Changes in Mrna Expression of Arcuate Nucleus Appetite-Regulating Peptides During Lactation in Rats
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Multifaceted Physiological Roles of Adiponectin in Inflammation And
International Journal of Molecular Sciences Review Multifaceted Physiological Roles of Adiponectin in Inflammation and Diseases Hyung Muk Choi 1, Hari Madhuri Doss 1,2 and Kyoung Soo Kim 1,2,* 1 Department of Clinical Pharmacology and Therapeutics, Kyung Hee University School of Medicine, Seoul 02447, Korea; [email protected] (H.M.C.); [email protected] (H.M.D.) 2 East-West Bone & Joint Disease Research Institute, Kyung Hee University Hospital at Gangdong, Gandong-gu, Seoul 02447, Korea * Correspondence: [email protected]; Tel.: +82-2-961-9619 Received: 3 January 2020; Accepted: 10 February 2020; Published: 12 February 2020 Abstract: Adiponectin is the richest adipokine in human plasma, and it is mainly secreted from white adipose tissue. Adiponectin circulates in blood as high-molecular, middle-molecular, and low-molecular weight isoforms. Numerous studies have demonstrated its insulin-sensitizing, anti-atherogenic, and anti-inflammatory effects. Additionally, decreased serum levels of adiponectin is associated with chronic inflammation of metabolic disorders including Type 2 diabetes, obesity, and atherosclerosis. However, recent studies showed that adiponectin could have pro-inflammatory roles in patients with autoimmune diseases. In particular, its high serum level was positively associated with inflammation severity and pathological progression in rheumatoid arthritis, chronic kidney disease, and inflammatory bowel disease. Thus, adiponectin seems to have both pro-inflammatory and anti-inflammatory effects. This indirectly indicates that adiponectin has different physiological roles according to an isoform and effector tissue. Knowledge on the specific functions of isoforms would help develop potential anti-inflammatory therapeutics to target specific adiponectin isoforms against metabolic disorders and autoimmune diseases. -

Supporting Online Material
1 2 3 4 5 6 7 Supplementary Information for 8 9 Fractalkine-induced microglial vasoregulation occurs within the retina and is altered early in diabetic 10 retinopathy 11 12 *Samuel A. Mills, *Andrew I. Jobling, *Michael A. Dixon, Bang V. Bui, Kirstan A. Vessey, Joanna A. Phipps, 13 Ursula Greferath, Gene Venables, Vickie H.Y. Wong, Connie H.Y. Wong, Zheng He, Flora Hui, James C. 14 Young, Josh Tonc, Elena Ivanova, Botir T. Sagdullaev, Erica L. Fletcher 15 * Joint first authors 16 17 Corresponding author: 18 Prof. Erica L. Fletcher. Department of Anatomy & Neuroscience. The University of Melbourne, Grattan St, 19 Parkville 3010, Victoria, Australia. 20 Email: [email protected] ; Tel: +61-3-8344-3218; Fax: +61-3-9347-5219 21 22 This PDF file includes: 23 24 Supplementary text 25 Figures S1 to S10 26 Tables S1 to S7 27 Legends for Movies S1 to S2 28 SI References 29 30 Other supplementary materials for this manuscript include the following: 31 32 Movies S1 to S2 33 34 35 36 1 1 Supplementary Information Text 2 Materials and Methods 3 Microglial process movement on retinal vessels 4 Dark agouti rats were anaesthetized, injected intraperitoneally with rhodamine B (Sigma-Aldrich) to label blood 5 vessels and retinal explants established as described in the main text. Retinal microglia were labelled with Iba-1 6 and imaging performed on an inverted confocal microscope (Leica SP5). Baseline images were taken for 10 7 minutes, followed by the addition of PBS (10 minutes) and then either fractalkine or fractalkine + candesartan 8 (10 minutes) using concentrations outlined in the main text. -

Plasma Omentin Levels Are Inversely Associated with Atherosclerosis In
