleukin-6 and C-reactive protein in systemic sclerosis: a systematic literature review C. Muangchan1,2,3, J.E. Pope2 1Research Fellow, Rheumatology; ABSTRACT Introduction 2Schulich School of Medicine & Dentistry, Objectives. Interleukin-6 (IL-6) may Systemic sclerosis (SSc) or scleroder- Western University of Canada (formerly play a role in the pathogenesis of SSc. ma is a systemic autoimmune rheumat- University of Western Ontario), St Joseph C-reactive protein (CRP), an acute ic disease characterised by autoimmun- Health Care, London, ON; 3Division of Rheumatology, Department of phase reactant induced by IL-6, may be ity; fibrosis and dysfunction in vascular Medicine, Faculty of Medicine, Mahidol a prognostic marker in SSc. The goal of regulatory mechanisms highlighted by University, Siriraj Hospital, Bangkok, this systematic review was to address vasculopathy of microcirculation (1). Thailand. the significance and clinical applica- SSc has increased extracellular matrix Chayawee Muangchan, Research Fellow tion of IL-6 and CRP in systemic scle- protein deposition due to increased fi- Janet Elizabeth Pope, MD rosis (SSc). broblast biosynthetic activity (2). SSc is Please address correspondence Methods. A literature search was con- rare and has a female predisposition (3, and reprint requests to: ducted to identify English-language 4). It is classified into diffuse cutane- Dr Janet Pope, original articles within PubMed, Sco- ous SSc (dcSSc) and limited cutaneous St. Joseph’s Health Care, London, SSc (lcSSc) subsets according to extent 268 Grosvenor St., pus, and Medline database from incep- London N6A 4V2, ON, Canada. tion to May 30, 2013 using keywords of cutaneous involvement (5). -

Chronic Mtor Activation Induces a Degradative Smooth Muscle Cell Phenotype
The Journal of Clinical Investigation RESEARCH ARTICLE Chronic mTOR activation induces a degradative smooth muscle cell phenotype Guangxin Li,1,2 Mo Wang,1 Alexander W. Caulk,3 Nicholas A. Cilfone,4 Sharvari Gujja,4 Lingfeng Qin,1 Pei-Yu Chen,5 Zehua Chen,4 Sameh Yousef,1 Yang Jiao,1 Changshun He,1 Bo Jiang,1 Arina Korneva,3 Matthew R. Bersi,3 Guilin Wang,6 Xinran Liu,7,8 Sameet Mehta,9 Arnar Geirsson,1,10 Jeffrey R. Gulcher,4 Thomas W. Chittenden,4 Michael Simons,5,10 Jay D. Humphrey,3,10 and George Tellides1,10,11 1Department of Surgery, Yale School of Medicine, New Haven, Connecticut, USA. 2Department of Breast and Thyroid Surgery, Peking University Shenzhen Hospital, Shenzhen, Guangdong Province, China. 3Department of Biomedical Engineering, Yale School of Engineering and Applied Science, New Haven, Connecticut, USA. 4Computational Statistics and Bioinformatics Group, Advanced Artificial Intelligence Research Laboratory, WuXi NextCODE, Cambridge, Massachusetts, USA. 5Internal Medicine, 6Molecular Biophysics and Biochemistry, and 7Cell Biology, Yale School of Medicine, New Haven, Connecticut, USA. 8Center for Cellular and Molecular Imaging, EM Core Facility, Yale School of Medicine, New Haven, Connecticut, USA. 9Genetics and 10Program in Vascular Biology and Therapeutics, Yale School of Medicine, New Haven, Connecticut, USA. 11Veterans Affairs Connecticut Healthcare System, West Haven, Connecticut, USA. Smooth muscle cell (SMC) proliferation has been thought to limit the progression of thoracic aortic aneurysm and dissection (TAAD) because loss of medial cells associates with advanced disease. We investigated effects of SMC proliferation in the aortic media by conditional disruption of Tsc1, which hyperactivates mTOR complex 1. -

