Noncanonical Coproporphyrin-Dependent Bacterial Heme Biosynthesis Pathway That Does Not Use Protoporphyrin
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
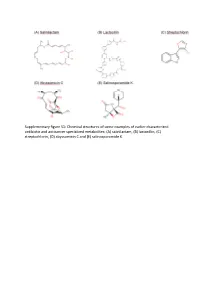
Chemical Structures of Some Examples of Earlier Characterized Antibiotic and Anticancer Specialized
Supplementary figure S1: Chemical structures of some examples of earlier characterized antibiotic and anticancer specialized metabolites: (A) salinilactam, (B) lactocillin, (C) streptochlorin, (D) abyssomicin C and (E) salinosporamide K. Figure S2. Heat map representing hierarchical classification of the SMGCs detected in all the metagenomes in the dataset. Table S1: The sampling locations of each of the sites in the dataset. Sample Sample Bio-project Site depth accession accession Samples Latitude Longitude Site description (m) number in SRA number in SRA AT0050m01B1-4C1 SRS598124 PRJNA193416 Atlantis II water column 50, 200, Water column AT0200m01C1-4D1 SRS598125 21°36'19.0" 38°12'09.0 700 and above the brine N "E (ATII 50, ATII 200, 1500 pool water layers AT0700m01C1-3D1 SRS598128 ATII 700, ATII 1500) AT1500m01B1-3C1 SRS598129 ATBRUCL SRS1029632 PRJNA193416 Atlantis II brine 21°36'19.0" 38°12'09.0 1996– Brine pool water ATBRLCL1-3 SRS1029579 (ATII UCL, ATII INF, N "E 2025 layers ATII LCL) ATBRINP SRS481323 PRJNA219363 ATIID-1a SRS1120041 PRJNA299097 ATIID-1b SRS1120130 ATIID-2 SRS1120133 2168 + Sea sediments Atlantis II - sediments 21°36'19.0" 38°12'09.0 ~3.5 core underlying ATII ATIID-3 SRS1120134 (ATII SDM) N "E length brine pool ATIID-4 SRS1120135 ATIID-5 SRS1120142 ATIID-6 SRS1120143 Discovery Deep brine DDBRINP SRS481325 PRJNA219363 21°17'11.0" 38°17'14.0 2026– Brine pool water N "E 2042 layers (DD INF, DD BR) DDBRINE DD-1 SRS1120158 PRJNA299097 DD-2 SRS1120203 DD-3 SRS1120205 Discovery Deep 2180 + Sea sediments sediments 21°17'11.0" -

Deconstruction of Lignin: from Enzymes to Microorganisms
molecules Review Deconstruction of Lignin: From Enzymes to Microorganisms Jéssica P. Silva 1, Alonso R. P. Ticona 1 , Pedro R. V. Hamann 1, Betania F. Quirino 2 and Eliane F. Noronha 1,* 1 Enzymology Laboratory, Cell Biology Department, University of Brasilia, 70910-900 Brasília, Brazil; [email protected] (J.P.S.); [email protected] (A.R.P.T.); [email protected] (P.R.V.H.) 2 Genetics and Biotechnology Laboratory, Embrapa-Agroenergy, 70770-901 Brasília, Brazil; [email protected] * Correspondence: [email protected]; Tel.: +55-61-3307-2152 Abstract: Lignocellulosic residues are low-cost abundant feedstocks that can be used for industrial applications. However, their recalcitrance currently makes lignocellulose use limited. In natural environments, microbial communities can completely deconstruct lignocellulose by synergistic action of a set of enzymes and proteins. Microbial degradation of lignin by fungi, important lignin degraders in nature, has been intensively studied. More recently, bacteria have also been described as able to break down lignin, and to have a central role in recycling this plant polymer. Nevertheless, bacterial deconstruction of lignin has not been fully elucidated yet. Direct analysis of environmental samples using metagenomics, metatranscriptomics, and metaproteomics approaches is a powerful strategy to describe/discover enzymes, metabolic pathways, and microorganisms involved in lignin breakdown. Indeed, the use of these complementary techniques leads to a better understanding of the composition, function, and dynamics of microbial communities involved in lignin deconstruction. We focus on omics approaches and their contribution to the discovery of new enzymes and reactions that impact the development of lignin-based bioprocesses. -

