(KHV) from Potential Vector Species to Common Carp
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Neurotoxin Β-N-Methylamino-L-Alanine (BMAA)
The neurotoxin β-N-methylamino-L-alanine (BMAA) Sources, bioaccumulation and extraction procedures Sandra Ferreira Lage ©Sandra Ferreira Lage, Stockholm University 2016 Cover image: Cyanobacteria, diatoms and dinoflagellates microscopic pictures taken by Sandra Ferreira Lage ISBN 978-91-7649-455-4 Printed in Sweden by Holmbergs, Malmö 2016 Distributor: Department of Ecology, Environment and Plant Sciences, Stockholm University “Sinto mais longe o passado, sinto a saudade mais perto.” Fernando Pessoa, 1914. Abstract β-methylamino-L-alanine (BMAA) is a neurotoxin linked to neurodegeneration, which is manifested in the devastating human diseases amyotrophic lateral sclerosis, Alzheimer’s and Parkinson’s disease. This neurotoxin is known to be produced by almost all tested species within the cyanobacterial phylum including free living as well as the symbiotic strains. The global distribution of the BMAA producers ranges from a terrestrial ecosystem on the Island of Guam in the Pacific Ocean to an aquatic ecosystem in Northern Europe, the Baltic Sea, where annually massive surface blooms occur. BMAA had been shown to accumulate in the Baltic Sea food web, with highest levels in the bottom dwelling fish-species as well as in mollusks. One of the aims of this thesis was to test the bottom-dwelling bioaccumulation hy- pothesis by using a larger number of samples allowing a statistical evaluation. Hence, a large set of fish individuals from the lake Finjasjön, were caught and the BMAA concentrations in different tissues were related to the season of catching, fish gender, total weight and species. The results reveal that fish total weight and fish species were positively correlated with BMAA concentration in the fish brain. -
Labidesthes Sicculus
Version 2, 2015 United States Fish and Wildlife Service Lower Great Lakes Fish and Wildlife Conservation Office 1 Atherinidae Atherinidae Sand Smelt Distinguishing Features: — (Atherina boyeri) — Sand Smelt (Non-native) Old World Silversides Old World Silversides Old World (Atherina boyeri) Two widely separated dorsal fins Eye wider than Silver color snout length 39-49 lateral line scales 2 anal spines, 13-15.5 rays Rainbow Smelt (Non -Native) (Osmerus mordax) No dorsal spines Pale green dorsally Single dorsal with adipose fin Coloring: Silver Elongated, pointed snout No anal spines Size: Length: up to 145mm SL Pink/purple/blue iridescence on sides Distinguishing Features: Dorsal spines (total): 7-10 Brook Silverside (Native) 1 spine, 10-11 rays Dorsal soft rays (total): 8-16 (Labidesthes sicculus) 4 spines Anal spines: 2 Anal soft rays: 13-15.5 Eye diameter wider than snout length Habitat: Pelagic in lakes, slow or still waters Similar Species: Rainbow Smelt (Osmerus mordax), 75-80 lateral line scales Brook Silverside (Labidesthes sicculus) Elongated anal fin Images are not to scale 2 3 Centrarchidae Centrarchidae Redear Sunfish Distinguishing Features: (Lepomis microlophus) Redear Sunfish (Non-native) — — Sunfishes (Lepomis microlophus) Sunfishes Red on opercular flap No iridescent lines on cheek Long, pointed pectoral fins Bluegill (Native) Dark blotch at base (Lepomis macrochirus) of dorsal fin No red on opercular flap Coloring: Brownish-green to gray Blue-purple iridescence on cheek Bright red outer margin on opercular flap -

