A Clade Uniting the Green Algae Mesostigma Viride and Chlorokybus
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Early Photosynthetic Eukaryotes Inhabited Low-Salinity Habitats
Early photosynthetic eukaryotes inhabited PNAS PLUS low-salinity habitats Patricia Sánchez-Baracaldoa,1, John A. Ravenb,c, Davide Pisanid,e, and Andrew H. Knollf aSchool of Geographical Sciences, University of Bristol, Bristol BS8 1SS, United Kingdom; bDivision of Plant Science, University of Dundee at the James Hutton Institute, Dundee DD2 5DA, United Kingdom; cPlant Functional Biology and Climate Change Cluster, University of Technology Sydney, Ultimo, NSW 2007, Australia; dSchool of Biological Sciences, University of Bristol, Bristol BS8 1TH, United Kingdom; eSchool of Earth Sciences, University of Bristol, Bristol BS8 1TH, United Kingdom; and fDepartment of Organismic and Evolutionary Biology, Harvard University, Cambridge, MA 02138 Edited by Peter R. Crane, Oak Spring Garden Foundation, Upperville, Virginia, and approved July 7, 2017 (received for review December 7, 2016) The early evolutionary history of the chloroplast lineage remains estimates for the origin of plastids ranging over 800 My (7). At the an open question. It is widely accepted that the endosymbiosis that same time, the ecological setting in which this endosymbiotic event established the chloroplast lineage in eukaryotes can be traced occurred has not been fully explored (8), partly because of phy- back to a single event, in which a cyanobacterium was incorpo- logenetic uncertainties and preservational biases of the fossil re- rated into a protistan host. It is still unclear, however, which cord. Phylogenomics and trait evolution analysis have pointed to a Cyanobacteria are most closely related to the chloroplast, when the freshwater origin for Cyanobacteria (9–11), providing an approach plastid lineage first evolved, and in what habitats this endosym- to address the early diversification of terrestrial biota for which the biotic event occurred. -

Complete Plastome Sequences Of
Karol et al. BMC Evolutionary Biology 2010, 10:321 http://www.biomedcentral.com/1471-2148/10/321 RESEARCH ARTICLE Open Access Complete plastome sequences of Equisetum arvense and Isoetes flaccida: implications for phylogeny and plastid genome evolution of early land plant lineages Kenneth G Karol1*, Kathiravetpillai Arumuganathan2, Jeffrey L Boore3,4, Aaron M Duffy5, Karin DE Everett6, John D Hall1, S Kellon Hansen5, Jennifer V Kuehl7, Dina F Mandoli6,8, Brent D Mishler9, Richard G Olmstead6, Karen S Renzaglia10, Paul G Wolf5 Abstract Background: Despite considerable progress in our understanding of land plant phylogeny, several nodes in the green tree of life remain poorly resolved. Furthermore, the bulk of currently available data come from only a subset of major land plant clades. Here we examine early land plant evolution using complete plastome sequences including two previously unexamined and phylogenetically critical lineages. To better understand the evolution of land plants and their plastomes, we examined aligned nucleotide sequences, indels, gene and nucleotide composition, inversions, and gene order at the boundaries of the inverted repeats. Results: We present the plastome sequences of Equisetum arvense, a horsetail, and of Isoetes flaccida,a heterosporous lycophyte. Phylogenetic analysis of aligned nucleotides from 49 plastome genes from 43 taxa supported monophyly for the following clades: embryophytes (land plants), lycophytes, monilophytes (leptosporangiate ferns + Angiopteris evecta + Psilotum nudum + Equisetum arvense), and seed plants. Resolution among the four monilophyte lineages remained moderate, although nucleotide analyses suggested that P. nudum and E. arvense form a clade sister to A. evecta + leptosporangiate ferns. Results from phylogenetic analyses of nucleotides were consistent with the distribution of plastome gene rearrangements and with analysis of sequence gaps resulting from insertions and deletions (indels). -

