Deconstruction of Lignin: from Enzymes to Microorganisms
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
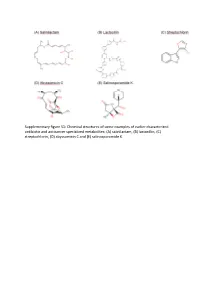
Chemical Structures of Some Examples of Earlier Characterized Antibiotic and Anticancer Specialized
Supplementary figure S1: Chemical structures of some examples of earlier characterized antibiotic and anticancer specialized metabolites: (A) salinilactam, (B) lactocillin, (C) streptochlorin, (D) abyssomicin C and (E) salinosporamide K. Figure S2. Heat map representing hierarchical classification of the SMGCs detected in all the metagenomes in the dataset. Table S1: The sampling locations of each of the sites in the dataset. Sample Sample Bio-project Site depth accession accession Samples Latitude Longitude Site description (m) number in SRA number in SRA AT0050m01B1-4C1 SRS598124 PRJNA193416 Atlantis II water column 50, 200, Water column AT0200m01C1-4D1 SRS598125 21°36'19.0" 38°12'09.0 700 and above the brine N "E (ATII 50, ATII 200, 1500 pool water layers AT0700m01C1-3D1 SRS598128 ATII 700, ATII 1500) AT1500m01B1-3C1 SRS598129 ATBRUCL SRS1029632 PRJNA193416 Atlantis II brine 21°36'19.0" 38°12'09.0 1996– Brine pool water ATBRLCL1-3 SRS1029579 (ATII UCL, ATII INF, N "E 2025 layers ATII LCL) ATBRINP SRS481323 PRJNA219363 ATIID-1a SRS1120041 PRJNA299097 ATIID-1b SRS1120130 ATIID-2 SRS1120133 2168 + Sea sediments Atlantis II - sediments 21°36'19.0" 38°12'09.0 ~3.5 core underlying ATII ATIID-3 SRS1120134 (ATII SDM) N "E length brine pool ATIID-4 SRS1120135 ATIID-5 SRS1120142 ATIID-6 SRS1120143 Discovery Deep brine DDBRINP SRS481325 PRJNA219363 21°17'11.0" 38°17'14.0 2026– Brine pool water N "E 2042 layers (DD INF, DD BR) DDBRINE DD-1 SRS1120158 PRJNA299097 DD-2 SRS1120203 DD-3 SRS1120205 Discovery Deep 2180 + Sea sediments sediments 21°17'11.0" -

Oxidative Degradation of Non-Phenolic Lignin During Lipid Peroxidation by Fungal Manganese Peroxidase
FEBS Letters 354 (1994) 297-300 FEBS 14759 Oxidative degradation of non-phenolic lignin during lipid peroxidation by fungal manganese peroxidase Wuli Bao, Yaichi Fukushima, Kenneth A. Jensen Jr., Mark A. Moen, Kenneth E. Hammel* USDA Forest Products Laboratory, Madison, WI 53705, USA Received 3 October 1994 Abstract A non-phenolic lignin model dimer, 1-(4-ethoxy-3-methoxyphenyl)-2-phenoxypropane-l ,3-diol, was oxidized by a lipid peroxidation system that consisted of a fungal manganese peroxidase, Mn(II), and unsaturated fatty acid esters. The reaction products included 1-(4-ethoxy-3- methoxyphenyl)-1-oxo-2-phenoxy-3-hydroxypropane and 1-(4-ethoxy-3-methoxyphenyl)- 1-oxo-3-hydroxypropane, indicating that substrate oxida- tion occurred via benzylic hydrogen abstraction. The peroxidation system depolymerized both exhaustively methylated (non-phenolic) and unmeth- ylated (phenolic) synthetic lignins efficiently. It may therefore enable white-rot fungi to accomplish the initial delignification of wood. Key words: Manganese peroxidase; Lipid peroxidation; Ligninolysis; Non-phenolic lignin; White-rot fungus 1. Introduction 2. Materials and methods White-rot fungi have evolved unique ligninolytic mecha- 2.1. Reagents l-(4-Ethoxy-3-methoxyphenyl)-2-phenoxypropane-l,3-diol (I) [12] nisms, which enable them to play an essential role in terrestrial 1-(4-ethoxy-3-methoxyphenyl)-l-oxo-2-phenoxy-3-hydroxypropane ecosystems by degrading and recycling lignified biomass. The (II) [13], and l-(4-ethoxy-3-methoxyphenyl)-l-oxo-3-hydroxypropane chemical recalcitrance, random structure, and large size of lig- (III) [14] were prepared by the indicated methods.I labeled with 14C at Cl (0.066 mCi· mmol-1) was prepared in the same manner as the unla- nin require that these mechanisms be oxidative, non-specific, 14 beled compound, using [1- C]acetic acid as the labeled precursor. -

