Beryllium in Drinking-Water
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Radiochemistry of Beryllium
National Academy of Sciences National Research Council I NUCLEAR SCIENCE SERIES The Radiochemistry ·of Beryllium COMMITTEE ON NUCLEAR SCIENCE L. F. CURTISS, Chairman ROBLEY D. EVANS, Vice Chairman National Bureau of Standards MassaChusetts Institute of Technol0gy J. A. DeJUREN, Secretary ./Westinghouse Electric Corporation H.J. CURTIS G. G. MANOV Brookhaven National' LaboratOry Tracerlab, Inc. SAMUEL EPSTEIN W. WAYNE MEINKE CalUornia Institute of Technology University of Michigan HERBERT GOLDSTEIN A.H. SNELL Nuclear Development Corporation of , oak Ridge National Laboratory America E. A. UEHLING H.J. GOMBERG University of Washington University of Michigan D. M. VAN PATTER E.D.KLEMA Bartol Research Foundation Northwestern University ROBERT L. PLATZMAN Argonne National Laboratory LIAISON MEMBERS PAUL C .. AEBERSOLD W.D.URRY Atomic Energy Commission U. S. Air Force J. HOW ARD McMILLEN WILLIAM E. WRIGHT National Science Foundation Office of Naval Research SUBCOMMITTEE ON RADIOCHEMISTRY W. WAYNE MEINKE, Chairman HAROLD KIRBY University of Michigan Mound Laboratory GREGORY R. CHOPPIN GEORGE LEDDICOTTE Florida State University. Oak Ridge National Laboratory GEORGE A. COW AN JULIAN NIELSEN Los Alamos Scientific Laboratory Hanford Laboratories ARTHUR W. FAIRHALL ELLIS P. STEINBERG University of Washington Argonne National Laboratory JEROME HUDIS PETER C. STEVENSON Brookhaven National Laboratory University of California (Livermore) EARL HYDE LEO YAFFE University of CalUornia (Berkeley) McGill University CONSULTANTS NATHAN BALLOU WILLIAM MARLOW Naval Radiological Defense Laboratory N atlonal Bureau of Standards JAMESDeVOE University of Michigan CHF.MISTRY-RADIATION AND RADK>CHEMIST The Radiochemistry of Beryllium By A. W. FAIRHALL. Department of Chemistry University of Washington Seattle, Washington May 1960 ' Subcommittee on Radiochemistry National Academy of Sciences - National Research Council Printed in USA. -

Exposure Data
BERYLLIUM AND BERYLLIUM eOMPOUNDS Beryllium and beryllium compounds were considered by previous Working Groups, In 1971,1979 and 1987 (lARe, 1972, 1980, 1987a). New data have since become available, and these are included in the present monograph and have been taken into consideration In the evaluation. The agents considered herein Include (a) metallic beryllium, (b) beryllium- aluminium and -copper alloys and (c) some beryllum compounds. 1. Exposure Data 1.1 Chemical and physical data and analysis 1.1.1 Synonyms, trade names and molecular formulae Synonyms, trade names and molecular formulae for beryllium, beryllum-aluminium and -copper alloys and certain beryllium compounds are presented in Thble 1. The list is not exhaustive, nor does it comprise necessarily the most commercially important beryllum- containing substances; rather, it indicates the range of beryllum compounds available. 1. 1.2 Chemical and physical properties of the pure substances Selected chemical and physical properties of beryllium, beryllum-aluminium and -copper alloys and the beryllium compounds covered in this monograph are presented in Thble 2. The French chemist Vauquelin discovered beryllium in 1798 as the oxide, while analysing emerald to prove an analogous composition (Newland, 1984). The metallc element was first isolated in independent experiments by Wöhler (1828) and Bussy (1828), who called it 'glucinium' owing to the sweet taste of its salts; that name is stil used in the French chemical literature. Wöhler's name 'beryllum' was offcially recognized by IUPAe in 1957 (WHO, 1990). The atomic weight and corn mon valence of beryllum were originally the subject of much controversy but were correctly predicted by Mendeleev to be 9 and + 2, respectively (Everest, 1973). -