Neoproterozoic Origin and Multiple Transitions to Macroscopic Growth in Green Seaweeds
Neoproterozoic origin and multiple transitions to macroscopic growth in green seaweeds Andrea Del Cortonaa,b,c,d,1, Christopher J. Jacksone, François Bucchinib,c, Michiel Van Belb,c, Sofie D’hondta, f g h i,j,k e Pavel Skaloud , Charles F. Delwiche , Andrew H. Knoll , John A. Raven , Heroen Verbruggen , Klaas Vandepoeleb,c,d,1,2, Olivier De Clercka,1,2, and Frederik Leliaerta,l,1,2 aDepartment of Biology, Phycology Research Group, Ghent University, 9000 Ghent, Belgium; bDepartment of Plant Biotechnology and Bioinformatics, Ghent University, 9052 Zwijnaarde, Belgium; cVlaams Instituut voor Biotechnologie Center for Plant Systems Biology, 9052 Zwijnaarde, Belgium; dBioinformatics Institute Ghent, Ghent University, 9052 Zwijnaarde, Belgium; eSchool of Biosciences, University of Melbourne, Melbourne, VIC 3010, Australia; fDepartment of Botany, Faculty of Science, Charles University, CZ-12800 Prague 2, Czech Republic; gDepartment of Cell Biology and Molecular Genetics, University of Maryland, College Park, MD 20742; hDepartment of Organismic and Evolutionary Biology, Harvard University, Cambridge, MA 02138; iDivision of Plant Sciences, University of Dundee at the James Hutton Institute, Dundee DD2 5DA, United Kingdom; jSchool of Biological Sciences, University of Western Australia, WA 6009, Australia; kClimate Change Cluster, University of Technology, Ultimo, NSW 2006, Australia; and lMeise Botanic Garden, 1860 Meise, Belgium Edited by Pamela S. Soltis, University of Florida, Gainesville, FL, and approved December 13, 2019 (received for review June 11, 2019) The Neoproterozoic Era records the transition from a largely clear interpretation of how many times and when green seaweeds bacterial to a predominantly eukaryotic phototrophic world, creat- emerged from unicellular ancestors (8). ing the foundation for the complex benthic ecosystems that have There is general consensus that an early split in the evolution sustained Metazoa from the Ediacaran Period onward. -

Evolution and Comparative Morphology of the Euglenophyte
EVOLUTION AND COMPARATIVE MORPHOLOGY OF THE EUGLENOPHYTE PLASTID by PATRICK JERRY PAUL BROWN (Under the Direction of Mark A. Farmer) ABSTRACT My doctoral research centered on understanding the evolution of the euglenophyte protists, with special attention paid to their plastids. The euglenophytes are a widely distributed group of euglenid protists that have acquired a chloroplast via secondary symbiogenesis. The goals of my research were to 1) test the efficacy of plastid morphological and ultrastructural characters in phylogenetic analysis; 2) understand the process of plastid development and partitioning in the euglenophytes; 3) to use a plastid- encoded protein gene to determine a euglenophyte phylogeny; and 4) to perform a multi- gene analysis to uncover clues about the origins of the euglenophyte plastid. My work began with an alpha-taxonomic study that redefined the rare euglenophyte Euglena rustica. This work not only validly circumscribed the species, but also noted novel features of its habitat, cyclic migration habits, and cellular biology. This was followed by a study of plastid morphology and development in a number of diverse euglenophytes. The results of this study showed that the plastids of euglenophytes undergo drastic changes in morphology and ultrastructure over the course of a single cell division cycle. I concluded that there are four main classes of plastid development and partitioning in the euglenophytes, and that the class a given species will use is dependant on its interphase plastid morphology and the rigidity of the cell. The discovery of the class IV partitioning strategy in which cells with only one or very few plastids fragment their plastids prior to cell division was very significant. -