Evolution of the 3-Hydroxypropionate Bicycle and Recent Transfer of Anoxygenic Photosynthesis Into the Chloroflexi
Evolution of the 3-hydroxypropionate bicycle and recent transfer of anoxygenic photosynthesis into the Chloroflexi Patrick M. Shiha,b,1, Lewis M. Wardc, and Woodward W. Fischerc,1 aFeedstocks Division, Joint BioEnergy Institute, Emeryville, CA 94608; bEnvironmental Genomics and Systems Biology Division, Lawrence Berkeley National Laboratory, Berkeley, CA 94720; and cDivision of Geological and Planetary Sciences, California Institute of Technology, Pasadena, CA 91125 Edited by Bob B. Buchanan, University of California, Berkeley, CA, and approved August 21, 2017 (received for review June 14, 2017) Various lines of evidence from both comparative biology and the provide a hard geological constraint on these analyses, the timing geologic record make it clear that the biochemical machinery for of these evolutionary events remains relative, thus highlighting anoxygenic photosynthesis was present on early Earth and provided the uncertainty in our understanding of when and how anoxy- the evolutionary stock from which oxygenic photosynthesis evolved genic photosynthesis may have originated. ca. 2.3 billion years ago. However, the taxonomic identity of these A less recognized alternative is that anoxygenic photosynthesis early anoxygenic phototrophs is uncertain, including whether or not might have been acquired in modern bacterial clades relatively they remain extant. Several phototrophic bacterial clades are thought recently. This possibility is supported by the observation that to have evolved before oxygenic photosynthesis emerged, including anoxygenic photosynthesis often sits within a derived position in the Chloroflexi, a phylum common across a wide range of modern the phyla in which it is found (3). Moreover, it is increasingly environments. Although Chloroflexi have traditionally been thought being recognized that horizontal gene transfer (HGT) has likely to be an ancient phototrophic lineage, genomics has revealed a much played a major role in the distribution of phototrophy (8–10). -

Bioresources.Com
PEER-REVIEWED ARTICLE bioresources.com HYPERPRODUCTIVITY OF EXTRACELLULAR ENZYMES FROM INDIGENOUS WHITE ROT FUNGI (P. chrysosporium IBL-03) BY UTILIZING AGRO-WASTES Muhammad Asgher, Nadeem Ahmed, and Hafiz Muhammad Nasir Iqbal* An indigenous locally isolated white rot fungal strain Phanerochaete chrysosporium IBL-03 was investigated for the hyper-production of ligninolytic enzymes from different agro-industrial wastes including wheat straw, rice straw, banana stalks, corncobs, corn stover, and sugarcane bagasse as substrate material in still culture fermentation technique. Screening experiments were performed at 30oC from 1 to 10 days and maximum enzyme activities were recorded after the 5th day of incubation on banana stalk. P. chrysosporium IBL-03 produced highest activities of lignin peroxidase (LiP) and manganese peroxidase (MnP) but no laccase activity was detected in any fermented culture media. Production of ligninolytic enzymes was substantially enhanced through the optimization process. When banana stalk at 66.6 % moisture level and pH 4.5 was inoculated with 5mL spore suspension of P. chrysosporium IBL-03 at 35oC in the presence of molasses (1%) as carbon source, ammonium sulfate (0.2%) as nitrogen supplement, (1%) Tween-80 (0.3 mL) as surfactant and mediators (MnSO4 and veratryl alcohol) enhanced the LiP and MnP production up to 1040 and 965 (U/mL), respectively. Keywords: Phanerochaete chrysosporium IBL-03; Extracellular enzymes; Agro-wastes; SSF; Optimization Contact information: Department of Chemistry and Biochemistry, University of Agriculture, Faisalabad Pakistan; * Corresponding Author: Telephone# +92-307-6715243 E-mail: [email protected] INTRODUCTION White rot fungi (WRF) are among the most robust micro-organisms, having the ability to degrade the major components of lignocellulosic sources, cellulose, hemicelluloses, and lignin as available hydrolyzable nutrients efficiently through their nonspecific and non stereo-selective extracellular enzyme system (Papinutti and Forchiassin 2007; Ruhl et al. -