The Periodic Table of Elements
The Periodic Table of Elements 1 2 6 Atomic Number = Number of Protons = Number of Electrons HYDROGENH HELIUMHe 1 Chemical Symbol NON-METALS 4 3 4 C 5 6 7 8 9 10 Li Be CARBON Chemical Name B C N O F Ne LITHIUM BERYLLIUM = Number of Protons + Number of Neutrons* BORON CARBON NITROGEN OXYGEN FLUORINE NEON 7 9 12 Atomic Weight 11 12 14 16 19 20 11 12 13 14 15 16 17 18 SODIUMNa MAGNESIUMMg ALUMINUMAl SILICONSi PHOSPHORUSP SULFURS CHLORINECl ARGONAr 23 24 METALS 27 28 31 32 35 40 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 POTASSIUMK CALCIUMCa SCANDIUMSc TITANIUMTi VANADIUMV CHROMIUMCr MANGANESEMn FeIRON COBALTCo NICKELNi CuCOPPER ZnZINC GALLIUMGa GERMANIUMGe ARSENICAs SELENIUMSe BROMINEBr KRYPTONKr 39 40 45 48 51 52 55 56 59 59 64 65 70 73 75 79 80 84 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 RUBIDIUMRb STRONTIUMSr YTTRIUMY ZIRCONIUMZr NIOBIUMNb MOLYBDENUMMo TECHNETIUMTc RUTHENIUMRu RHODIUMRh PALLADIUMPd AgSILVER CADMIUMCd INDIUMIn SnTIN ANTIMONYSb TELLURIUMTe IODINEI XeXENON 85 88 89 91 93 96 98 101 103 106 108 112 115 119 122 128 127 131 55 56 72 73 74 75 76 77 78 79 80 81 82 83 84 85 86 CESIUMCs BARIUMBa HAFNIUMHf TANTALUMTa TUNGSTENW RHENIUMRe OSMIUMOs IRIDIUMIr PLATINUMPt AuGOLD MERCURYHg THALLIUMTl PbLEAD BISMUTHBi POLONIUMPo ASTATINEAt RnRADON 133 137 178 181 184 186 190 192 195 197 201 204 207 209 209 210 222 87 88 104 105 106 107 108 109 110 111 112 113 114 115 116 117 118 FRANCIUMFr RADIUMRa RUTHERFORDIUMRf DUBNIUMDb SEABORGIUMSg BOHRIUMBh HASSIUMHs MEITNERIUMMt DARMSTADTIUMDs ROENTGENIUMRg COPERNICIUMCn NIHONIUMNh -

Llfltfits Paul Stanek, MST-6
COAJF- 9 50cDo/-~^0 LA-UR- 9 5-1666 Title: AGE HARDENING IN RAPIDLY SOLIDIFIED AND HOT ISOSTATICALLY PRESSED BERYLLIUM-ALUM1UM SILVER ALLOYS HVAUli Author(s): David H. Carter, MST-6 Andrew McGeorge, MST-6 Loren A. Jacobson, MST-6 llfltfits Paul Stanek, MST-6 IflllS,Ic 8-o° ,S8 si Proceedings of TMS Annual Meeting, Las Vegas, jJffilJSif—- Nevada, February 1995 as-IVUiS' i in Los Alamos NATIONAL LABORATORY Los Alamos National Laboratory, an affirmative action/equal opportunity employer, is operated by the University of California for the U.S. Department of Energy under contract W-7405-ENG-36. By acceptance of this article, the publisher recognizes that the U.S. Government retains a nonexclusive, royalty-free license to publish or reproduce the published form of this contribution, or to allow others to do so, for U.S. Government purposes. The Los Alamos National Laboratory requests that the publisher identify this article as work performed under the auspices of the U.S. Department of Energy* FotmNo.B36R5 MCTOIQimnM f\c mie hAMMr&T 10 i mi in ST26291M1 DISCLAIMER Portions of this document may be illegible in electronic image products. Images are produced from the best available original document. Age Hardening in Rapidly Solidified and Hot Isostatically Pressed Beryllium-Aluminum-Silver Alloys David H. Carter, Andrew C. McGeorge,* Loren A. Jacobson, and Paul W. Stanek Los Alamos National Laboratory, Los Alamos NM 87545 Abstract Three different alloys of beryllium, aluminum and silver were processed to powder by centrifugal atomization in a helium atmosphere. Alloy com• positions were, by weight, 50% Be, 47.5% Al, 2.5% Ag, 50% Be, 47% Al, 3% Ag, and 50% Be, 46% Al, 4% Ag. -