Commercial Inland Fishing in Member Countries of the European Inland Fisheries Advisory Commission (EIFAC)
Commercial inland fishing in member countries of the European Inland Fisheries Advisory Commission (EIFAC): Operational environments, property rights regimes and socio-economic indicators Country Profiles May 2010 Mitchell, M., Vanberg, J. & Sipponen, M. EIFAC Ad Hoc Working Party on Socio-Economic Aspects of Inland Fisheries The designations employed and the presentation of material in this information product do not imply the expression of any opinion whatsoever on the part of the Food and Agriculture Organization of the United Nations (FAO) concerning the legal or development status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. The mention of specific companies or products of manufacturers, whether or not these have been patented, does not imply that these have been endorsed or recommended by FAO in preference to others of a similar nature that are not mentioned. The views expressed in this information product are those of the author(s) and do not necessarily reflect the views of FAO. All rights reserved. FAO encourages the reproduction and dissemination of material in this information product. Non-commercial uses will be authorized free of charge, upon request. Reproduction for resale or other commercial purposes, including educational purposes, may incur fees. Applications for permission to reproduce or disseminate FAO copyright materials, and all queries concerning rights and licences, should be addressed by e-mail to [email protected] or to the Chief, Publishing Policy and Support Branch, Office of Knowledge Exchange, Research and Extension, FAO, Viale delle Terme di Caracalla, 00153 Rome, Italy. © FAO 2012 All papers have been reproduced as submitted. -

Artificial Reproduction of Blue Bream (Ballerus Ballerus L.) As A
animals Article Artificial Reproduction of Blue Bream (Ballerus ballerus L.) as a Conservative Method under Controlled Conditions Przemysław Piech * and Roman Kujawa Department of Ichthyology and Aquaculture, Faculty of Animal Bioengineering, University of Warmia and Mazury in Olsztyn, PL 10-719 Olsztyn, Poland; reofi[email protected] * Correspondence: [email protected] Simple Summary: Quite severe biological imbalances have been caused by the often ill-conceived and destructive actions of humans. The natural environment, with its flora and fauna, has been subjected to a strong, direct or indirect, anthropogenic impact. In consequence, the total population of wild animals has been considerably reduced, despite efforts to compensate for these errors and expand the scope of animal legal protection to include endangered species. Many animal populations on the verge of extinction have been saved. These actions are ongoing and embrace endangered species as well as those which may be threatened with extinction in the near future as a result of climate change. The changes affect economically valuable species and those of low value, whose populations are still relatively strong and stable. Pre-emptive protective actions and developing methods for the reproduction and rearing of rare species may ensure their survival when the ecological balance is upset. The blue bream is one such species which should be protected while there is still time. Abstract: The blue bream Ballerus ballerus (L.) is one of two species of the Ballerus genus occurring in Citation: Piech, P.; Kujawa, R. Europe. The biotechnology for its reproduction under controlled conditions needs to be developed to Artificial Reproduction of Blue Bream conserve its local populations. -

Applied Freshwater Fish Biology an Introduction to Methods of Research and Management
How to preserve and how to exploit natural populations to be sustained for the future. Plain questions without equally plain answers Applied freshwater fish biology An introduction to methods of research and management Arne N. Linløkken, ass. professor Inland Norway University of Applied Sciences Arne N. 1 CONTENT INTRODUCTION .................................................................................................................................................. 3 Prehistory and evolution ......................................................................................................................................... 3 Short on construction and function ......................................................................................................................... 4 Morphology ........................................................................................................................................................ 4 Anatomy and physiology .................................................................................................................................... 5 European freshwater fish species ............................................................................................................................ 6 Immigration and distribution of freshwater fish in western Scandinavia ........................................................... 7 Western immigrants ........................................................................................................................ 8 -