Kinetic Approaches to Measuring Peroxiredoxin Reactivity
Mol. Cells 2016; 39(1): 26-30 http://dx.doi.org/10.14348/molcells.2016.2325 Molecules and Cells http://molcells.org Established in 1990 Kinetic Approaches to Measuring Peroxiredoxin Reactivity Christine C. Winterbourn*, and Alexander V. Peskin Peroxiredoxins are ubiquitous thiol proteins that catalyse demonstrated surprisingly low reactivity with thiol reagents such the breakdown of peroxides and regulate redox activity in as iodoacetamide and other oxidants such as chloramines the cell. Kinetic analysis of their reactions is required in (Peskin et al., 2007) and it is clear that the low pKa of the active order to identify substrate preferences, to understand how site thiol is insufficient to confer the high peroxide reactivity. In molecular structure affects activity and to establish their fact, typical low molecular weight and protein thiolates react -1 -1 physiological functions. Various approaches can be taken, with H2O2 with a rate constant of 20 M s whereas values of including the measurement of rates of individual steps in Prxs are 105-106 fold higher (Winterbourn and Hampton, 2008). the reaction pathway by stopped flow or competitive kinet- An elegant series of structural and mutational studies (Hall et al., ics, classical enzymatic analysis and measurement of pe- 2010; Nakamura et al., 2010; Nagy et al., 2011) have shown roxidase activity. Each methodology has its strengths and that to get sufficient rate enhancement, it is necessary to acti- they can often give complementary information. However, vate the peroxide. As discussed in detail elsewhere (Hall et al., it is important to understand the experimental conditions 2010; 2011), this involves formation of a transition state in of the assay so as to interpret correctly what parameter is which hydrogen bonding of the peroxide to conserved Arg and being measured. -

Prokaryotic Origins of the Non-Animal Peroxidase Superfamily and Organelle-Mediated Transmission to Eukaryotes
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector Genomics 89 (2007) 567–579 www.elsevier.com/locate/ygeno Prokaryotic origins of the non-animal peroxidase superfamily and organelle-mediated transmission to eukaryotes Filippo Passardi a, Nenad Bakalovic a, Felipe Karam Teixeira b, Marcia Margis-Pinheiro b,c, ⁎ Claude Penel a, Christophe Dunand a, a Laboratory of Plant Physiology, University of Geneva, Quai Ernest-Ansermet 30, CH-1211 Geneva 4, Switzerland b Department of Genetics, Institute of Biology, Federal University of Rio de Janeiro, Rio de Janeiro, Brazil c Department of Genetics, Federal University of Rio Grande do Sul, Rio Grande do Sul, Brazil Received 16 June 2006; accepted 18 January 2007 Available online 13 March 2007 Abstract Members of the superfamily of plant, fungal, and bacterial peroxidases are known to be present in a wide variety of living organisms. Extensive searching within sequencing projects identified organisms containing sequences of this superfamily. Class I peroxidases, cytochrome c peroxidase (CcP), ascorbate peroxidase (APx), and catalase peroxidase (CP), are known to be present in bacteria, fungi, and plants, but have now been found in various protists. CcP sequences were detected in most mitochondria-possessing organisms except for green plants, which possess only ascorbate peroxidases. APx sequences had previously been observed only in green plants but were also found in chloroplastic protists, which acquired chloroplasts by secondary endosymbiosis. CP sequences that are known to be present in prokaryotes and in Ascomycetes were also detected in some Basidiomycetes and occasionally in some protists. -