Botany Without Bias
editorial Botany without bias In the Gospel According to Matthew Chapter seven, Verse fve, Jesus says “frst cast out the beam out of thine own eye; and then shalt thou see clearly to cast out the mote out of thy brother’s eye”. We should remember this entreaty before too casually casting accusations of ‘plant blindness’. anguage usage helps maintain unconscious biases. In plant biology Lfor example, there is the careless use of the term ‘higher plants’, without thinking about its meaning or implication. If there is a definition of ‘higher plants’ then it is synonymous with vascular plants, but the image it conjures is of upstanding, leafy land-dwelling plants. The problem is that ‘higher’ is a charged term implying superiority of this group over their non-vascular cousins. This stratification is a manifestation of orthogenesis, the idea that evolution has both a direction and a goal. A perfect illustration of orthogenesis is the frequent meme of The Road to Homo Sapiens, the original version of which was drawn by Rudolph Zallinger for a 1965 edition of Life Nature Library1. Also known as The March of Progress, it shows a line of assumed human ancestors, starting with a gibbon-like in their News and Views3, “innovations spend much of their discussions on what Pliopithecus, processing from left to right, associated with improving water use the similarities of these organisms to becoming taller and more upright in stance, efficiency […] may be more fundamental angiosperms can tell about the history and culminating in a modern human. to the evolution of vascular plants than the of plants’ colonization of dry land, and The implication is clear, our evolutionary vascular system from which they derive much less on their characteristics and ancestors are only of interest as waypoints their name”. -

JUDD W.S. Et. Al. (2002) Plant Systematics: a Phylogenetic Approach. Chapter 7. an Overview of Green
UNCORRECTED PAGE PROOFS An Overview of Green Plant Phylogeny he word plant is commonly used to refer to any auto- trophic eukaryotic organism capable of converting light energy into chemical energy via the process of photosynthe- sis. More specifically, these organisms produce carbohydrates from carbon dioxide and water in the presence of chlorophyll inside of organelles called chloroplasts. Sometimes the term plant is extended to include autotrophic prokaryotic forms, especially the (eu)bacterial lineage known as the cyanobacteria (or blue- green algae). Many traditional botany textbooks even include the fungi, which differ dramatically in being heterotrophic eukaryotic organisms that enzymatically break down living or dead organic material and then absorb the simpler products. Fungi appear to be more closely related to animals, another lineage of heterotrophs characterized by eating other organisms and digesting them inter- nally. In this chapter we first briefly discuss the origin and evolution of several separately evolved plant lineages, both to acquaint you with these important branches of the tree of life and to help put the green plant lineage in broad phylogenetic perspective. We then focus attention on the evolution of green plants, emphasizing sev- eral critical transitions. Specifically, we