Notropis Atherinoides) in the Upper Niagara River, New York: Growth, Diversity, and Importance As a Forage Species Jacob L
State University of New York College at Buffalo - Buffalo State College Digital Commons at Buffalo State Great Lakes Center Masters Theses Great Lakes Center 8-2017 Ecology of the Young-of-the-Year Emerald Shiner (Notropis Atherinoides) in the Upper Niagara River, New York: Growth, Diversity, and Importance as a Forage Species Jacob L. Cochran State University of New York College at Buffalo, [email protected] Advisor Dr. Alicia Pérez-Fuentetaja First Reader Dr. Alicia Pérez-Fuentetaja Second Reader Dr. Randal J. Snyder Third Reader Mark D. Clapsadl Department Chair Dr. Alexander Y. Karatayev To learn more about the Great Lakes Center and its educational programs, research, and resources, go to http://greatlakescenter.buffalostate.edu/. Recommended Citation Cochran, Jacob L., "Ecology of the Young-of-the-Year Emerald Shiner (Notropis Atherinoides) in the Upper Niagara River, New York: Growth, Diversity, and Importance as a Forage Species" (2017). Great Lakes Center Masters Theses. 3. http://digitalcommons.buffalostate.edu/greatlakes_theses/3 Follow this and additional works at: http://digitalcommons.buffalostate.edu/greatlakes_theses Part of the Other Ecology and Evolutionary Biology Commons, Population Biology Commons, and the Terrestrial and Aquatic Ecology Commons Abstract of Thesis The emerald shiner (Notropis atherinoides) is a relatively understudied Cyprinid that fills a major keystone role in the Niagara River. Little is known about the emerald shiner’s early life history, such as the ecology of their larval and juvenile stages, which is the focus of this study. In the upper Niagara River, larvae first recruited into sampling gear in early July at a mean water temperature of 23oC, with larvae appearing into August. -

Description of Myxobolus Xiantaoensis N. Sp. from the Fins of Yellow Catfish in China: a Species Previously Attributed to Myxobo
Parasitology Research (2019) 118:1137–1146 https://doi.org/10.1007/s00436-019-06244-7 FISH PARASITOLOGY - ORIGINAL PAPER Description of Myxobolus xiantaoensis n. sp. from the fins of yellow catfish in China: a species previously attributed to Myxobolus physophilus Reuss, 1906 in Chinese records Urfa Bin Tahir1,2 & QingXiang Guo1,2 & DanDan Zhao1,2 & Yang Liu1,2 & Zemao Gu1,2 Received: 8 June 2018 /Accepted: 31 January 2019 /Published online: 5 March 2019 # Springer-Verlag GmbH Germany, part of Springer Nature 2019 Abstract Myxozoans are economically important cnidarian endoparasites. Members of this group have been traditionally characterized by a morphology-based taxonomic system. Because myxozoans possess few morphological characters, these data are routinely accompanied by biological traits (host/organ/tissue specificity) and molecular data when describing or identifying myxozoan species. In the present study, a species of Myxobolus was collected from the fins of yellow catfish Tachysurus fulvidraco Richardson, 1846, which was consistent in spore morphology and host/organ specificity with Chinese records of Myxobolus physophilus Reuss, 1906. However, these earlier records and our own findings are inconsistent with the original description of M. physophilus from Russia. Specifically, there are differences in spore morphology (shape, intercapsular appendix, and polar capsule size), the infection site (air bladder vs. fins), and the host affinity (common rudd vs. yellow catfish). The inconsistencies allow us to conclude that both the present Myxobolus species and Chinese records of M. physophilus are distinct from the original description of M. physophilus and represent a new Myxobolus species, which we named Myxobolus xiantaoensis n. sp. Keywords Myxozoan . -

Scardiniusgenus in Molecular Studies – a Review
Analele Ştiinţifice ale Universităţii „Alexandru Ioan Cuza”, Secţiunea Genetică şi Biologie Moleculară TOM XVI, Fascicula 4, 2015 SCARDINIUS GENUS IN MOLECULAR STUDIES – A REVIEW OVIDIU POPESCUL1, DUMITRU COJOCARU1, MITICĂ CIORPAC1* Received: 20 November 2015 / Revised: 21 November 2015 / Accepted: 23 November2015 / Published: 14 December 2015 Keywords: Scardinius, molecular markers, phylogeny, phylogeography. Abstract. Scardinius is a genus of ray-finned fish in the Cyprinidae family commonly called rudds. The common rudd (Scardinius erithrophthalmus) is a bentho-pelagic freshwater fish that occurs mainly in nutrient-rich, well vegetated lowland rivers, backwaters, oxbows, ponds and lakes and it is widespread in Europe and middle Asia. It has a medium length of 20-30 cm, but it can reach 50 cm. The classification of cyprinids has always been controversial the morphological traits have an unclear homology this led to the idea that the recognized monophyletic groups are surely misinterpreted. This paper aims to assess the current level of molecular data regarding Scardinius genera. Some of the molecular data obtained for Scardinius genus is from DNA barcoding studies on fresh water fishes, but studies regarding this genus and Cyprinidae family used mitochondrial genes like cytochrome b (cyt b) and cytochrome oxidase (CO), but nuclear genes or nuclear microsatellites were also used. We found that molecular data exists for both nuclear and mitochondrial genes, but this genus wasn’t studied separately and as many of the researchers suggest more taxonomic studies are required in order to solve the uncertainties within it. INTRODUCTION Scardinius is a genus of ray-finned fish in the Cyprinidae family commonly called rudds. -

