Biomineralization Forming Process and Bio-Inspired Nanomaterials for Biomedical Application: a Review
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Four Hundred Million Years of Silica Biomineralization in Land Plants
Four hundred million years of silica biomineralization in land plants Elizabeth Trembath-Reicherta,1, Jonathan Paul Wilsonb, Shawn E. McGlynna,c, and Woodward W. Fischera aDivision of Geological and Planetary Sciences, California Institute of Technology, Pasadena, CA 91125; bDepartment of Biology, Haverford College, Haverford, PA 19041; and cGraduate School of Science and Engineering, Tokyo Metropolitan University, Hachioji-shi, Tokyo 192-0397, Japan Edited by Thure E. Cerling, University of Utah, Salt Lake City, UT, and approved February 20, 2015 (received for review January 7, 2015) Biomineralization plays a fundamental role in the global silicon Silica is widely used within plants for structural support and cycle. Grasses are known to mobilize significant quantities of Si in pathogen defense (19–21), but it remains a poorly understood the form of silica biominerals and dominate the terrestrial realm aspect of plant biology. Recent work on the angiosperm Oryza today, but they have relatively recent origins and only rose to sativa demonstrated that silica accumulation is facilitated by taxonomic and ecological prominence within the Cenozoic Era. transmembrane proteins expressed in root cells (21–24). Phy- This raises questions regarding when and how the biological silica logenetic analysis revealed that these silicon transport proteins cycle evolved. To address these questions, we examined silica were derived from a diverse family of modified aquaporins that abundances of extant members of early-diverging land plant include arsenite and glycerol transporters (19, 21, 25, 26). A clades, which show that silica biomineralization is widespread different member of this aquaporin family was recently identi- across terrestrial plant linages. Particularly high silica abundances fied that enables silica uptake in the horsetail Equisetum,an are observed in lycophytes and early-diverging ferns. -

SHELLS in ACID Adapted from NAMEPA’S an Educator’S Guide to the Marine Environment: Shells in Acid
SHELLS IN ACID Adapted from NAMEPA’s An Educator’s Guide to the Marine Environment: Shells in Acid PURPOSE Students will test the strength of normal seashells versus shells that have been soaked in vinegar to simulate the weakening effect of ocean acidification. Students identify the correlation between decreasing oceanic pH (ocean acidification) and the weakening of shells and discuss the effect this could have on the health of shellfish in the world’s oceans. MATERIALS (PER GROUP OF 4) • *white vinegar • *small, thin seashells • *non-reactive containers (glass beakers, Pyrex, measuring glass) • *water • heavy books (several) • paper towels For #6: • shells (1 per student) • snack size plastic bags (1 per student) • Small amount of vinegar • magnifying glass *Before beginning this activity, shells should be pre-soaked overnight in a 1:1 solution of vinegar and fresh water. PROCEDURE 1. Engage/Elicit Ask the students to give examples of different species of shellfish. Answers may include clams, oysters, mussels, scallops, etc. Ask students why and where they have seen these creatures. Students’ knowledge may come from eating seafood, or perhaps from having seen them in an aquarium, a marina or in coastal areas. Ask the students why these animals are important to the marine environment and to human beings. 2. Explore Lay out an assemblage of the non-soaked shells. Have the students observe the shells. Allow the students to handle the shells and ask them why the development of shells is advantageous to such animals. Explain that shellfish are invertebrates, meaning that instead of having an internal skeleton like humans, invertebrates produce a hard, protective covering. -

Phytoplankton As Key Mediators of the Biological Carbon Pump: Their Responses to a Changing Climate
sustainability Review Phytoplankton as Key Mediators of the Biological Carbon Pump: Their Responses to a Changing Climate Samarpita Basu * ID and Katherine R. M. Mackey Earth System Science, University of California Irvine, Irvine, CA 92697, USA; [email protected] * Correspondence: [email protected] Received: 7 January 2018; Accepted: 12 March 2018; Published: 19 March 2018 Abstract: The world’s oceans are a major sink for atmospheric carbon dioxide (CO2). The biological carbon pump plays a vital role in the net transfer of CO2 from the atmosphere to the oceans and then to the sediments, subsequently maintaining atmospheric CO2 at significantly lower levels than would be the case if it did not exist. The efficiency of the biological pump is a function of phytoplankton physiology and community structure, which are in turn governed by the physical and chemical conditions of the ocean. However, only a few studies have focused on the importance of phytoplankton community structure to the biological pump. Because global change is expected to influence carbon and nutrient availability, temperature and light (via stratification), an improved understanding of how phytoplankton community size structure will respond in the future is required to gain insight into the biological pump and the ability of the ocean to act as a long-term sink for atmospheric CO2. This review article aims to explore the potential impacts of predicted changes in global temperature and the carbonate system on phytoplankton cell size, species and elemental composition, so as to shed light on the ability of the biological pump to sequester carbon in the future ocean. -