Nishimura et al. Cardiovasc Diabetol (2019) 18:167 https://doi.org/10.1186/s12933-019-0973-3 Cardiovascular Diabetology ORIGINAL INVESTIGATION Open Access Plasma omentin levels are inversely associated with atherosclerosis in type 2 diabetes patients with increased plasma adiponectin levels: a cross-sectional study Masami Nishimura1, Tomoaki Morioka1* , Mariko Hayashi1, Yoshinori Kakutani1, Yuko Yamazaki1, Masafumi Kurajoh1, Katsuhito Mori2, Shinya Fukumoto3, Atsushi Shioi4,5, Tetsuo Shoji4,5, Masaaki Inaba1,5 and Masanori Emoto1 Abstract Background: Omentin and adiponectin are among the anti-infammatory and anti-atherogenic adipokines that have potentially benefcial efects on cardiovascular disorders. Recent studies indicate a paradoxical relationship between adiponectin and cardiovascular mortality across many clinical settings including type 2 diabetes. In this study, we characterized the clinical features of type 2 diabetes patients with increased adiponectin levels and exam- ined the association between omentin and atherosclerosis in those patients. Methods: The subjects were 413 patients with type 2 diabetes. Fasting plasma omentin and total adiponectin levels were measured by enzyme-linked immunosorbent assay. The intima-media thickness (IMT) of the common carotid artery was measured by ultrasonography. The subjects were stratifed according to the median value of plasma adiponectin. Results: In high-adiponectin group, omentin levels were higher, while IMT tended to be greater than those in low- adiponectin group. The high-adiponectin group also exhibited older age, higher systolic blood pressure, lower kidney function, body mass index, and insulin resistance index compared to the low-adiponectin group. Multivariate analysis revealed that omentin levels were independently and negatively associated with IMT in high-adiponectin group, but not in low-adiponectin group, after adjusting for adiponectin levels and traditional cardiovascular risk factors. -

(Title of the Thesis)*
THE PHYSIOLOGICAL ACTIONS OF ADIPONECTIN IN CENTRAL AUTONOMIC NUCLEI: IMPLICATIONS FOR THE INTEGRATIVE CONTROL OF ENERGY HOMEOSTASIS by Ted Donald Hoyda A thesis submitted to the Department of Physiology In conformity with the requirements for the degree of Doctor of Philosophy Queen‟s University Kingston, Ontario, Canada (September, 2009) Copyright © Ted Donald Hoyda, 2009 ABSTRACT Adiponectin regulates feeding behavior, energy expenditure and autonomic function through the activation of two receptors present in nuclei throughout the central nervous system, however much remains unknown about the mechanisms mediating these effects. Here I investigate the actions of adiponectin in autonomic centers of the hypothalamus (the paraventricular nucleus) and brainstem (the nucleus of the solitary tract) through examining molecular, electrical, hormonal and physiological consequences of peptidergic signalling. RT-PCR and in situ hybridization experiments demonstrate the presence of AdipoR1 and AdipoR2 mRNA in the paraventricular nucleus. Investigation of the electrical consequences following receptor activation in the paraventricular nucleus indicates that magnocellular-oxytocin cells are homogeneously inhibited while magnocellular-vasopressin neurons display mixed responses. Single cell RT-PCR analysis shows oxytocin neurons express both receptors while vasopressin neurons express either both receptors or one receptor. Co-expressing oxytocin and vasopressin neurons express neither receptor and are not affected by adiponectin. Median eminence projecting corticotropin releasing hormone neurons, brainstem projecting oxytocin neurons, and thyrotropin releasing hormone neurons are all depolarized by adiponectin. Plasma adrenocorticotropin hormone concentration is increased following intracerebroventricular injections of adiponectin. I demonstrate that the nucleus of the solitary tract, the primary cardiovascular regulation site of the medulla, expresses mRNA for AdipoR1 and AdipoR2 and mediates adiponectin induced hypotension. -