Bioresources.Com
PEER-REVIEWED REVIEW ARTICLE bioresources.com BIOLOGICAL PRETREATMENT OF LIGNOCELLULOSES WITH WHITE-ROT FUNGI AND ITS APPLICATIONS: A REVIEW a,b c, d Isroi, Ria Millati, * Siti Syamsiah, Claes Niklasson,b Muhammad Nur Cahyanto,c f Knut Lundquist,e and Mohammad J. Taherzadeh Lignocellulosic carbohydrates, i.e. cellulose and hemicellulose, have abundant potential as feedstock for production of biofuels and chemicals. However, these carbohydrates are generally infiltrated by lignin. Breakdown of the lignin barrier will alter lignocelluloses structures and make the carbohydrates accessible for more efficient bioconversion. White-rot fungi produce ligninolytic enzymes (lignin peroxidase, manganese peroxidase, and laccase) and efficiently mineralise lignin into CO2 and H2O. Biological pretreatment of lignocelluloses using white-rot fungi has been used for decades for ruminant feed, enzymatic hydrolysis, and biopulping. Application of white-rot fungi capabilities can offer environmentally friendly processes for utilising lignocelluloses over physical or chemical pretreatment. This paper reviews white-rot fungi, ligninolytic enzymes, the effect of biological pretreatment on biomass characteristics, and factors affecting biological pretreatment. Application of biological pretreatment for enzymatic hydrolysis, biofuels (bioethanol, biogas and pyrolysis), biopulping, biobleaching, animal feed, and enzymes production are also discussed. Keywords: Biological pretreatment; White-rot fungi; Lignocellulose; Solid-state fermentation; Lignin peroxidase; -

Textile Dye Biodecolorization by Manganese Peroxidase: a Review
molecules Review Textile Dye Biodecolorization by Manganese Peroxidase: A Review Yunkang Chang 1,2, Dandan Yang 2, Rui Li 2, Tao Wang 2,* and Yimin Zhu 1,* 1 Institute of Environmental Remediation, Dalian Maritime University, Dalian 116026, China; [email protected] 2 The Lab of Biotechnology Development and Application, School of Biological Science, Jining Medical University, No. 669 Xueyuan Road, Donggang District, Rizhao 276800, China; [email protected] (D.Y.); [email protected] (R.L.) * Correspondence: [email protected] (T.W.); [email protected] (Y.Z.); Tel.: +86-063-3298-3788 (T.W.); +86-0411-8472-6992 (Y.Z.) Abstract: Wastewater emissions from textile factories cause serious environmental problems. Man- ganese peroxidase (MnP) is an oxidoreductase with ligninolytic activity and is a promising biocatalyst for the biodegradation of hazardous environmental contaminants, and especially for dye wastewater decolorization. This article first summarizes the origin, crystal structure, and catalytic cycle of MnP, and then reviews the recent literature on its application to dye wastewater decolorization. In addition, the application of new technologies such as enzyme immobilization and genetic engineering that could improve the stability, durability, adaptability, and operating costs of the enzyme are highlighted. Finally, we discuss and propose future strategies to improve the performance of MnP-assisted dye decolorization in industrial applications. Keywords: manganese peroxidase; biodecolorization; dye wastewater; immobilization; recombi- nant enzyme Citation: Chang, Y.; Yang, D.; Li, R.; Wang, T.; Zhu, Y. Textile Dye Biodecolorization by Manganese Peroxidase: A Review. Molecules 2021, 26, 4403. https://doi.org/ 1. Introduction 10.3390/molecules26154403 The textile industry produces large quantities of wastewater containing different types of dyes used during the dyeing process, which cause great harm to the environment [1,2]. -