Table S4. Phylogenetic Distribution of Bacterial and Archaea Genomes in Groups A, B, C, D, and X
Table S4. Phylogenetic distribution of bacterial and archaea genomes in groups A, B, C, D, and X. Group A a: Total number of genomes in the taxon b: Number of group A genomes in the taxon c: Percentage of group A genomes in the taxon a b c cellular organisms 5007 2974 59.4 |__ Bacteria 4769 2935 61.5 | |__ Proteobacteria 1854 1570 84.7 | | |__ Gammaproteobacteria 711 631 88.7 | | | |__ Enterobacterales 112 97 86.6 | | | | |__ Enterobacteriaceae 41 32 78.0 | | | | | |__ unclassified Enterobacteriaceae 13 7 53.8 | | | | |__ Erwiniaceae 30 28 93.3 | | | | | |__ Erwinia 10 10 100.0 | | | | | |__ Buchnera 8 8 100.0 | | | | | | |__ Buchnera aphidicola 8 8 100.0 | | | | | |__ Pantoea 8 8 100.0 | | | | |__ Yersiniaceae 14 14 100.0 | | | | | |__ Serratia 8 8 100.0 | | | | |__ Morganellaceae 13 10 76.9 | | | | |__ Pectobacteriaceae 8 8 100.0 | | | |__ Alteromonadales 94 94 100.0 | | | | |__ Alteromonadaceae 34 34 100.0 | | | | | |__ Marinobacter 12 12 100.0 | | | | |__ Shewanellaceae 17 17 100.0 | | | | | |__ Shewanella 17 17 100.0 | | | | |__ Pseudoalteromonadaceae 16 16 100.0 | | | | | |__ Pseudoalteromonas 15 15 100.0 | | | | |__ Idiomarinaceae 9 9 100.0 | | | | | |__ Idiomarina 9 9 100.0 | | | | |__ Colwelliaceae 6 6 100.0 | | | |__ Pseudomonadales 81 81 100.0 | | | | |__ Moraxellaceae 41 41 100.0 | | | | | |__ Acinetobacter 25 25 100.0 | | | | | |__ Psychrobacter 8 8 100.0 | | | | | |__ Moraxella 6 6 100.0 | | | | |__ Pseudomonadaceae 40 40 100.0 | | | | | |__ Pseudomonas 38 38 100.0 | | | |__ Oceanospirillales 73 72 98.6 | | | | |__ Oceanospirillaceae -

Evolution of the 3-Hydroxypropionate Bicycle and Recent Transfer of Anoxygenic Photosynthesis Into the Chloroflexi
Evolution of the 3-hydroxypropionate bicycle and recent transfer of anoxygenic photosynthesis into the Chloroflexi Patrick M. Shiha,b,1, Lewis M. Wardc, and Woodward W. Fischerc,1 aFeedstocks Division, Joint BioEnergy Institute, Emeryville, CA 94608; bEnvironmental Genomics and Systems Biology Division, Lawrence Berkeley National Laboratory, Berkeley, CA 94720; and cDivision of Geological and Planetary Sciences, California Institute of Technology, Pasadena, CA 91125 Edited by Bob B. Buchanan, University of California, Berkeley, CA, and approved August 21, 2017 (received for review June 14, 2017) Various lines of evidence from both comparative biology and the provide a hard geological constraint on these analyses, the timing geologic record make it clear that the biochemical machinery for of these evolutionary events remains relative, thus highlighting anoxygenic photosynthesis was present on early Earth and provided the uncertainty in our understanding of when and how anoxy- the evolutionary stock from which oxygenic photosynthesis evolved genic photosynthesis may have originated. ca. 2.3 billion years ago. However, the taxonomic identity of these A less recognized alternative is that anoxygenic photosynthesis early anoxygenic phototrophs is uncertain, including whether or not might have been acquired in modern bacterial clades relatively they remain extant. Several phototrophic bacterial clades are thought recently. This possibility is supported by the observation that to have evolved before oxygenic photosynthesis emerged, including anoxygenic photosynthesis often sits within a derived position in the Chloroflexi, a phylum common across a wide range of modern the phyla in which it is found (3). Moreover, it is increasingly environments. Although Chloroflexi have traditionally been thought being recognized that horizontal gene transfer (HGT) has likely to be an ancient phototrophic lineage, genomics has revealed a much played a major role in the distribution of phototrophy (8–10). -