Feedback Regulation of Growth Hormone Synthesis and Secretion in Fish and the Emerging Concept of Intrapituitary Feedback Loop ☆ ⁎ Anderson O.L
http://www.paper.edu.cn Comparative Biochemistry and Physiology, Part A 144 (2006) 284–305 Review Feedback regulation of growth hormone synthesis and secretion in fish and the emerging concept of intrapituitary feedback loop ☆ ⁎ Anderson O.L. Wong , Hong Zhou, Yonghua Jiang, Wendy K.W. Ko Department of Zoology, University of Hong Kong, Pokfulam Road, Hong Kong, P.R. China Received 29 July 2005; received in revised form 21 November 2005; accepted 21 November 2005 Available online 9 January 2006 Abstract Growth hormone (GH) is known to play a key role in the regulation of body growth and metabolism. Similar to mammals, GH secretion in fish is under the control of hypothalamic factors. Besides, signals generated within the pituitary and/or from peripheral tissues/organs can also exert a feedback control on GH release by effects acting on both the hypothalamus and/or anterior pituitary. Among these feedback signals, the functional role of IGF is well conserved from fish to mammals. In contrast, the effects of steroids and thyroid hormones are more variable and appear to be species-specific. Recently, a novel intrapituitary feedback loop regulating GH release and GH gene expression has been identified in fish. This feedback loop has three functional components: (i) LH induction of GH release from somatotrophs, (ii) amplification of GH secretion by GH autoregulation in somatotrophs, and (iii) GH feedback inhibition of LH release from neighboring gonadotrophs. In this article, the mechanisms for feedback control of GH synthesis and secretion are reviewed and functional implications of this local feedback loop are discussed. This intrapituitary feedback loop may represent a new facet of pituitary research with potential applications in aquaculture and clinical studies. -

NIH Public Access Author Manuscript Rev Endocr Metab Disord
NIH Public Access Author Manuscript Rev Endocr Metab Disord. Author manuscript; available in PMC 2014 September 03. NIH-PA Author ManuscriptPublished NIH-PA Author Manuscript in final edited NIH-PA Author Manuscript form as: Rev Endocr Metab Disord. 2014 June ; 15(2): 137–147. doi:10.1007/s11154-013-9280-6. ADIPONECTIN SIGNALING IN THE LIVER Terry P. Combs1,2,3 and Errol B. Marliss2 1Department of Medicine, University of North Carolina at Chapel Hill 2Crabtree Nutrition Laboratories, Department of Medicine, McGill University Abstract High glucose production contributes to fed and fasted hyperglycemia in Type 1 Diabetes (T1D) and Type 2 Diabetes (T2D). The breakdown of the adiponectin signaling pathway in T1D and the reduction of circulating adiponectin in T2D contribute to this abnormal increase in glucose production. Sufficient amounts of insulin could compensate for the loss of adiponectin signaling in T1D and T2D and reduce hyperglycemia. However, the combination of low adiponectin signaling and high insulin resembles an insulin resistance state associated with cardiovascular disease and decreased life expectancy. Future development of medications that correct the deficiency of adiponectin signaling in the liver could restore the metabolic balance in T1D and T2D and reduce the need for insulin. This article reviews the adiponectin signaling pathway in the liver through T- cadherin, AdipoR1, AdipoR2, AMPK, ceramidase activity, APPL1 and the recently discovered Suppressor Of Glucose from Autophagy (SOGA). 1. Liver contribution to circulating glucose The liver releases glucose ensuring sufficient amounts of this essential fuel are always available in the circulation for other tissues. Endocrine and neural mechanisms stimulate the liver during fasting, exercise and pregnancy to meet the increased demand for glucose. -

Metabolic Syndrome: Is It Time to Add the Central Nervous System?