A Genomic Journey Through a Genus of Large DNA Viruses
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Virology Papers Virology, Nebraska Center for 2013 Towards defining the chloroviruses: a genomic journey through a genus of large DNA viruses Adrien Jeanniard Aix-Marseille Université David D. Dunigan University of Nebraska-Lincoln, [email protected] James Gurnon University of Nebraska-Lincoln, [email protected] Irina V. Agarkova University of Nebraska-Lincoln, [email protected] Ming Kang University of Nebraska-Lincoln, [email protected] See next page for additional authors Follow this and additional works at: https://digitalcommons.unl.edu/virologypub Part of the Biological Phenomena, Cell Phenomena, and Immunity Commons, Cell and Developmental Biology Commons, Genetics and Genomics Commons, Infectious Disease Commons, Medical Immunology Commons, Medical Pathology Commons, and the Virology Commons Jeanniard, Adrien; Dunigan, David D.; Gurnon, James; Agarkova, Irina V.; Kang, Ming; Vitek, Jason; Duncan, Garry; McClung, O William; Larsen, Megan; Claverie, Jean-Michel; Van Etten, James L.; and Blanc, Guillaume, "Towards defining the chloroviruses: a genomic journey through a genus of large DNA viruses" (2013). Virology Papers. 245. https://digitalcommons.unl.edu/virologypub/245 This Article is brought to you for free and open access by the Virology, Nebraska Center for at DigitalCommons@University of Nebraska - Lincoln. It has been accepted for inclusion in Virology Papers by an authorized administrator of DigitalCommons@University of Nebraska - Lincoln. Authors Adrien Jeanniard, David D. Dunigan, James Gurnon, Irina V. Agarkova, Ming Kang, Jason Vitek, Garry Duncan, O William McClung, Megan Larsen, Jean-Michel Claverie, James L. Van Etten, and Guillaume Blanc This article is available at DigitalCommons@University of Nebraska - Lincoln: https://digitalcommons.unl.edu/ virologypub/245 Jeanniard, Dunigan, Gurnon, Agarkova, Kang, Vitek, Duncan, McClung, Larsen, Claverie, Van Etten & Blanc in BMC Genomics (2013) 14. -

Lignocellulolytic Enzyme Production from Wood Rot Fungi Collected in Chiapas, Mexico, and Their Growth on Lignocellulosic Material
Journal of Fungi Article Lignocellulolytic Enzyme Production from Wood Rot Fungi Collected in Chiapas, Mexico, and Their Growth on Lignocellulosic Material Lina Dafne Sánchez-Corzo 1 , Peggy Elizabeth Álvarez-Gutiérrez 1 , Rocío Meza-Gordillo 1 , Juan José Villalobos-Maldonado 1, Sofía Enciso-Pinto 2 and Samuel Enciso-Sáenz 1,* 1 National Technological of Mexico-Technological Institute of Tuxtla Gutiérrez, Carretera Panamericana, km. 1080, Boulevares, C.P., Tuxtla Gutiérrez 29050, Mexico; [email protected] (L.D.S.-C.); [email protected] (P.E.Á.-G.); [email protected] (R.M.-G.); [email protected] (J.J.V.-M.) 2 Institute of Biomedical Research, National Autonomous University of Mexico, Circuito, Mario de La Cueva s/n, C.U., Coyoacán, México City 04510, Mexico; sofi[email protected] * Correspondence: [email protected]; Tel.: +52-96-150461 (ext. 304) Abstract: Wood-decay fungi are characterized by ligninolytic and hydrolytic enzymes that act through non-specific oxidation and hydrolytic reactions. The objective of this work was to evaluate the production of lignocellulolytic enzymes from collected fungi and to analyze their growth on lignocellulosic material. The study considered 18 species isolated from collections made in the state Citation: Sánchez-Corzo, L.D.; of Chiapas, Mexico, identified by taxonomic and molecular techniques, finding 11 different families. Álvarez-Gutiérrez, P.E.; The growth rates of each isolate were obtained in culture media with African palm husk (PH), coffee Meza-Gordillo, R.; husk (CH), pine sawdust (PS), and glucose as control, measuring daily growth with images analyzed Villalobos-Maldonado, J.J.; in ImageJ software, finding the highest growth rate in the CH medium. -