Of the Periodic Table
of the Periodic Table teacher notes Give your students a visual introduction to the families of the periodic table! This product includes eight mini- posters, one for each of the element families on the main group of the periodic table: Alkali Metals, Alkaline Earth Metals, Boron/Aluminum Group (Icosagens), Carbon Group (Crystallogens), Nitrogen Group (Pnictogens), Oxygen Group (Chalcogens), Halogens, and Noble Gases. The mini-posters give overview information about the family as well as a visual of where on the periodic table the family is located and a diagram of an atom of that family highlighting the number of valence electrons. Also included is the student packet, which is broken into the eight families and asks for specific information that students will find on the mini-posters. The students are also directed to color each family with a specific color on the blank graphic organizer at the end of their packet and they go to the fantastic interactive table at www.periodictable.com to learn even more about the elements in each family. Furthermore, there is a section for students to conduct their own research on the element of hydrogen, which does not belong to a family. When I use this activity, I print two of each mini-poster in color (pages 8 through 15 of this file), laminate them, and lay them on a big table. I have students work in partners to read about each family, one at a time, and complete that section of the student packet (pages 16 through 21 of this file). When they finish, they bring the mini-poster back to the table for another group to use. -

Beryllium Compounds (A) BERYLLIUM COMPOUNDS
Beryllium Compounds (A) BERYLLIUM COMPOUNDS 107-02-8 Hazard Summary Inhalation exposure to beryllium primarily occurs in the workplaces where it is mined, processed, or converted into alloys and chemicals, or from the burning of coal or fuel oil and in tobacco smoke. Acute (short-term) inhalation exposure to high levels of beryllium has been observed to cause inflammation of the lungs or acute pneumonitis (reddening and swelling of the lungs) in humans; after exposure ends, these symptoms may be reversible. Chronic (long-term) inhalation exposure of humans to beryllium has been reported to cause chronic beryllium disease (berylliosis), in which granulomatous lesions (noncancerous) develop in the lung. Human epidemiology studies are limited, but suggest a causal relationship between beryllium exposure and an increased risk of lung cancer. Inhalation exposure to beryllium has been demonstrated to cause lung cancer in rats and monkeys. EPA has classified beryllium as a Group B1, probable human carcinogen. Please Note: The main sources of information for this fact sheet are EPA's Integrated Risk Information System (IRIS) (3), which contains information on oral chronic toxicity and the RfD and inhalation chronic toxicity and the RfC, and the carcinogenic effects of beryllium including the unit cancer risk for inhalation exposure, EPA's Toxicological Review of Beryllium and Compounds (2), and the Agency for Toxic Substances and Disease Registry's (ATSDR's) Toxicological Profile for Beryllium. (1) Uses Pure beryllium and its metal alloys have applications in electrical components, tools, structural components for aircraft, missiles, and satellites, and other metal-fabricating uses. (1) Beryllium is also used in consumer products, such as televisions, calculators, and personal computers. -