concentrate on the origins of land plants (embryophytes), of vascular plants (tracheophytes), of 1 UNCORRECTED PAGE PROOFS 2 CHAPTER SEVEN seed plants (spermatophytes), and of flowering plants dons.” In some cases it is possible to abandon such (angiosperms). names entirely, but in others it is tempting to retain Although knowledge of fossil plants is critical to a them, either as common names for certain forms of orga- deep understanding of each of these shifts and some key nization (e.g., the “bryophytic” life cycle), or to refer to a fossils are mentioned, much of our discussion focuses on clade (e.g., applying “gymnosperms” to a hypothesized extant groups. -

Lateral Gene Transfer of Anion-Conducting Channelrhodopsins Between Green Algae and Giant Viruses
bioRxiv preprint doi: https://doi.org/10.1101/2020.04.15.042127; this version posted April 23, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. 1 5 Lateral gene transfer of anion-conducting channelrhodopsins between green algae and giant viruses Andrey Rozenberg 1,5, Johannes Oppermann 2,5, Jonas Wietek 2,3, Rodrigo Gaston Fernandez Lahore 2, Ruth-Anne Sandaa 4, Gunnar Bratbak 4, Peter Hegemann 2,6, and Oded 10 Béjà 1,6 1Faculty of Biology, Technion - Israel Institute of Technology, Haifa 32000, Israel. 2Institute for Biology, Experimental Biophysics, Humboldt-Universität zu Berlin, Invalidenstraße 42, Berlin 10115, Germany. 3Present address: Department of Neurobiology, Weizmann 15 Institute of Science, Rehovot 7610001, Israel. 4Department of Biological Sciences, University of Bergen, N-5020 Bergen, Norway. 5These authors contributed equally: Andrey Rozenberg, Johannes Oppermann. 6These authors jointly supervised this work: Peter Hegemann, Oded Béjà. e-mail: [email protected] ; [email protected] 20 ABSTRACT Channelrhodopsins (ChRs) are algal light-gated ion channels widely used as optogenetic tools for manipulating neuronal activity 1,2. Four ChR families are currently known. Green algal 3–5 and cryptophyte 6 cation-conducting ChRs (CCRs), cryptophyte anion-conducting ChRs (ACRs) 7, and the MerMAID ChRs 8. Here we 25 report the discovery of a new family of phylogenetically distinct ChRs encoded by marine giant viruses and acquired from their unicellular green algal prasinophyte hosts. -

Neoproterozoic Origin and Multiple Transitions to Macroscopic Growth in Green Seaweeds
bioRxiv preprint doi: https://doi.org/10.1101/668475; this version posted June 12, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Neoproterozoic origin and multiple transitions to macroscopic growth in green seaweeds Andrea Del Cortonaa,b,c,d,1, Christopher J. Jacksone, François Bucchinib,c, Michiel Van Belb,c, Sofie D’hondta, Pavel Škaloudf, Charles F. Delwicheg, Andrew H. Knollh, John A. Raveni,j,k, Heroen Verbruggene, Klaas Vandepoeleb,c,d,1,2, Olivier De Clercka,1,2 Frederik Leliaerta,l,1,2 aDepartment of Biology, Phycology Research Group, Ghent University, Krijgslaan 281, 9000 Ghent, Belgium bDepartment of Plant Biotechnology and Bioinformatics, Ghent University, Technologiepark 71, 9052 Zwijnaarde, Belgium cVIB Center for Plant Systems Biology, Technologiepark 71, 9052 Zwijnaarde, Belgium dBioinformatics Institute Ghent, Ghent University, Technologiepark 71, 9052 Zwijnaarde, Belgium eSchool of Biosciences, University of Melbourne, Melbourne, Victoria, Australia fDepartment of Botany, Faculty of Science, Charles University, Benátská 2, CZ-12800 Prague 2, Czech Republic gDepartment of Cell Biology and Molecular Genetics, University of Maryland, College Park, MD 20742, USA hDepartment of Organismic and Evolutionary Biology, Harvard University, Cambridge, Massachusetts, 02138, USA. iDivision of Plant Sciences, University of Dundee at the James Hutton Institute, Dundee, DD2 5DA, UK jSchool of Biological Sciences, University of Western Australia (M048), 35 Stirling Highway, WA 6009, Australia kClimate Change Cluster, University of Technology, Ultimo, NSW 2006, Australia lMeise Botanic Garden, Nieuwelaan 38, 1860 Meise, Belgium 1To whom correspondence may be addressed. Email [email protected], [email protected], [email protected] or [email protected]. -