Bioaugmentation with Vaults: Novel in Situ Remediation Strategy for Transformation of Perfluoroalkyl Compounds
FINAL REPORT Bioaugmentation with Vaults: Novel In Situ Remediation Strategy for Transformation of Perfluoroalkyl Compounds SERDP Project ER-2422 JANUARY 2016 Shaily Mahendra, PhD Leonard H. Rome, PhD Valerie A. Kickhoefer, PhD Meng Wang, MS University of California, Los Angeles Distribution Statement A Page Intentionally Left Blank This report was prepared under contract to the Department of Defense Strategic Environmental Research and Development Program (SERDP). The publication of this report does not indicate endorsement by the Department of Defense, nor should the contents be construed as reflecting the official policy or position of the Department of Defense. Reference herein to any specific commercial product, process, or service by trade name, trademark, manufacturer, or otherwise, does not necessarily constitute or imply its endorsement, recommendation, or favoring by the Department of Defense. Page Intentionally Left Blank Form ApprowJd REPORTDOCUMENTATJONPAGE 0MB No.070UJ1BB The publicror repartlngbunlen 1h11 C0lec:liDIIlnf0rmat100 af Is estlmaled ID a-age1haurpr.-flllPIIIIN,lncludlng ror lhelime nlYillwlng lrwlructions, .-dq alstlng dalll -. gathering needed,and makltakmglhedata and camplrl&lgilformalbL andflMIIIWIIIthec:dledl0nof Send aimmenll '9g8l'dlng thisbunlenestlmaleo,anyat!.- asped rl llisi1bmallcx,, C0lllldi0ncf Including suggestiarmDepar".mr11d farl'llduc:lnglheburden,ID ot Defame,Semcm, W8stlinglDl'IHwlqmr1ln Dinlclaralll lnfannatian far Operltiara(0704-0188}, andRllparta 1215 JefferlonD11'111 Highway, Sulla 1204+ ArllnglDn.VA 22202-<1302.. Rmp011de,itastia.lld be awm9 thatnalwlltlaandlnganyDlhlll' provillan at law, 1111 pr,rsan lhall be IUbjr,c:ta ID8l'lfpenallyfarfallinglocarnplywllhacalecllar!afblfor11iiilkx,If dotil nat dilplay0MB acumtnllyvalid c:anlnll number. PLEASENOT DO RETURNYOUR FORMTO TIIE ABOVE ADDRESS, 1. REPORTDATE (DD-MM-YVVY) REPORTTYPE 3. DATES COVERED(From - To) 15/01/2016 12.Technical 20/08/2014 - 20/08/2016 4. -

Inhibition of Lignin Peroxidase-Mediated Oxidation Activity by Ethylenediamine Tetraacetic Acid and N-N-N’-N’-Tetramethylenediamine
Proc. Natl. Sci. Counc. ROC(B) H.C. Chang and J.A. Bumpus Vol. 25, No. 1, 2001. pp. 26-33 Inhibition of Lignin Peroxidase-Mediated Oxidation Activity by Ethylenediamine Tetraacetic Acid and N-N-N’-N’-Tetramethylenediamine HEBRON C. CHANG* AND JOHN A. BUMPUS** *Division of Biochemical Toxicology National Center for Toxicology Research Food and Drug Administration Jefferson, AR, U.S.A. **Department of Chemistry University of Northern Iowa Cedar Fall, IA, U.S.A (Received April 11, 2000; Accepted June 15, 2000) ABSTRACT The mineralization rate of 14C-[1,1,1-trichloro-2,2-bis(4-chlorophenyl)ethane] (DDT) was reduced by 90% on the 18th day in fungal cultures of Phanerochaete chrysosporium in the presence of 8 mM ethylenediamine tetraacetic acid (EDTA). In the presence of 8 mM N-N-N’-N’-tetramethylenediamine (TEMED), the mineralization rate of 14C-DDT was reduced by 80%. In the presence of 2 mM or 10 mM EDTA, 95% inhibition of lignin peroxidase (LiP) mediated veratryl alcohol oxidase activity and 97% inhibition of LiP mediated iodide oxidase activity occurred. TEMED caused 79% inhibition of veratryl alcohol oxidase activity and 92% inhibition of iodide oxidase activity when the amount used was 2 mM and 10 mM, respectively. In the presence of Zn(II) with slight molar excess of the EDTA concentration, reversed the EDTA mediated non-competitive inhibition of LiP catalyzed veratryl alcohol or iodide oxidation. Zn(II) also reversed the inhibition of LiP catalyzed veratryl alcohol oxidase activity caused by chelators other than EDTA and TEMED. In addition to Zn(II), several other metal ions also relieved EDTA mediated inhibition of veratryl alcohol and iodide oxidase activity catalyzed by LiP.