Murphey Et Al. 2019 Best Practices in Mitigation Paleontology
PROCEEDINGS of the San Diego Society of Natural History Founded 1874 Number 47 1 May 2019 BEST PRACTICES IN MITIGATION PALEONTOLOGY By Paul C. Murphey Paleo Solutions, 2785 Speer Boulevard, Suite 1, Denver, CO 80211, U.S.A.; [email protected]; Department of Paleontology, San Diego Natural History Museum, 1788 El Prado, San Diego, CA 92101, U.S.A.; [email protected] Department of Earth Sciences, Denver Museum of Nature and Science, 2001 Colorado Boulevard, Denver, CO 80201, U.S.A. Georgia E. Knauss SWCA Environmental Consultants, 1892 S. Sheridan Avenue, Sheridan, WY 82801 U.S.A.; [email protected] Lanny H. Fisk PaleoResource Consultants, 550 High Street, Suite 108, Auburn, CA 95603, U.S.A. (deceased) Thomas A. Deméré Department of Paleontology, San Diego Natural History Museum, 1788 El Prado, San Diego, CA 92101, U.S.A.; [email protected] Robert E. Reynolds Department of Paleontology, San Diego Natural History Museum, 1788 El Prado, San Diego, CA 92101, U.S.A.; [email protected] For correspondence, write to: Paul C. Murphey, Paleo Solutions, 4614 Lonespur Ct. Oceanside, CA 92056 Email: [email protected] [email protected] bpmp-19-01-fm Page 2 PDF Created: 2019-4-12: 9:20:AM 2 Paul C. Murphey, Georgia E. Knauss, Lanny H. Fisk, Thomas A. Deméré, and Robert E. Reynolds TABLE OF CONTENTS Abstract . 4 Introduction . 4 History and Scientific Contributions . 5 History of Mitigation Paleontology in the United States . 5 Methods Best Practice Categories . 7 1. Qualifications. 7 Confusion between Resource Disciplines . 7 Professional Geologists as Mitigation Paleontologists. 8 Mitigation Paleontologist Categories . -
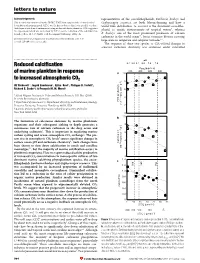
Reduced Calcification of Marine Plankton in Response to Increased
letters to nature Acknowledgements representatives of the coccolithophorids, Emiliania huxleyi and This research was sponsored by the EPSRC. T.W.F. ®rst suggested the electrochemical Gephyrocapsa oceanica, are both bloom-forming and have a deoxidation of titanium metal. G.Z.C. was the ®rst to observe that it was possible to reduce world-wide distribution. G. oceanica is the dominant coccolitho- thick layers of oxide on titanium metal using molten salt electrochemistry. D.J.F. suggested phorid in neritic environments of tropical waters9, whereas the experiment, which was carried out by G.Z.C., on the reduction of the solid titanium dioxide pellets. M. S. P. Shaffer took the original SEM image of Fig. 4a. E. huxleyi, one of the most prominent producers of calcium carbonate in the world ocean10, forms extensive blooms covering Correspondence and requests for materials should be addressed to D. J. F. large areas in temperate and subpolar latitudes9,11. (e-mail: [email protected]). The response of these two species to CO2-related changes in seawater carbonate chemistry was examined under controlled ................................................................. pH Reduced calci®cation 8.4 8.2 8.1 8.0 7.9 7.8 PCO2 (p.p.m.v.) of marine plankton in response 200 400 600 800 a 10 to increased atmospheric CO2 ) 8 –1 Ulf Riebesell *, Ingrid Zondervan*, BjoÈrn Rost*, Philippe D. Tortell², d –1 Richard E. Zeebe*³ & FrancËois M. M. Morel² 6 * Alfred Wegener Institute for Polar and Marine Research, P.O. Box 120161, 4 D-27515 Bremerhaven, Germany mol C cell –13 ² Department of Geosciences & Department of Ecology and Evolutionary Biology, POC production Princeton University, Princeton, New Jersey 08544, USA (10 2 ³ Lamont-Doherty Earth Observatory, Columbia University, Palisades, New York 10964, USA 0 ............................................................................................................................................. -