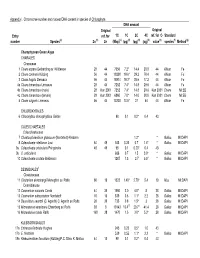
Green Appendix I
Appendix I. Chromsome number and nuclear DNA content in species of Chlorophyta DNA amount Original Original Entry ref. for 1C 1C 2C 4C ref. for C- Standard number Species(a) 2n (b) 2n (Mbp)(c) (pg)(d) (pg)(d) (pg)(d) value(e) species(f) Method(g) Charophycean Green Algae CHARALES Characeae 1 Chara aspera Detharding ex Willdenow 28 44 7056 7.2* 14.4 28.8 44 Allium Fe 2 Chara contraria Kützing 56 44 19208 19.6* 39.2 78.4 44 Allium Fe 3 Chara fragilis Desvaux 56 44 18914 19.3* 38.6 77.2 44 Allium Fe 4a Chara tomentosa Linnaeus 28 44 7252 7.4* 14.8 29.6 44 Allium Fe 4b Chara tomentosa (male) 28 Kun 2001 7252 7.4* 14.8 29.6 Kun 2001 Chara MI:EB 4c Chara tomentsoa (female) 28 Kun 2001 6860 7.0* 14.0 28.0 Kun 2001 Chara MI:EB 5 Chara vulgaris Linnaeus 56 44 13230 13.5* 27 54 44 Allium Fe CHLOROKYBALES 6 Chlorokybus atmosphyticus Geitler 98 0.1 0.2* 0.4 43 COLEOCHAETALES Coleochaetaceae 7 Chaetosphaeridium globosum (Nordstedt) Klebahn 1.2* * Gallus MI:DAPI 8 Coleochaete nitellarum Jost 84 49 343 0.35 0.7 1.4* * Gallus MI:DAPI 9a Coleochaete orbicularis Pringsheim 48 49 98 0.1 0.20* 0.4 43 9b C. orbicularis 686 0.7 1.5 3.0* * Gallus MI:DAPI 10 Coleochaete scutata Brébisson 1287 1.3 2.7 5.5* * Gallus MI:DAPI DESMIDIALES1 Closteriaceae 11 Closterium ehrenbergii Meneghini ex Ralfs 60 19 1323 1.40* 2.70* 5.4 19 Mus MI:DAPI Desmidiaceae 12 Cosmarium cucumis Corda 44 39 1960 2.0 4.0* 8 28 Gallus MI:DAPI 13 Cosmarium subcostatum Nordstedt 10 16 539 0.6 1.1* 2.2 28 Gallus MI:DAPI 14 Desmidium swartzii (C. -

Diversity and Evolution of Algae: Primary Endosymbiosis
CHAPTER TWO Diversity and Evolution of Algae: Primary Endosymbiosis Olivier De Clerck1, Kenny A. Bogaert, Frederik Leliaert Phycology Research Group, Biology Department, Ghent University, Krijgslaan 281 S8, 9000 Ghent, Belgium 1Corresponding author: E-mail: [email protected] Contents 1. Introduction 56 1.1. Early Evolution of Oxygenic Photosynthesis 56 1.2. Origin of Plastids: Primary Endosymbiosis 58 2. Red Algae 61 2.1. Red Algae Defined 61 2.2. Cyanidiophytes 63 2.3. Of Nori and Red Seaweed 64 3. Green Plants (Viridiplantae) 66 3.1. Green Plants Defined 66 3.2. Evolutionary History of Green Plants 67 3.3. Chlorophyta 68 3.4. Streptophyta and the Origin of Land Plants 72 4. Glaucophytes 74 5. Archaeplastida Genome Studies 75 Acknowledgements 76 References 76 Abstract Oxygenic photosynthesis, the chemical process whereby light energy powers the conversion of carbon dioxide into organic compounds and oxygen is released as a waste product, evolved in the anoxygenic ancestors of Cyanobacteria. Although there is still uncertainty about when precisely and how this came about, the gradual oxygenation of the Proterozoic oceans and atmosphere opened the path for aerobic organisms and ultimately eukaryotic cells to evolve. There is a general consensus that photosynthesis was acquired by eukaryotes through endosymbiosis, resulting in the enslavement of a cyanobacterium to become a plastid. Here, we give an update of the current understanding of the primary endosymbiotic event that gave rise to the Archaeplastida. In addition, we provide an overview of the diversity in the Rhodophyta, Glaucophyta and the Viridiplantae (excluding the Embryophyta) and highlight how genomic data are enabling us to understand the relationships and characteristics of algae emerging from this primary endosymbiotic event. -