WO 2016/074683 Al 19 May 2016 (19.05.2016) W P O P C T
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2016/074683 Al 19 May 2016 (19.05.2016) W P O P C T (51) International Patent Classification: (81) Designated States (unless otherwise indicated, for every C12N 15/10 (2006.01) kind of national protection available): AE, AG, AL, AM, AO, AT, AU, AZ, BA, BB, BG, BH, BN, BR, BW, BY, (21) International Application Number: BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, PCT/DK20 15/050343 DO, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, (22) International Filing Date: HN, HR, HU, ID, IL, IN, IR, IS, JP, KE, KG, KN, KP, KR, 11 November 2015 ( 11. 1 1.2015) KZ, LA, LC, LK, LR, LS, LU, LY, MA, MD, ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, OM, (25) Filing Language: English PA, PE, PG, PH, PL, PT, QA, RO, RS, RU, RW, SA, SC, (26) Publication Language: English SD, SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (30) Priority Data: PA 2014 00655 11 November 2014 ( 11. 1 1.2014) DK (84) Designated States (unless otherwise indicated, for every 62/077,933 11 November 2014 ( 11. 11.2014) US kind of regional protection available): ARIPO (BW, GH, 62/202,3 18 7 August 2015 (07.08.2015) US GM, KE, LR, LS, MW, MZ, NA, RW, SD, SL, ST, SZ, TZ, UG, ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, RU, (71) Applicant: LUNDORF PEDERSEN MATERIALS APS TJ, TM), European (AL, AT, BE, BG, CH, CY, CZ, DE, [DK/DK]; Nordvej 16 B, Himmelev, DK-4000 Roskilde DK, EE, ES, FI, FR, GB, GR, HR, HU, IE, IS, IT, LT, LU, (DK). -
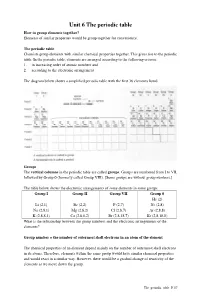
Unit 6 the Periodic Table How to Group Elements Together? Elements of Similar Properties Would Be Group Together for Convenience
Unit 6 The periodic table How to group elements together? Elements of similar properties would be group together for convenience. The periodic table Chemists group elements with similar chemical properties together. This gives rise to the periodic table. In the periodic table, elements are arranged according to the following criteria: 1. in increasing order of atomic numbers and 2. according to the electronic arrangement The diagram below shows a simplified periodic table with the first 36 elements listed. Groups The vertical columns in the periodic table are called groups . Groups are numbered from I to VII, followed by Group 0 (formerly called Group VIII). [Some groups are without group numbers.] The table below shows the electronic arrangements of some elements in some groups. Group I Group II Group VII Group 0 He (2) Li (2,1) Be (2,2) F (2,7) Ne (2,8) Na (2,8,1) Mg (2,8,2) Cl (2,8,7) Ar (2,8,8) K (2,8,8,1) Ca (2,8,8,2) Br (2,8,18,7) Kr (2,8,18,8) What is the relationship between the group numbers and the electronic arrangements of the elements? Group number = the number of outermost shell electrons in an atom of the element The chemical properties of an element depend mainly on the number of outermost shell electrons in its atoms. Therefore, elements within the same group would have similar chemical properties and would react in a similar way. However, there would be a gradual change of reactivity of the elements as we move down the group. -

Beryllium and Beryllium Compounds
BERYLLIUM AND BERYLLIUM COMPOUNDS Beryllium and beryllium compounds were considered by previous IARC Working Groups in 1971, 1979, 1987, and 1993 (IARC, 1972, 1980, 1987, 1993). Since that time, new data have become available, these have been incorporated in the Monograph, and taken into consid- eration in the present evaluation. 1. Exposure Data a very high strength-to-weight ratio. Beryllium is lighter than aluminium but is greater than 40% 1.1 Identification of the agents more rigid than steel. It has excellent electrical and thermal conductivities. Its only markedly Synonyms and molecular formulae for beryl- adverse feature is relatively pronounced brittle- lium, beryllium–aluminium and beryllium– ness, which restricts the use of metallic beryl- copper alloys, and certain beryllium compounds lium to specialized applications (WHO, 1990). are presented in Table 1.1. The list is not exhaus- Because of its low atomic number, beryllium tive, nor does it comprise necessarily the most is very permeable to X-rays. Neutron emission commercially important beryllium-containing after bombardment with α or γ rays is the most substances; rather, it indicates the range of beryl- important of its nuclear physical properties, lium compounds available. and beryllium can be used as a neutron source. Moreover, its low neutron absorptiveness and high-scattering cross-section make it a suitable 1.2 Chemical and physical properties moderator and reflector in structural materials of the agents in nuclear facilities; where most other metals absorb neutrons emitted during the fission Beryllium (atomic number, 4; relative atomic of nuclear fuel, beryllium atoms only reduce mass, 9.01) is a metal, which belongs to Group the energy of such neutrons, and reflect them IIA of the Periodic Table. -