Silicate Weathering in Anoxic Marine Sediment As a Requirement for Authigenic Carbonate Burial T
Earth-Science Reviews 200 (2020) 102960 Contents lists available at ScienceDirect Earth-Science Reviews journal homepage: www.elsevier.com/locate/earscirev Silicate weathering in anoxic marine sediment as a requirement for authigenic carbonate burial T Marta E. Torresa,*, Wei-Li Hongb, Evan A. Solomonc, Kitty Millikend, Ji-Hoon Kime, James C. Samplef, Barbara M.A. Teichertg, Klaus Wallmannh a College of Earth, Ocean and Atmospheric Science, Oregon State University, Corvallis OR 97331, USA b Geological Survey of Norway, Trondheim, Norway c School of Oceanography, University of Washington, Seattle, WA 98195, USA d Bureau of Economic Geology, University of Texas at Austin, Austin, TX 78713, USA e Petroleum and Marine Research Division, Korea Institute of Geoscience and Mineral Resources, Daejeon, South Korea f School of Earth and Sustainability, Northern Arizona University, Flagstaff, AZ 86011, USA g Geologisch-Paläontologisches Institut, Wilhelms-Universität Münster, Corrensstr. 24, 48149 Münster, Germany h IFM-GEOMAR Leibniz Institute of Marine Sciences, Wischhofstrasse 1-3, 24148 Kiel, Germany ARTICLE INFO ABSTRACT Keywords: We emphasize the importance of marine silicate weathering (MSiW) reactions in anoxic sediment as funda- Silicate weathering mental in generating alkalinity and cations needed for carbonate precipitation and preservation along con- Authigenic carbonate tinental margins. We use a model that couples thermodynamics with aqueous geochemistry to show that the CO2 Organogenic dolomite released during methanogenesis results in a drop in pH to 6.0; unless these protons are buffered by MSiW, Alkalinity carbonate minerals will dissolve. We present data from two regions: the India passive margin and the active Carbon cycling subduction zone off Japan, where ash and/or rivers supply the reactive silicate phase, as reflected in strontium isotope data. -

The Silicate Structure Analysis of Hydrated Portland Cement Paste
The Silicate Structure Analysis of Hydrated Portland Cement Paste CHARLES W. LENTZ, Research Department, Dow Corning Corporation, Midland, Michigan A new technique for recovering silicate structures as trimeth- ylsilyl derivatives has been used to study the hydration of port- land cement. By this method only the changes iii the silicate portion of the structure can be determined as a function of hydration time. Cement pastes ranging in age from one day to 14. 7 years were analyzedfor the study. The hydration reaction is shown to be similar to a condensation type polymerization. The orthosilicate content of cement paste (which probably rep- resents the original calcium silicates in the portland cement) gradually decreases as the paste ages. Concurrentiy a disilicate structure is formed which reaches a maximum quantity in about four weeks and then it too diminishes as the paste ages. Minor quantities of a trisiicate and a cyclic tetrasiicate are shown to be present in hydrated cement paste. An unidentified polysilicate structure is produced by the hydration reaction which not only increases in quantity throughout the age period studied, but also increases in molecular weight as the paste ages. THE SILICON atoms in silicate minerals are always in fourfold coordination with oxygen. These silicon-oxygen tetrahedra can be completely separated from each other, as in the orthosilicates, they can be paired, as in the pyrosilicates, or they can be in other combinations with each other to give a variety of silicate structures. If the min- eral is composed only of Si and 0, then there is a three-dimensional network of silicate tetrahedra. -