nutrients Review Metabolic Syndrome: Is It Time to Add the Central Nervous System? Milagros Rojas 1, Mervin Chávez-Castillo 2, Daniela Pirela 1, Heliana Parra 1 , Manuel Nava 1, Maricarmen Chacín 3, Lissé Angarita 4 , Roberto Añez 5, Juan Salazar 1 , Rina Ortiz 6, Samuel Durán Agüero 7, Marbel Gravini-Donado 8, Valmore Bermúdez 9 and Edgar Díaz-Camargo 9,* 1 Endocrine and Metabolic Diseases Research Center, School of Medicine, University of Zulia, Maracaibo 4004, Venezuela; migarocafi@gmail.com (M.R.); [email protected] (D.P.); [email protected] (H.P.); [email protected] (M.N.); [email protected] (J.S.) 2 Psychiatric Hospital of Maracaibo, Maracaibo 4004, Venezuela; [email protected] 3 Facultad de Ciencias de la Salud, Universidad Simón Bolívar, Barranquilla 08002, Colombia; [email protected] 4 Escuela de Nutrición y Dietética, Facultad de Medicina, Universidad Andrés Bello, Sede Concepción 4260000, Chile; [email protected] 5 Departamento de Endocrinología y Nutrición, Hospital General Universitario Gregorio Marañón, 28007 Madrid, Spain; [email protected] 6 Posgrado, Carrera de Medicina, Universidad Católica de Cuenca, Cantón de Cuenca 010101, Ecuador; [email protected] 7 Facultad de Ciencias Para el Cuidado de la Salud, Universidad San Sebastián, Los Leones 8420524, Chile; [email protected] 8 Facultad de Ciencias Jurídicas y Sociales, Universidad Simón Bolívar, Barranquilla 080002, Colombia; [email protected] 9 Facultad de Ciencias Jurídicas y Sociales, Universidad Simón Bolívar, Cúcuta 540006, Colombia; [email protected] * Correspondence: [email protected] Citation: Rojas, M.; Chávez-Castillo, M.; Pirela, D.; Parra, H.; Nava, M.; Abstract: Metabolic syndrome (MS) is a set of cardio-metabolic risk factors that includes central Chacín, M.; Angarita, L.; Añez, R.; obesity, hyperglycemia, hypertension, and dyslipidemias. -

ADIPOQ) and the Type 1 Receptor (ADIPOR1), Obesity and Prostate Cancer in African Americans
Prostate Cancer and Prostatic Diseases (2010) 13, 362–368 & 2010 Macmillan Publishers Limited All rights reserved 1365-7852/10 www.nature.com/pcan ORIGINAL ARTICLE Genetic variation in adiponectin (ADIPOQ) and the type 1 receptor (ADIPOR1), obesity and prostate cancer in African Americans JL Beebe-Dimmer1, KA Zuhlke2, AM Ray2, EM Lange3 and KA Cooney4 1Department of Population Studies and Prevention, Karmanos Cancer Institute, Department of Internal Medicine, Wayne State University, Detroit, MI, USA; 2Departments of Internal Medicine and Urology, University of Michigan Medical School, Ann Arbor, MI, USA; 3Departments of Genetics and Biostatistics, University of North Carolina, Chapel Hill, NC, USA and 4Departments of Internal Medicine and Urology, University of Michigan Medical School, University of Michigan Comprehensive Cancer Center, Ann Arbor, MI, USA Adiponectin is a protein derived from adipose tissue suspected to have an important role in prostate carcinogenesis. Variants in the adiponectin gene (ADIPOQ) and its type 1 receptor (ADIPOR1) have been recently linked to risk of both breast and colorectal cancer. Therefore, we set out to examine the relationship between polymorphisms in these genes, obesity and prostate cancer in study of African-American men. Ten single-nucleotide polymorphisms (SNPs) in ADIPOQ and ADIPOR1 were genotyped in DNA samples from 131 African-American prostate cancer cases and 344 controls participating in the Flint Men’s Health Study. Logistic regression was then used to estimate their association with prostate cancer and obesity. While no significant associations were detected between any of the tested SNPs and prostate cancer, the rs1501299 SNP in ADIPOQ was significantly associated with body mass (P ¼ 0.03). -