Single-Cell RNA Sequencing Demonstrates the Molecular and Cellular Reprogramming of Metastatic Lung Adenocarcinoma
ARTICLE https://doi.org/10.1038/s41467-020-16164-1 OPEN Single-cell RNA sequencing demonstrates the molecular and cellular reprogramming of metastatic lung adenocarcinoma Nayoung Kim 1,2,3,13, Hong Kwan Kim4,13, Kyungjong Lee 5,13, Yourae Hong 1,6, Jong Ho Cho4, Jung Won Choi7, Jung-Il Lee7, Yeon-Lim Suh8,BoMiKu9, Hye Hyeon Eum 1,2,3, Soyean Choi 1, Yoon-La Choi6,10,11, Je-Gun Joung1, Woong-Yang Park 1,2,6, Hyun Ae Jung12, Jong-Mu Sun12, Se-Hoon Lee12, ✉ ✉ Jin Seok Ahn12, Keunchil Park12, Myung-Ju Ahn 12 & Hae-Ock Lee 1,2,3,6 1234567890():,; Advanced metastatic cancer poses utmost clinical challenges and may present molecular and cellular features distinct from an early-stage cancer. Herein, we present single-cell tran- scriptome profiling of metastatic lung adenocarcinoma, the most prevalent histological lung cancer type diagnosed at stage IV in over 40% of all cases. From 208,506 cells populating the normal tissues or early to metastatic stage cancer in 44 patients, we identify a cancer cell subtype deviating from the normal differentiation trajectory and dominating the metastatic stage. In all stages, the stromal and immune cell dynamics reveal ontological and functional changes that create a pro-tumoral and immunosuppressive microenvironment. Normal resident myeloid cell populations are gradually replaced with monocyte-derived macrophages and dendritic cells, along with T-cell exhaustion. This extensive single-cell analysis enhances our understanding of molecular and cellular dynamics in metastatic lung cancer and reveals potential diagnostic and therapeutic targets in cancer-microenvironment interactions. 1 Samsung Genome Institute, Samsung Medical Center, Seoul 06351, Korea. -

Cell Communication and Signaling Biomed Central
Cell Communication and Signaling BioMed Central Review Open Access Extravasation of leukocytes in comparison to tumor cells Carina Strell and Frank Entschladen* Address: Institute of Immunology, Witten/Herdecke University, Stockumer Str. 10, 58448 Witten, Germany Email: Carina Strell - [email protected]; Frank Entschladen* - [email protected] * Corresponding author Published: 4 December 2008 Received: 18 November 2008 Accepted: 4 December 2008 Cell Communication and Signaling 2008, 6:10 doi:10.1186/1478-811X-6-10 This article is available from: http://www.biosignaling.com/content/6/1/10 © 2008 Strell and Entschladen; licensee BioMed Central Ltd. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Abstract The multi-step process of the emigration of cells from the blood stream through the vascular endothelium into the tissue has been termed extravasation. The extravasation of leukocytes is fairly well characterized down to the molecular level, and has been reviewed in several aspects. Comparatively little is known about the extravasation of tumor cells, which is part of the hematogenic metastasis formation. Although the steps of the process are basically the same in leukocytes and tumor cells, i.e. rolling, adhesion, transmigration (diapedesis), the molecules that are involved are different. A further important difference is that leukocyte interaction with the endothelium changes the endothelial integrity only temporarily, whereas tumor cell interaction leads to an irreversible damage of the endothelial architecture. Moreover, tumor cells utilize leukocytes for their extravasation as linkers to the endothelium. -