Biomineralization and Global Biogeochemical Cycles Philippe Van Cappellen Faculty of Geosciences, Utrecht University P.O
1122 Biomineralization and Global Biogeochemical Cycles Philippe Van Cappellen Faculty of Geosciences, Utrecht University P.O. Box 80021 3508 TA Utrecht, The Netherlands INTRODUCTION Biological activity is a dominant force shaping the chemical structure and evolution of the earth surface environment. The presence of an oxygenated atmosphere- hydrosphere surrounding an otherwise highly reducing solid earth is the most striking consequence of the rise of life on earth. Biological evolution and the functioning of ecosystems, in turn, are to a large degree conditioned by geophysical and geological processes. Understanding the interactions between organisms and their abiotic environment, and the resulting coupled evolution of the biosphere and geosphere is a central theme of research in biogeology. Biogeochemists contribute to this understanding by studying the transformations and transport of chemical substrates and products of biological activity in the environment. Biogeochemical cycles provide a general framework in which geochemists organize their knowledge and interpret their data. The cycle of a given element or substance maps out the rates of transformation in, and transport fluxes between, adjoining environmental reservoirs. The temporal and spatial scales of interest dictate the selection of reservoirs and processes included in the cycle. Typically, the need for a detailed representation of biological process rates and ecosystem structure decreases as the spatial and temporal time scales considered increase. Much progress has been made in the development of global-scale models of biogeochemical cycles. Although these models are based on fairly simple representations of the biosphere and hydrosphere, they account for the large-scale changes in the composition, redox state and biological productivity of the earth surface environment that have occurred over geological time. -

University of Nevada Reno SILICATE and CARBONATE SEDIMENT
University of Nevada Reno /SILICATE AND CARBONATE SEDIMENT-WATER RELATIONSHIPS IN WALKER LAKE, NEVADA A Thesis Submitted in Partial Fulfillment of the Requirements for the Degree of Master of Science in Geochemistry by Ronald J. Spencer MINES 2 - UBKAKY %?: The thesis of Ronald James Spencer is approved: University of Nevada Reno May 1977 ACKNOWLEDGMENTS The author gratefully acknowledges the contributions of Dr. L. V. Benson, who directed the thesis, and Drs. L. C. Hsu and R. D. Burkhart, who served on the thesis ccmnittee. A special note of thanks is given to Pat Harris of the Desert Research Institute, who supervised the wet chemical analyses on the lake water and pore fluids. I also would like to thank John Sims and Mike Rymer of the U. S. Geological Survey, Menlo Park, for their effort in obtaining the piston core; and Blair Jones of the U. S. Geological Survey, Reston, for the use of equipment and many very helpful suggestions. Much of the work herein was done as a part of the study of the "Dynamic Ecological Relationships in Walker Lake, Nevada", and was supported by the Office of Water Research and Technology through grant number C-6158 to the Desert Research Institute. I thank the other members involved in the study; Drs. Dave Koch and Roger Jacobson, and Joe Mahoney, Jim Cooper, and Jim Hainiine; for advice in their fields of expertise and help in sample collection. Special thanks are extended to my wife, Laurie, without whose help and support this thesis could not have been completed. A final note of thanks to Tina Nesler, who advised Laurie in the typing of the manuscript. -

Facies, Phosphate, and Fossil Preservation Potential Across a Lower Cambrian Carbonate Shelf, Arrowie Basin, South Australia
Palaeogeography, Palaeoclimatology, Palaeoecology 533 (2019) 109200 Contents lists available at ScienceDirect Palaeogeography, Palaeoclimatology, Palaeoecology journal homepage: www.elsevier.com/locate/palaeo Facies, phosphate, and fossil preservation potential across a Lower Cambrian T carbonate shelf, Arrowie Basin, South Australia ⁎ Sarah M. Jacqueta,b, , Marissa J. Bettsc,d, John Warren Huntleya, Glenn A. Brockb,d a Department of Geological Sciences, University of Missouri, Columbia, MO 65211, USA b Department of Biological Sciences, Macquarie University, Sydney, New South Wales 2109, Australia c Palaeoscience Research Centre, School of Environmental and Rural Science, University of New England, Armidale, New South Wales 2351, Australia d Early Life Institute and Department of Geology, State Key Laboratory for Continental Dynamics, Northwest University, Xi'an 710069, China ARTICLE INFO ABSTRACT Keywords: The efects of sedimentological, depositional and taphonomic processes on preservation potential of Cambrian Microfacies small shelly fossils (SSF) have important implications for their utility in biostratigraphy and high-resolution Calcareous correlation. To investigate the efects of these processes on fossil occurrence, detailed microfacies analysis, Organophosphatic biostratigraphic data, and multivariate analyses are integrated from an exemplar stratigraphic section Taphonomy intersecting a suite of lower Cambrian carbonate palaeoenvironments in the northern Flinders Ranges, South Biominerals Australia. The succession deepens upsection, across a low-gradient shallow-marine shelf. Six depositional Facies Hardgrounds Sequences are identifed ranging from protected (FS1) and open (FS2) shelf/lagoonal systems, high-energy inner ramp shoal complex (FS3), mid-shelf (FS4), mid- to outer-shelf (FS5) and outer-shelf (FS6) environments. Non-metric multi-dimensional scaling ordination and two-way cluster analysis reveal an underlying bathymetric gradient as the main control on the distribution of SSFs. -