Meiothermus Ruber Type Strain (21T)
Standards in Genomic Sciences (2010) 3:26-36 DOI:10.4056/sigs.1032748 Complete genome sequence of Meiothermus ruber type strain (21T) Brian J Tindall1, Johannes Sikorski1, Susan Lucas2, Eugene Goltsman2, Alex Copeland2, Tijana Glavina Del Rio2, Matt Nolan2, Hope Tice2, Jan-Fang Cheng2, Cliff Han2,3, Sam Pitluck2, Konstantinos Liolios2, Natalia Ivanova2, Konstantinos Mavromatis2, Galina Ovchinnikova2, Amrita Pati2, Regine Fähnrich1, Lynne Goodwin2,3, Amy Chen4, Krishna Palaniappan4, Miriam Land2,5, Loren Hauser2,5, Yun-Juan Chang2,5, Cynthia D. Jeffries2,5, Manfred Rohde6, Markus Göker1, Tanja Woyke2, James Bristow2, Jonathan A. Eisen2,7, Victor Markowitz4, Philip Hugenholtz2, Nikos C. Kyrpides2, Hans-Peter Klenk1, and Alla Lapidus2* 1 DSMZ - German Collection of Microorganisms and Cell Cultures GmbH, Braunschweig, Germany 2 DOE Joint Genome Institute, Walnut Creek, California, USA 3 Los Alamos National Laboratory, Bioscience Division, Los Alamos, New Mexico, USA 4 Biological Data Management and Technology Center, Lawrence Berkeley National Laboratory, Berkeley, California, USA 5 Oak Ridge National Laboratory, Oak Ridge, Tennessee, USA 6 HZI – Helmholtz Centre for Infection Research, Braunschweig, Germany 7 University of California Davis Genome Center, Davis, California, USA *Corresponding author: Alla Lapidus Keywords: thermophilic, aerobic, non-motile, free-living, Gram-negative, Thermales, Deino- cocci, GEBA Meiothermus ruber (Loginova et al. 1984) Nobre et al. 1996 is the type species of the genus Meiothermus. This thermophilic genus is of special interest, as its members share relatively low degrees of 16S rRNA gene sequence similarity and constitute a separate evolutionary li- neage from members of the genus Thermus, from which they can generally be distinguished by their slightly lower temperature optima. -

Distribution of Arsenite-Oxidizing Bacteria and Its Correlation with Environmental Factors in Geothermal Areas of Tengchong, Yunnan, China
E3S Web of Conferences 98, 02002 (2019) https://doi.org/10.1051/e3sconf/20199802002 WRI-16 Distribution of arsenite-oxidizing bacteria and its correlation with environmental factors in geothermal areas of Tengchong, Yunnan, China Ping Li1,*, Dawei Jiang1, and Zhou Jiang1,2 1State Key Laboratory of Biogeology and Environmental Geology, China University of Geosciences, Wuhan, China 2School of Environmental Studies, China University of Geosciences, Wuhan, China Abstract. Arsenic (As) is an ubiquitous constituent in geothermal water. Arsenite (AsIII) is oxidized via microbial processes as the waters equilibrate with oxygen in the geothermal effluent. The distribution of arsenite oxdizing bacteria and its correlation with environment factors were studied in Tengchong geothermal areas of Yunnan, China. A total of 230 aioA clone sequences were obtained and these sequences were affiliated with four phyla: Betaproteobacteria, Alphaproteobacteria, Deinococcus- Thermus and Aquificae. Temperature was negatively correlated with aioA diversity and was the only environment factor that had correlation with diversity index. Betaproteobacteria was mainly distributed in low temperature (T = 28 to 43 oC) and circumneutral or light alkaline (pH = 7 to 9) springs; Alphaproteobacteria was mainly predominant in low pH (pH = 3.3 to 3.6) springs; Deinococcus-Thermus and Aquificae mainly inhabited in high temperature (T=55 to 78 oC) springs with a wide range of pH. Usually, Deinococcus-Thermus was dominant when springs had a pH within 4.0 to 8.0. Aquificae was dominated in springs with pH > 8.0 or pH < 4.0. 1 Diversity of aioA gene A total of 230 aioA gene clone sequences from 10 sample sites were subjected to sequence similarity analysis. -