The Regulation of Interleukin 7 Receptor Alpha Internalization, Recycling and Degradation by IL-7
Universidade de Lisboa Faculdade de Medicina Unidade de Biologia do Cancro, Instituto de Medicina Molecular The regulation of Interleukin 7 receptor alpha internalization, recycling and degradation by IL-7 - Possible implications in T-cell homeostasis, migration and leukaemogenesis - Catarina Martins de Oliveira Henriques Doutoramento em Ciências Biomédicas For the degree of Doctor of Philosophy 2009 Universidade de Lisboa Faculdade de Medicina Unidade de Biologia do Cancro, Instituto de Medicina Molecular The regulation of Interleukin 7 receptor alpha internalization, recycling and degradation by IL-7 - Possible implications in T-cell homeostasis, migration and leukaemogenesis - Catarina Martins de Oliveira Henriques (Recipient of a scholarship- SFRH7BD/21940/2005 from Fundação para a Ciência e Tecnologia) Tese orientada pelo Doutor João T. Barata, Prof Doutor.Gerard Graham e Prof. Doutora Leonor Parreira Doutoramento em Ciências Biomédicas, especialidade em Ciências Biopatológicas For the degree of Doctor of Philosophy 2009 As opiniões expressas são da exclusiva responsabilidade do seu autor A impressão desta dissertação foi aprovada pela Comissão Coordenadora do Conselho Científico da Faculdade de Medicina de Lisboa em reunião de 13 de Outubro de 2009. Para a Prof. Filomena Mota. Sem o seu apoio e inspiração há muitos anos atrás, eu não seria hoje uma bióloga nem esta tese teria alguma vez existido… Table of Contents Table of contents……………………………………………………….………………….................. i Aknowledgements……………………………………………………………………………………………. -
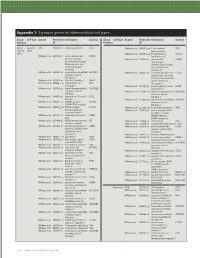
Technical Note, Appendix: an Analysis of Blood Processing Methods to Prepare Samples for Genechip® Expression Profiling (Pdf, 1
Appendix 1: Signature genes for different blood cell types. Blood Cell Type Source Probe Set Description Symbol Blood Cell Type Source Probe Set Description Symbol Fraction ID Fraction ID Mono- Lympho- GSK 203547_at CD4 antigen (p55) CD4 Whitney et al. 209813_x_at T cell receptor TRG nuclear cytes gamma locus cells Whitney et al. 209995_s_at T-cell leukemia/ TCL1A Whitney et al. 203104_at colony stimulating CSF1R lymphoma 1A factor 1 receptor, Whitney et al. 210164_at granzyme B GZMB formerly McDonough (granzyme 2, feline sarcoma viral cytotoxic T-lymphocyte- (v-fms) oncogene associated serine homolog esterase 1) Whitney et al. 203290_at major histocompatibility HLA-DQA1 Whitney et al. 210321_at similar to granzyme B CTLA1 complex, class II, (granzyme 2, cytotoxic DQ alpha 1 T-lymphocyte-associated Whitney et al. 203413_at NEL-like 2 (chicken) NELL2 serine esterase 1) Whitney et al. 203828_s_at natural killer cell NK4 (H. sapiens) transcript 4 Whitney et al. 212827_at immunoglobulin heavy IGHM Whitney et al. 203932_at major histocompatibility HLA-DMB constant mu complex, class II, Whitney et al. 212998_x_at major histocompatibility HLA-DQB1 DM beta complex, class II, Whitney et al. 204655_at chemokine (C-C motif) CCL5 DQ beta 1 ligand 5 Whitney et al. 212999_x_at major histocompatibility HLA-DQB Whitney et al. 204661_at CDW52 antigen CDW52 complex, class II, (CAMPATH-1 antigen) DQ beta 1 Whitney et al. 205049_s_at CD79A antigen CD79A Whitney et al. 213193_x_at T cell receptor beta locus TRB (immunoglobulin- Whitney et al. 213425_at Homo sapiens cDNA associated alpha) FLJ11441 fis, clone Whitney et al. 205291_at interleukin 2 receptor, IL2RB HEMBA1001323, beta mRNA sequence Whitney et al. -

HIF-1A Regulates IL-1B and IL-17 in Sarcoidosis
RESEARCH ARTICLE HIF-1a regulates IL-1b and IL-17 in sarcoidosis Jaya Talreja1, Harvinder Talwar1, Christian Bauerfeld2, Lawrence I Grossman3, Kezhong Zhang3, Paul Tranchida4, Lobelia Samavati1* 1Department of Internal Medicine, Division of Pulmonary, Critical Care and Sleep Medicine, Wayne State University School of Medicine and Detroit Medical Center, Detroit, United States; 2Department of Pediatrics, Division of Critical Care, Wayne State University School of Medicine and Detroit Medical Center, Detroit, United States; 3Center for Molecular Medicine and Genetics, Wayne State University School of Medicine, Detroit, United States; 4Department of Pathology, Wayne State University School of Medicine and Detroit Medical Center, Detroit, United States Abstract Sarcoidosis is a complex systemic granulomatous disease of unknown etiology characterized by the presence of activated macrophages and Th1/Th17 effector cells. Data mining of our RNA-Seq analysis of CD14+monocytes showed enrichment for metabolic and hypoxia inducible factor (HIF) pathways in sarcoidosis. Further investigation revealed that sarcoidosis macrophages and monocytes exhibit higher protein levels for HIF-a isoforms, HIF-1b, and their transcriptional co-activator p300 as well as glucose transporter 1 (Glut1). In situ hybridization of sarcoidosis granulomatous lung tissues showed abundance of HIF-1a in the center of granulomas. The abundance of HIF isoforms was mechanistically linked to elevated IL-1b and IL-17 since targeted down regulation of HIF-1a via short interfering RNA or a HIF-1a inhibitor decreased their production. Pharmacological intervention using chloroquine, a lysosomal inhibitor, decreased lysosomal associated protein 2 (LAMP2) and HIF-1a levels and modified cytokine production. These data suggest that increased activity of HIF-a isoforms regulate Th1/Th17 mediated inflammation in sarcoidosis. -