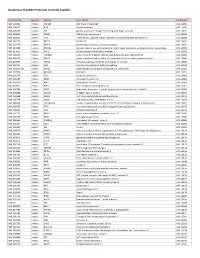
Quantigene Flowrna Probe Sets Currently Available
QuantiGene FlowRNA Probe Sets Currently Available Accession No. Species Symbol Gene Name Catalog No. NM_003452 Human ZNF189 zinc finger protein 189 VA1-10009 NM_000057 Human BLM Bloom syndrome VA1-10010 NM_005269 Human GLI glioma-associated oncogene homolog (zinc finger protein) VA1-10011 NM_002614 Human PDZK1 PDZ domain containing 1 VA1-10015 NM_003225 Human TFF1 Trefoil factor 1 (breast cancer, estrogen-inducible sequence expressed in) VA1-10016 NM_002276 Human KRT19 keratin 19 VA1-10022 NM_002659 Human PLAUR plasminogen activator, urokinase receptor VA1-10025 NM_017669 Human ERCC6L excision repair cross-complementing rodent repair deficiency, complementation group 6-like VA1-10029 NM_017699 Human SIDT1 SID1 transmembrane family, member 1 VA1-10032 NM_000077 Human CDKN2A cyclin-dependent kinase inhibitor 2A (melanoma, p16, inhibits CDK4) VA1-10040 NM_003150 Human STAT3 signal transducer and activator of transcripton 3 (acute-phase response factor) VA1-10046 NM_004707 Human ATG12 ATG12 autophagy related 12 homolog (S. cerevisiae) VA1-10047 NM_000737 Human CGB chorionic gonadotropin, beta polypeptide VA1-10048 NM_001017420 Human ESCO2 establishment of cohesion 1 homolog 2 (S. cerevisiae) VA1-10050 NM_197978 Human HEMGN hemogen VA1-10051 NM_001738 Human CA1 Carbonic anhydrase I VA1-10052 NM_000184 Human HBG2 Hemoglobin, gamma G VA1-10053 NM_005330 Human HBE1 Hemoglobin, epsilon 1 VA1-10054 NR_003367 Human PVT1 Pvt1 oncogene homolog (mouse) VA1-10061 NM_000454 Human SOD1 Superoxide dismutase 1, soluble (amyotrophic lateral sclerosis 1 (adult)) -

Supplementary Table S4. FGA Co-Expressed Gene List in LUAD
Supplementary Table S4. FGA co-expressed gene list in LUAD tumors Symbol R Locus Description FGG 0.919 4q28 fibrinogen gamma chain FGL1 0.635 8p22 fibrinogen-like 1 SLC7A2 0.536 8p22 solute carrier family 7 (cationic amino acid transporter, y+ system), member 2 DUSP4 0.521 8p12-p11 dual specificity phosphatase 4 HAL 0.51 12q22-q24.1histidine ammonia-lyase PDE4D 0.499 5q12 phosphodiesterase 4D, cAMP-specific FURIN 0.497 15q26.1 furin (paired basic amino acid cleaving enzyme) CPS1 0.49 2q35 carbamoyl-phosphate synthase 1, mitochondrial TESC 0.478 12q24.22 tescalcin INHA 0.465 2q35 inhibin, alpha S100P 0.461 4p16 S100 calcium binding protein P VPS37A 0.447 8p22 vacuolar protein sorting 37 homolog A (S. cerevisiae) SLC16A14 0.447 2q36.3 solute carrier family 16, member 14 PPARGC1A 0.443 4p15.1 peroxisome proliferator-activated receptor gamma, coactivator 1 alpha SIK1 0.435 21q22.3 salt-inducible kinase 1 IRS2 0.434 13q34 insulin receptor substrate 2 RND1 0.433 12q12 Rho family GTPase 1 HGD 0.433 3q13.33 homogentisate 1,2-dioxygenase PTP4A1 0.432 6q12 protein tyrosine phosphatase type IVA, member 1 C8orf4 0.428 8p11.2 chromosome 8 open reading frame 4 DDC 0.427 7p12.2 dopa decarboxylase (aromatic L-amino acid decarboxylase) TACC2 0.427 10q26 transforming, acidic coiled-coil containing protein 2 MUC13 0.422 3q21.2 mucin 13, cell surface associated C5 0.412 9q33-q34 complement component 5 NR4A2 0.412 2q22-q23 nuclear receptor subfamily 4, group A, member 2 EYS 0.411 6q12 eyes shut homolog (Drosophila) GPX2 0.406 14q24.1 glutathione peroxidase -