Planctomycetes Cell Viability Studies: Perspectives of Toxicity Assessment Using Zeta Potential 2.º CICLO FCUP CIIMAR 2013
MSc FCUP 0 Planctomycetes cell viability studies: perspectives of toxicity assessment using zeta potential 2.º CICLO FCUP CIIMAR 2013 Planctomycetes of toxicity assessment Planctomycetes cell cell viability viability studies: using perspectives of studies zeta potential toxicity assessment : perspectives using zeta potential Carlos Eduardo de Bento Flores Dissertação de Mestrado apresentada à BentoEduardo Flores de Carlos Faculdade de Ciências da Universidade do Porto, Centro Interdisciplinar de Investigação Marinha Ambiental. Biologia Celular e Molecular 2012 / 2013 FCUP 1 Planctomycetes cell viability studies: perspectives of toxicity assessment using zeta potential Planctomycetes cell viability studies: perspectives of toxicity assessment using zeta potential Carlos Eduardo de Bento Flores Mestrado em Biologia Celular e Molecular Departamento de Biologia 2013 Orientador Olga Maria Oliveira da Silva Lage, Professora Auxiliar, Faculdade de Ciências da Universidade do Porto FCUP 2 Planctomycetes cell viability studies: perspectives of toxicity assessment using zeta potential FCUP 3 Planctomycetes cell viability studies: perspectives of toxicity assessment using zeta potential Todas as correções determinadas pelo júri, e só essas, foram efetuadas. O Presidente do Júri, Porto, ______/______/_________ FCUP 4 Planctomycetes cell viability studies: perspectives of toxicity assessment using zeta potential FCUP 5 Planctomycetes cell viability studies: perspectives of toxicity assessment using zeta potential Dissertação para a candidatura ao grau de Mestre em Biologia Celular e Molecular submetida à Faculdade de Ciências da Universidade do Porto. O presente trabalho foi desenvolvido sob a orientação científica da Professora Doutora Olga Maria Oliveira da Silva Lage e foi realizado na Faculdade de Ciências da Universidade do Porto, com a colaboração do Centro Interdisciplinar de Investigação Marinha e Ambiental. -

Phylogeny of Nitrogenase Structural and Assembly Components Reveals New Insights Into the Origin and Distribution of Nitrogen Fixation Across Bacteria and Archaea
microorganisms Article Phylogeny of Nitrogenase Structural and Assembly Components Reveals New Insights into the Origin and Distribution of Nitrogen Fixation across Bacteria and Archaea Amrit Koirala 1 and Volker S. Brözel 1,2,* 1 Department of Biology and Microbiology, South Dakota State University, Brookings, SD 57006, USA; [email protected] 2 Department of Biochemistry, Genetics and Microbiology, University of Pretoria, Pretoria 0004, South Africa * Correspondence: [email protected]; Tel.: +1-605-688-6144 Abstract: The phylogeny of nitrogenase has only been analyzed using the structural proteins NifHDK. As nifHDKENB has been established as the minimum number of genes necessary for in silico predic- tion of diazotrophy, we present an updated phylogeny of diazotrophs using both structural (NifHDK) and cofactor assembly proteins (NifENB). Annotated Nif sequences were obtained from InterPro from 963 culture-derived genomes. Nif sequences were aligned individually and concatenated to form one NifHDKENB sequence. Phylogenies obtained using PhyML, FastTree, RapidNJ, and ASTRAL from individuals and concatenated protein sequences were compared and analyzed. All six genes were found across the Actinobacteria, Aquificae, Bacteroidetes, Chlorobi, Chloroflexi, Cyanobacteria, Deferribacteres, Firmicutes, Fusobacteria, Nitrospira, Proteobacteria, PVC group, and Spirochaetes, as well as the Euryarchaeota. The phylogenies of individual Nif proteins were very similar to the overall NifHDKENB phylogeny, indicating the assembly proteins have evolved together. Our higher resolution database upheld the three cluster phylogeny, but revealed undocu- Citation: Koirala, A.; Brözel, V.S. mented horizontal gene transfers across phyla. Only 48% of the 325 genera containing all six nif genes Phylogeny of Nitrogenase Structural and Assembly Components Reveals are currently supported by biochemical evidence of diazotrophy. -