Sensitization to the Lysosomal Cell Death Pathway by Oncogene- Induced Down-Regulation of Lysosome-Associated Membrane Proteins 1 and 2
Research Article Sensitization to the Lysosomal Cell Death Pathway by Oncogene- Induced Down-regulation of Lysosome-Associated Membrane Proteins 1 and 2 Nicole Fehrenbacher,1 Lone Bastholm,2 Thomas Kirkegaard-Sørensen,1 Bo Rafn,1 Trine Bøttzauw,1 Christina Nielsen,1 Ekkehard Weber,3 Senji Shirasawa,4 Tuula Kallunki,1 and Marja Ja¨a¨ttela¨1 1Apoptosis Department and Centre for Genotoxic Stress Response, Institute for Cancer Biology, Danish Cancer Society; 2Institute of Molecular Pathology, Faculty of Health Sciences, University of Copenhagen, Copenhagen, Denmark; 3Institute of Physiological Chemistry, Medical Faculty, Martin-Luther-University Halle-Wittenberg, Halle, Germany; and 4Department of Cell Biology, School of Medicine, Fukuoka University, Fukuoka, Japan Abstract molecules of the cell to breakdown products available for Expression and activity of lysosomal cysteine cathepsins metabolic reuse (3, 4). Cathepsin proteases are among the best- correlate with the metastatic capacity and aggressiveness of studied lysosomal hydrolases. They are maximally active at the tumors. Here, we show that transformation of murine acidic pH of lysosomes (pH 4–5). However, many of them can be Y527F embryonic fibroblasts with v-H-ras or c-src changes the active at the neutral pH outside lysosomes, albeit with a decreased distribution, density, and ultrastructure of the lysosomes, efficacy and/or altered specificity (5). For example, transformation and tumor environment enhance the expression of lysosomal decreases the levels of lysosome-associated membrane pro- teins (LAMP-1 and LAMP-2) in an extracellular signal- cysteine cathepsins and increase their secretion into the extracel- regulated kinase (ERK)- and cathepsin-dependent manner, lular space (6). Once outside the tumor cells, cathepsins stimulate and sensitizes the cells to lysosomal cell death pathways angiogenesis, tumor growth, and invasion in murine cancer induced by various anticancer drugs (i.e., cisplatin, etoposide, models, thereby enhancing cancer progression (7, 8). -

Abnormal Lysosomal Trafficking and Enhanced Exosomal Export of Cisplatin in Drug-Resistant Human Ovarian Carcinoma Cells
1595 Abnormal lysosomal trafficking and enhanced exosomal export of cisplatin in drug-resistant human ovarian carcinoma cells Roohangiz Safaei, Barrett J. Larson, of human ovarian carcinoma cells selected for stable Timothy C. Cheng, Michael A. Gibson, resistance to CDDP is markedly reduced in size, and that Shinji Otani, Wiltrud Naerdemann, these cells abnormally sort some lysosomal proteins and Stephen B. Howell and the putative CDDP transporters into an exosomal pathway that also exports CDDP. [Mol Cancer Ther 2005; Rebecca and John Moore University of California at San Diego 4(10):1595–604] Cancer Center, University of California at San Diego, La Jolla, California Introduction Repeated exposure of tumor cells to cisplatin (CDDP) Abstract results in rapid development of resistance, which eventu- Previous work has shown that cisplatin (CDDP) becomes ally contributes to therapeutic failure (1). Resistance to concentrated in lysosomes, and that acquired resistance CDDP is often accompanied by resistance to metalloids to CDDP is associated with abnormalities of protein and is associated with alterations in DNA repair (2), thiol trafficking and secretion. The lysosomal compartment in content (3), and drug accumulation (4). Recent studies have CDDP-sensitive 2008 human ovarian carcinoma cells was linked the expression of several transporters, particularly compared with that in CDDP-resistant 2008/C13*5.25 the copper export proteins ATP7A and ATP7B, with CDDP subline using deconvoluting imaging and specific dyes and resistance and suggest that they play a direct role in the antibodies. The lysosomal compartment in CDDP-resistant efflux of CDDP (5). cells was reduced to just 40% of that in the parental In tumor cells, CDDP accumulates in a variety of vesic- CDDP-sensitive cells (P < 0.002). -