From Algae to Flowering Plants
PP62CH23-Popper ARI 4 April 2011 14:20 Evolution and Diversity of Plant Cell Walls: From Algae to Flowering Plants Zoe¨ A. Popper,1 Gurvan Michel,3,4 Cecile´ Herve,´ 3,4 David S. Domozych,5 William G.T. Willats,6 Maria G. Tuohy,2 Bernard Kloareg,3,4 and Dagmar B. Stengel1 1Botany and Plant Science, and 2Molecular Glycotechnology Group, Biochemistry, School of Natural Sciences, National University of Ireland, Galway, Ireland; email: [email protected] 3CNRS and 4UPMC University Paris 6, UMR 7139 Marine Plants and Biomolecules, Station Biologique de Roscoff, F-29682 Roscoff, Bretagne, France 5Department of Biology and Skidmore Microscopy Imaging Center, Skidmore College, Saratoga Springs, New York 12866 6Department of Plant Biology and Biochemistry, Faculty of Life Sciences, University of Copenhagen, Bulowsvej,¨ 17-1870 Frederiksberg, Denmark Annu. Rev. Plant Biol. 2011. 62:567–90 Keywords First published online as a Review in Advance on xyloglucan, mannan, arabinogalactan proteins, genome, environment, February 22, 2011 multicellularity The Annual Review of Plant Biology is online at plant.annualreviews.org Abstract This article’s doi: All photosynthetic multicellular Eukaryotes, including land plants 10.1146/annurev-arplant-042110-103809 by Universidad Veracruzana on 01/08/14. For personal use only. and algae, have cells that are surrounded by a dynamic, complex, Copyright c 2011 by Annual Reviews. carbohydrate-rich cell wall. The cell wall exerts considerable biologi- All rights reserved cal and biomechanical control over individual cells and organisms, thus Annu. Rev. Plant Biol. 2011.62:567-590. Downloaded from www.annualreviews.org 1543-5008/11/0602-0567$20.00 playing a key role in their environmental interactions. -

Freshwater Algae in Britain and Ireland - Bibliography
Freshwater algae in Britain and Ireland - Bibliography Floras, monographs, articles with records and environmental information, together with papers dealing with taxonomic/nomenclatural changes since 2003 (previous update of ‘Coded List’) as well as those helpful for identification purposes. Theses are listed only where available online and include unpublished information. Useful websites are listed at the end of the bibliography. Further links to relevant information (catalogues, websites, photocatalogues) can be found on the site managed by the British Phycological Society (http://www.brphycsoc.org/links.lasso). Abbas A, Godward MBE (1964) Cytology in relation to taxonomy in Chaetophorales. Journal of the Linnean Society, Botany 58: 499–597. Abbott J, Emsley F, Hick T, Stubbins J, Turner WB, West W (1886) Contributions to a fauna and flora of West Yorkshire: algae (exclusive of Diatomaceae). Transactions of the Leeds Naturalists' Club and Scientific Association 1: 69–78, pl.1. Acton E (1909) Coccomyxa subellipsoidea, a new member of the Palmellaceae. Annals of Botany 23: 537–573. Acton E (1916a) On the structure and origin of Cladophora-balls. New Phytologist 15: 1–10. Acton E (1916b) On a new penetrating alga. New Phytologist 15: 97–102. Acton E (1916c) Studies on the nuclear division in desmids. 1. Hyalotheca dissiliens (Smith) Bréb. Annals of Botany 30: 379–382. Adams J (1908) A synopsis of Irish algae, freshwater and marine. Proceedings of the Royal Irish Academy 27B: 11–60. Ahmadjian V (1967) A guide to the algae occurring as lichen symbionts: isolation, culture, cultural physiology and identification. Phycologia 6: 127–166 Allanson BR (1973) The fine structure of the periphyton of Chara sp.