A Perspective on Underlying Crystal Growth Mechanisms in Biomineralization: Solution Mediated Growth Versus Nanosphere Particle Accretion
CrystEngComm A perspective on underlying crystal growth mechanisms in biomineralization: solution mediated growth versus nanosphere particle accretion Journal: CrystEngComm Manuscript ID: CE-HIG-07-2014-001474.R1 Article Type: Highlight Date Submitted by the Author: 01-Dec-2014 Complete List of Authors: Gal, Assaf; Weizmann Institute of Science, Structural Biology Weiner, Steve; Weizmann Institute of Science, Structural Biology Addadi, Lia; Weizmann Institute of Science, Structural Biology Page 1 of 23 CrystEngComm A perspective on underlying crystal growth mechanisms in biomineralization: solution mediated growth versus nanosphere particle accretion Assaf Gal, Steve Weiner, and Lia Addadi Department of Structural Biology, Weizmann Institute of Science, Rehovot, Israel 76100 Abstract Many organisms form crystals from transient amorphous precursor phases. In the cases where the precursor phases were imaged, they consist of nanosphere particles. Interestingly, some mature biogenic crystals also have nanosphere particle morphology, but some are characterized by crystallographic faces that are smooth at the nanometer level. There are also biogenic crystals that have both crystallographic faces and nanosphere particle morphology. This highlight presents a working hypothesis, stating that some biomineralization processes involve growth by nanosphere particle accretion, where amorphous nanoparticles are incorporated as such into growing crystals and preserve their morphology upon crystallization. This process produces biogenic crystals with a nanosphere particle morphology. Other biomineralization processes proceed by ion-by-ion growth, and some cases of biological crystal growth involve both processes. We also identify several biomineralization processes which do not seem to fit this working hypothesis. It is our hope that this highlight will inspire studies that will shed more light on the underlying crystallization mechanisms in biology. -

An Overview of Biomineralization Processes and the Problem of The
11 An Overview of Biomineralization Processes and the Problem of the Vital Effect Steve Weiner Department of Structural Biology Weizmann Institute of Science 76100 Rehovot Israel Patricia M. Dove Department of GeoSciences Virginia Tech Blacksburg, Virginia 24061 U.S.A. “Biomineralization links soft organic tissues, which are compositionally akin to the atmosphere and oceans, with the hard materials of the solid Earth. It provides organisms with skeletons and shells while they are alive, and when they die these are deposited as sediment in environments from river plains to the deep ocean floor. It is also these hard, resistant products of life which are mainly responsible for the Earth’s fossil record. Consequently, biomineralization involves biologists, chemists, and geologists in interdisciplinary studies at one of the interfaces between Earth and life.” (Leadbeater and Riding 1986) INTRODUCTION Biomineralization refers to the processes by which organisms form minerals. The control exerted by many organisms over mineral formation distinguishes these processes from abiotic mineralization. The latter was the primary focus of earth scientists over the last century, but the emergence of biogeochemistry and the urgency of understanding the past and future evolution of the Earth are moving biological mineralization to the forefront of various fields of science, including the earth sciences. The growth in biogeochemistry has led to a number of new exciting research areas where the distinctions between the biological, chemical, and earth sciences disciplines melt away. Of the wonderful topics that are receiving renewed attention, the study of biomineral formation is perhaps the most fascinating. Truly at the interface of earth and life, biomineralization is a discipline that is certain to see major advancements as a new generation of scientists brings cross-disciplinary training and new experimental and computational methods to the most daunting problems.