Periodic Table Key Concepts
Periodic Table Key Concepts Periodic Table Basics The periodic table is a table of all the elements which make up matter Elements initially grouped in a table by Dmitri Mendeleev Symbols – each element has a symbol which is either a Capital Letter or a Capital Letter followed by a lower case letter Atomic Number – the number above an element’s symbol which shows the number of protons Atomic Mass – the number found below an elements symbol which shows the mass of the element. Mass = the number of protons + the number of neutrons Metals – the elements which have the properties of malleability, luster, and conductivity o These elements are good conductors of electricity & heat. o Found to the left of the zig-zag line on the periodic table Nonmetals – do not have the properties of metals. Found to the right of the zig-zag line Metalloids – elements found along the zig-zag line of the periodic table and have some properties of metals and nonmetals (B, Si, Ge, As, Sb, Te, and Po) Groups The columns going up and down (There are 18 groups) Group 1: Hydrogen, Lithium, Sodium, Potassium, Rubidium, Cesium, and Francium Elements arranged so that elements with similar properties would be in the same group. o Group 1 Alkali Metals - highly reactive metals o Group 2 Alkali Earth Metals – reactive metals o Group 3-12 Transition Metals o Group 17 Halogens – highly reactive non-metals o Group 18 Noble Gases - do not react or combine with any other elements. Elements are grouped according to their properties or reactivity Reactivity is determined by the number of electrons in an element’s outer energy level These electrons are called valence electrons Periods The rows that run from left to right on the periodic table (There are 7 periods) Period 1 contains 2 elements, Hydrogen and Helium. -

Chapter 3 Gamma-Ray and Neutron Sources R.J. Holmes
123 CHAPTER 3 GAMMA-RAY AND NEUTRON SOURCES R.J. HOLMES 125 1. GAMMA-RAY SOURCES Most y-ray sources in commercial use do not occur in nature because their half-lives are small compared with geological times. They must be produced from naturally occurring nuclides by a suitable nucle?r reaction; often this is by irradiation in a nuclear reactor. Some examples of radioisotope production are given below: 59Co (n,y)60Co T^ 5.26 y 123Sb (n,y)mSb T^ 60 d 6Li (n,a)3H T^ 12.3 y 55Mn (p,n)55Fe T, 2.7 y Commercially available sources are sealed in chemically inert capsules. The choice of the most suitable source for a particular application usually depends on the energy of the y-rays that are emitted and on the half-life of the radioisotope. In many applications, a monoenergetic source of long half-life is preferred. Calibration corrections for source decay can be made using the familiar equation - 0.693t/T, = Io e where I is the initial source intensity/ I(t) is its intensity at time o t, and T, is the half-life. Selection of the appropriate y-ray energy depends on such criteria as the energy threshold for a desired nuclear reaction and whether absorption should be due predominantly to the photoelectric effect or Compton scattering. Table 1 lists the commonly used y-ray sources together with their y-ray energies and half-lives. 2. X-RAY SOURCES Sources of radiation below an energy of about 150 keV are usually referred to as X-ray sources, although technically some of them are low energy y-ray sources, e.g. -

Copper Silica Feldspar Silver Gypsum Soda Ash Coal Sulfur Lead Zeolites
U.S. DEPARTMENT OF THE INTERIOR OPEN-FILE REPORT 01-360 U.S. GEOLOGICAL SURVEY Mineral Resources Out of the ground...into our daily lives MINERAL COMMODITIES PRECIOUS METALS AND ELEMENTS gold platinum-group elements silver NONFERROUS METALS soda ash cobalt LIGHT METALS windows paints aluminum glassware magnets beryllium tungsten fiberglass engines magnesium detergents tools titanium lights blubs light fixtures BASE METALS iron copper stained glass faucets lead paints tools zinc silica magnets abrasive and blasting sand aluminum refrigerators ceramics IRON AND FERROALLOYS plastics automobiles glass gypsum cans chromium cobalt concrete ferroalloy metals wallboard coal tar iron manganese shampoos molybdenum vermiculite asphalt nickel feldspar tungsten fire-protection boards fluorite plates glassware toothpaste wallboard paint pigments fertilizers paper concretefill material productsbricks ceramics FERTILIZER AND asphalt sand & gravel CHEMICAL INDUSTRIAL MATERIALS limestone, dolomite, and lime lithium sulfur silver nitrogen compounds and nitrate zeolites phosphate paints photographic materials soil additives potash gold fertilizers mirrors water softeners salts rubber products wiring stereo components cat litter sulfur jewelry jewelry telephones electronic components titanium beryllium CONSTRUCTION AND MANUFACTURING white paints stereos petroleum products MATERIALS toothpaste fluorescent lights enamel finishes CONSTRUCTION MATERIALS plastics concrete products micas DDDVVD automotive parts crushed stone natural gas sand and gravel lipstick