Concerted Gene Recruitment in Early Plant Evolution Jinling Huang* and J Peter Gogarten†
Open Access Research2008HuangVolume and 9, Issue Gogarten 7, Article R109 Concerted gene recruitment in early plant evolution Jinling Huang* and J Peter Gogarten† Addresses: *Department of Biology, Howell Science Complex, East Carolina University, Greenville, NC 27858, USA. †Department of Molecular and Cell Biology, University of Connecticut, 91 North Eagleville Road, Storrs, CT 06269, USA. Correspondence: Jinling Huang. Email: [email protected] Published: 8 July 2008 Received: 30 April 2008 Revised: 24 June 2008 Genome Biology 2008, 9:R109 (doi:10.1186/gb-2008-9-7-r109) Accepted: 8 July 2008 The electronic version of this article is the complete one and can be found online at http://genomebiology.com/2008/9/7/R109 © 2008 Huang and Gogarten; licensee BioMed Central Ltd. This is an open access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Eukaryotic<p>Analysesto the split ofhorizontal ofred the algae red gene andalgal transfergreen <it>Cyanidioschyzon</it> plants.</p> genome identified 37 genes that were acquired from non-organellar sources prior Abstract Background: Horizontal gene transfer occurs frequently in prokaryotes and unicellular eukaryotes. Anciently acquired genes, if retained among descendants, might significantly affect the long-term evolution of the recipient lineage. However, no systematic studies on the scope of anciently acquired genes and their impact on macroevolution are currently available in eukaryotes. Results: Analyses of the genome of the red alga Cyanidioschyzon identified 37 genes that were acquired from non-organellar sources prior to the split of red algae and green plants. -

Systema Naturae 2000 (Phylum, 6 Nov 2017)
The Taxonomicon Systema Naturae 2000 Classification of Domain Bacteria (prokaryotes) down to Phylum Compiled by Drs. S.J. Brands Universal Taxonomic Services 6 Nov 2017 Systema Naturae 2000 - Domain Bacteria - Domain Bacteria Woese et al. 1990 1 Genus †Eoleptonema Schopf 1983, incertae sedis 2 Genus †Primaevifilum Schopf 1983, incertae sedis 3 Genus †Archaeotrichion Schopf 1968, incertae sedis 4 Genus †Siphonophycus Schopf 1968, incertae sedis 5 Genus Bactoderma Tepper and Korshunova 1973 (Approved Lists 1980), incertae sedis 6 Genus Stibiobacter Lyalikova 1974 (Approved Lists 1980), incertae sedis 7.1.1.1.1.1 Superphylum "Proteobacteria" Craig et al. 2010 1.1 Phylum "Alphaproteobacteria" 1.2.1 Phylum "Acidithiobacillia" 1.2.2.1 Phylum "Gammaproteobacteria" 1.2.2.2.1 Candidate phylum Muproteobacteria (RIF23) Anantharaman et al. 2016 1.2.2.2.2 Phylum "Betaproteobacteria" 2 Phylum "Zetaproteobacteria" 7.1.1.1.1.2 Phylum "Deltaproteobacteria_1" 7.1.1.1.2.1.1.1 Phylum "Deltaproteobacteria" [polyphyletic] 7.1.1.1.2.1.1.2.1 Phylum "Deltaproteobacteria_2" 7.1.1.1.2.1.1.2.2 Phylum "Deltaproteobacteria_3" 7.1.1.1.2.1.2 Candidate phylum Dadabacteria (CSP1-2) Hug et al. 2015 7.1.1.1.2.2.1 Candidate phylum "MBNT15" 7.1.1.1.2.2.2 Candidate phylum "Uncultured Bacterial Phylum 10 (UBP10)" Parks et al. 2017 7.1.1.2.1 Phylum "Nitrospirae_1" 7.1.1.2.2 Phylum Chrysiogenetes Garrity and Holt 2001 7.1.2.1.1 Phylum "Nitrospirae" Garrity and Holt 2001 [polyphyletic] 7.1.2.1.2.1.1 Candidate phylum Rokubacteria (CSP1-6) Hug et al. -