Essential Young-Of-The-Year Fish Macrophyte Habitats Along the Swedish East Coast
Essential Young-of-the-Year Fish Macrophyte Habitats Along the Swedish East Coast – Evaluation of Representativity of Marine Protected Areas and Pressures on Habitats in the Gulf of Bothnia Zanna Zielfeldt Master’s degree project, 60 credits Swedish University of Agricultural Sciences, SLU Faculty of Natural Resources and Agricultural Sciences (NJ) Department of Aquatic resources (SLU Aqua) Landscape ecology, Stockholm University 1 Essential Young-of-the-Year Fish Macrophyte Habitats Along the Swedish East Coast – Evaluation of Representativity of Marine Protected Areas and Pressures on Habitats in the Gulf of Bothnia Essentiella Makrofythabitat för Årsyngel av Fisk längs den Svenska Ostkusten – Utvärdering av Representativiteten hos Marina Skyddade Områden och Påverkansfaktorer på Habitat i Bottniska Viken Zanna Zielfeldt Supervisor: Charlotte Berkström, Swedish University of Agriculture Sciences, Dept. of Aquatic Resources Assistant supervisor: Maria Eggertsen, Stockholm University, Dept. of Ecology, Environment and Plant Sciences Assistant supervisor: Ulf Bergström, Swedish University of Agriculture Sciences, Dept. of Aquatic Resources Assistant supervisor: Edmond Sacre, Swedish University of Agriculture Sciences, Dept. of Aquatic Resources Examiner: Josefin Sundin, Swedish University of Agriculture Sciences, Dept. of Aquatic Resources Credits: 60 credits Level: A2E for a Master´s degree Course title: Independent Project in Biology, A2E Course code: EX0900 Programme/education: Landscape Ecology, Dept. of Physical Geography, Stockholm University Course coordinating dept: Dept. of Aquatic Resources Place of publication: Stockholm/Öregrund Year of publication: 2021 Keywords Baltic Sea, fish habitats, vegetation cover, fish species richness, recreational activities, boating, marine protected areas Swedish University of Agricultural Sciences Faculty of Natural Resources and Agricultural Sciences (NJ) Department of Aquatic Resources (SLU Aqua) Publishing and archiving Approved students’ theses at SLU are published electronically. -

Reproductive Features of Roach, Bream and Common
International Letters of Natural Sciences Submitted: 2016-04-07 ISSN: 2300-9675, Vol. 57, pp 26-40 Revised: 2016-07-27 doi:10.18052/www.scipress.com/ILNS.57.26 Accepted: 2016-07-27 CC BY 4.0. Published by SciPress Ltd, Switzerland, 2016 Online: 2016-08-03 Reproductive Features of Roach, Bream and Common Carp of Zaporozhian (Dnipro) Reservoir in Contemporary Environmental Conditions Marenkov Oleh Oles Honchar Dnipropetrovsk National University, Ukraine Faculty of Biology, Ecology and Medicine, Department of General Biology and Water Bioresources P.M.B. 49050, Dnipropetrovsk, Ukraine Corresponding Email: [email protected] Keywords: roach, bream, common carp, developing gonads, young fish, artificial spawning nests, Zaporozhian Reservoir. Abstract. Over the past 50 years, the complete histological study of carp fish gonads was first carried out at Zaporozhian Reservoir. In conditions of environmental transformation of the reservoir the regularities of oogenesis and gonadogenesis, passing sexual cycles were found and the ecology of spawning carp fish was investigated. The adaptive potential of the carp fish reproductive systems, which is expressed through the asynchrony of reproductive products development as well as the functional peculiarities of the fish spawning process, was brought to light. Both environmental assessment and faunal analysis of biodiversity and distribution of young fish of Zaporozhian Reservoir were carried out. The growth and development of the fish larvae dwelling within the industrial agglomerations of Dnipropetrovsk city are found to be under anthropogenic influence. The efficiency of the use of artificial spawning nests for the carp fish spawning was studied, and practical recommendations for their installation and operation in the waters of the reservoir were elaborated. -