Protein Network Analyses of Pulmonary Endothelial Cells In
www.nature.com/scientificreports OPEN Protein network analyses of pulmonary endothelial cells in chronic thromboembolic pulmonary hypertension Sarath Babu Nukala1,8,9*, Olga Tura‑Ceide3,4,5,9, Giancarlo Aldini1, Valérie F. E. D. Smolders2,3, Isabel Blanco3,4, Victor I. Peinado3,4, Manuel Castell6, Joan Albert Barber3,4, Alessandra Altomare1, Giovanna Baron1, Marina Carini1, Marta Cascante2,7,9 & Alfonsina D’Amato1,9* Chronic thromboembolic pulmonary hypertension (CTEPH) is a vascular disease characterized by the presence of organized thromboembolic material in pulmonary arteries leading to increased vascular resistance, heart failure and death. Dysfunction of endothelial cells is involved in CTEPH. The present study describes for the frst time the molecular processes underlying endothelial dysfunction in the development of the CTEPH. The advanced analytical approach and the protein network analyses of patient derived CTEPH endothelial cells allowed the quantitation of 3258 proteins. The 673 diferentially regulated proteins were associated with functional and disease protein network modules. The protein network analyses resulted in the characterization of dysregulated pathways associated with endothelial dysfunction, such as mitochondrial dysfunction, oxidative phosphorylation, sirtuin signaling, infammatory response, oxidative stress and fatty acid metabolism related pathways. In addition, the quantifcation of advanced oxidation protein products, total protein carbonyl content, and intracellular reactive oxygen species resulted increased -

Dipeptidase Aminodipeptidase, Quiescent Cell Proline of a Novel
Vesicular Localization and Characterization of a Novel Post-Proline-Cleaving Aminodipeptidase, Quiescent Cell Proline Dipeptidase This information is current as of September 26, 2021. Murali Chiravuri, Fernando Agarraberes, Suzanne L. Mathieu, Henry Lee and Brigitte T. Huber J Immunol 2000; 165:5695-5702; ; doi: 10.4049/jimmunol.165.10.5695 http://www.jimmunol.org/content/165/10/5695 Downloaded from References This article cites 28 articles, 18 of which you can access for free at: http://www.jimmunol.org/content/165/10/5695.full#ref-list-1 http://www.jimmunol.org/ Why The JI? Submit online. • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication by guest on September 26, 2021 *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2000 by The American Association of Immunologists All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. Vesicular Localization and Characterization of a Novel Post-Proline-Cleaving Aminodipeptidase, Quiescent Cell Proline Dipeptidase1 Murali Chiravuri,* Fernando Agarraberes,† Suzanne L. Mathieu,* Henry Lee,* and Brigitte T. Huber2* A large number of chemokines, cytokines, and signal peptides share a highly conserved X-Pro motif on the N-terminus. -
Reprogramming of Lysosomal Gene Expression by Interleukin-4 and Stat6 Brignull Et Al
Reprogramming of lysosomal gene expression by interleukin-4 and Stat6 Brignull et al. Brignull et al. BMC Genomics 2013, 14:853 http://www.biomedcentral.com/1471-2164/14/853 Brignull et al. BMC Genomics 2013, 14:853 http://www.biomedcentral.com/1471-2164/14/853 RESEARCH ARTICLE Open Access Reprogramming of lysosomal gene expression by interleukin-4 and Stat6 Louise M Brignull1†, Zsolt Czimmerer2†, Hafida Saidi1,3, Bence Daniel2, Izabel Villela4,5, Nathan W Bartlett6, Sebastian L Johnston6, Lisiane B Meira4, Laszlo Nagy2,7 and Axel Nohturfft1* Abstract Background: Lysosomes play important roles in multiple aspects of physiology, but the problem of how the transcription of lysosomal genes is coordinated remains incompletely understood. The goal of this study was to illuminate the physiological contexts in which lysosomal genes are coordinately regulated and to identify transcription factors involved in this control. Results: As transcription factors and their target genes are often co-regulated, we performed meta-analyses of array-based expression data to identify regulators whose mRNA profiles are highly correlated with those of a core set of lysosomal genes. Among the ~50 transcription factors that rank highest by this measure, 65% are involved in differentiation or development, and 22% have been implicated in interferon signaling. The most strongly correlated candidate was Stat6, a factor commonly activated by interleukin-4 (IL-4) or IL-13. Publicly available chromatin immunoprecipitation (ChIP) data from alternatively activated mouse macrophages show that lysosomal genes are overrepresented among Stat6-bound targets. Quantification of RNA from wild-type and Stat6-deficient cells indicates that Stat6 promotes the expression of over 100 lysosomal genes, including hydrolases, subunits of the vacuolar H+ ATPase and trafficking factors.