Research Article Review Jmb
J. Microbiol. Biotechnol. (2017), 27(0), 1–7 https://doi.org/10.4014/jmb.1707.07027 Research Article Review jmb Methods 20,546 sequences and all the archaeal datasets were normalized to 21,154 sequences by the “sub.sample” Bioinformatics Analysis command. The filtered sequences were classified against The raw read1 and read2 datasets was demultiplexed by the SILVA 16S reference database (Release 119) using a trimming the barcode sequences with no more than 1 naïve Bayesian classifier built in Mothur with an 80% mismatch. Then the sequences with the same ID were confidence score [5]. Sequences passing through all the picked from the remaining read1 and read2 datasets by a filtration were also clustered into OTUs at 6% dissimilarity self-written python script. Bases with average quality score level. Then a “classify.otu” function was utilized to assign lower than 25 over a 25 bases sliding window were the phylogenetic information to each OTU. excluded and sequences which contained any ambiguous base or had a final length shorter than 200 bases were Reference abandoned using Sickle [1]. The paired reads were assembled into contigs and any contigs with an ambiguous 1. Joshi NA, FJ. 2011. Sickle: A sliding-window, adaptive, base, more than 8 homopolymeric bases and fewer than 10 quality-based trimming tool for FastQ files (Version 1.33) bp overlaps were culled. After that, the contigs were [Software]. further trimmed to get rid of the contigs that have more 2. Schloss PD. 2010. The Effects of Alignment Quality, than 1 forward primer mismatch and 2 reverse primer Distance Calculation Method, Sequence Filtering, and Region on the Analysis of 16S rRNA Gene-Based Studies. -

A Genomic Journey Through a Genus of Large DNA Viruses
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Virology Papers Virology, Nebraska Center for 2013 Towards defining the chloroviruses: a genomic journey through a genus of large DNA viruses Adrien Jeanniard Aix-Marseille Université David D. Dunigan University of Nebraska-Lincoln, [email protected] James Gurnon University of Nebraska-Lincoln, [email protected] Irina V. Agarkova University of Nebraska-Lincoln, [email protected] Ming Kang University of Nebraska-Lincoln, [email protected] See next page for additional authors Follow this and additional works at: https://digitalcommons.unl.edu/virologypub Part of the Biological Phenomena, Cell Phenomena, and Immunity Commons, Cell and Developmental Biology Commons, Genetics and Genomics Commons, Infectious Disease Commons, Medical Immunology Commons, Medical Pathology Commons, and the Virology Commons Jeanniard, Adrien; Dunigan, David D.; Gurnon, James; Agarkova, Irina V.; Kang, Ming; Vitek, Jason; Duncan, Garry; McClung, O William; Larsen, Megan; Claverie, Jean-Michel; Van Etten, James L.; and Blanc, Guillaume, "Towards defining the chloroviruses: a genomic journey through a genus of large DNA viruses" (2013). Virology Papers. 245. https://digitalcommons.unl.edu/virologypub/245 This Article is brought to you for free and open access by the Virology, Nebraska Center for at DigitalCommons@University of Nebraska - Lincoln. It has been accepted for inclusion in Virology Papers by an authorized administrator of DigitalCommons@University of Nebraska - Lincoln. Authors Adrien Jeanniard, David D. Dunigan, James Gurnon, Irina V. Agarkova, Ming Kang, Jason Vitek, Garry Duncan, O William McClung, Megan Larsen, Jean-Michel Claverie, James L. Van Etten, and Guillaume Blanc This article is available at DigitalCommons@University of Nebraska - Lincoln: https://digitalcommons.unl.edu/ virologypub/245 Jeanniard, Dunigan, Gurnon, Agarkova, Kang, Vitek, Duncan, McClung, Larsen, Claverie, Van Etten & Blanc in BMC Genomics (2013) 14.