Ministry of Rural Development, Environment and Agriculture of the Federal State of Brandenburg
Fishing for children and youth | MRDEA Seite 1 von 3 Ministry of Rural Development, Environment and Agriculture of the Federal State of Brandenburg Sitemap Site Map Fishing for children and youth (/cms/detail.php/bb1.c.210998.de) (/cms/detail.php/bb1.c.447071.de) My hobby is fishing! Fishing is a great hobby, and not just for grown-ups. Children and youth can also take an interest in the animals and plants living in and around water. Would you like to fish in the many rivers and lakes in Brandenburg? © MLUL Are children allowed to fish? In Brandenburg children and youth are allowed to fish for non-predatory fish without a fishing license since 1 August 2006. If you are between 8 and 18 years old, you need a valid fisheries fee stamp (price: 2.50 Euro) that should be stuck onto a green identity card. Both are available in the lower fisheries office (county administration), in the business offices of the state fishing club, and in further authorized places such as fishing stores, fisheries or campsite administrations. The identity card for the fisheries fee is an important document containing the name, address and age of the fisher. The card has to be taken with you wherever you fish. In addition a fishing pass is needed for permission to fish on the particular lake or river. This fishing pass is available at fisheries and fishing stores. Anyone with a membership card from a fishing club may fish in any of the rivers and lakes maintained by that fishing club. -
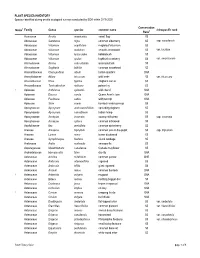
Appendix 11-D Plant and Wildlife Species Lists
PLANT SPECIES INVENTORY Species identified during on-site ecological surveys conducted by EDR within 2019-2020 Conservation Notes1 Family Genus species common name infraspecific rank Rank2 Acoraceae Acorus americanus sweet flag S5 Adoxaceae Sambucus nigra common elderberry S5 ssp. canadensis Adoxaceae Viburnum acerifolium mapleleaf viburnum S5 Adoxaceae Viburnum dentatum smooth arrowwood S5 var. lucidum Adoxaceae Viburnum lantanoides hobblebush S5 Adoxaceae Viburnum opulus highbush cranberry S4 var. americanum Alismataceae Alisma subcordatum water-plantain S5 Alismataceae Sagittaria latifolia common arrowhead S5 Amaranthaceae Chenopodium album lambs-quarters SNA Amaryllidaceae Allium tricoccum wild leeks S5 var. tricoccum Anacardiaceae Rhus typhina staghorn sumac S5 Anacardiaceae Toxicodendron radicans poison ivy S5 1 Apiaceae Anthriscus sylvestris wild chervil SNA Apiaceae Daucus carota Queen Anne's lace SNA Apiaceae Pastinaca sativa wild parsnip SNA Apiaceae Sium suave hemlock water-parnsip S5 Apocynaceae Apocynum androsaemifolium spreading dogbane S5 Apocynaceae Apocynum cannabinum Indian hemp S5 Apocynaceae Asclepias incarnata swamp milkweed S5 ssp. incarnata Apocynaceae Asclepias syriaca common milkweed S5 Aquifoliaceae Ilex verticillata common winterberry S5 Araceae Arisaema triphyllum common jack-in-the-pulpit S5 ssp. triphyllum Araceae Lemna minor lesser duckweed S5 Araceae Symplocarpus foetidus skunk cabbage S5 Araliaceae Aralia nudicaulis sarsaparilla S5 Asparagaceae Maianthemum canadense Canada mayflower S5 Asphodelaceae Hemerocallis