Supplementary Table 1. Clinico-Pathologic Features of Patients with Pancreatic Neuroendocrine Tumors Associated with Cushing’S Syndrome
Supplementary Table 1. Clinico-pathologic features of patients with pancreatic neuroendocrine tumors associated with Cushing’s syndrome. Review of the English/Spanish literature. Other hormones Age of Cortisol Other hormones Other ACTH Size (ICH or assay) ENETS Follow- Time Year Author Sex CS onset level (Blood syndromes or Site MET Type AH level (cm) corticotrophic Stage up (months) (years) (µg/dl) detection) NF other differentiation 1 1 1946 Crooke F 28 uk uk none no 4.5 H liver/peritoneum NET uk uk yes IV DOD 6 2 1956 Rosenberg 2 F 40 uk 92 none no 10 B liver NET uk uk yes IV PD 3 & p 3 1959 Balls F 36 uk elevated insulin Insulinoma 20 B liver/LNl/lung NET uk uk yes IV DOD 1 4 & 4 1962 Meador F 47 13~ elevated none no uk T liver/spleen NET ACTH uk yes IV uk 5 1963 Liddle5 uk uk uk elevated uk uk uk uk uk NET ACTH uk uk uk uk 6 1964 Hallwright6 F 32 uk uk none no large H LN/liver NET ACTH, MSH uk yes IV DOD 24 7 1965 Marks7 M 43 uk elevated insulin insulinomas 1.8 T liver NET uk uk yes IV DOD 7 8 1965 Sayle8 F 62 uk uk none carcinoidb 4 H LN/liver NET uk uk yes IV DOD 6 9 1965 Sayle8 F 15 uk uk none no 4 T no NET ACTH uk yes IIa DOD 5 9 b mesentery/ 10 1965 Geokas M 59 uk uk gastrin ZES large T NET uk uk yes IV DOD 2 LN/pleural/bone 11 1965 Law10 F 35 1.2 ∞ 91 gastrin ZESs uk B LN/liver NET ACTH, MSH gastrin yes IV PD 12 1967 Burkinshaw11 M 2 uk 72 none no large T no NET uk uk no uk AFD 12 13 1968 Uei12 F 9 uk uk none ZESs 7 T liver NET ACTH uk yes IV DOD 8 13 PTH, ADH, b gastrin, 14 1968 O'Neal F 52 13~ uk ZES uk T LN/liver/bone NET -

Suppression of Adiponectin Receptor 1 Promotes Memory Dysfunction And
www.nature.com/scientificreports OPEN Suppression of adiponectin receptor 1 promotes memory dysfunction and Alzheimer’s Received: 23 May 2017 Accepted: 8 September 2017 disease-like pathologies Published: xx xx xxxx Min Woo Kim, Noman bin Abid, Myeong Hoon Jo, Min Gi Jo, Gwang Ho Yoon & Myeong Ok Kim Recent studies on neurodegeneration have focused on dysfunction of CNS energy metabolism as well as proteinopathies. Adiponectin (ADPN), an adipocyte-derived hormone, plays a major role in the regulation of insulin sensitivity and glucose homeostasis in peripheral organs via adiponectin receptors. In spite of accumulating evidence that adiponectin has neuroprotective properties, the underlying role of adiponectin receptors has not been illuminated. Here, using gene therapy-mediated suppression with shRNA, we found that adiponectin receptor 1 (AdipoR1) suppression induces neurodegeneration as well as metabolic dysfunction. AdipoR1 knockdown mice exhibited increased body weight and abnormal plasma chemistry and also showed spatial learning and memory impairment in behavioural studies. Moreover, AdipoR1 suppression resulted in neurodegenerative phenotypes, diminished expression of the neuronal marker NeuN, and increased expression and activity of caspase 3. Furthermore, AD-like pathologies including insulin signalling dysfunction, abnormal protein aggregation and neuroinfammatory responses were highly exhibited in AdipoR1 knockdown groups, consistent with brain pathologies in ADPN knockout mice. Together, these results suggest that ADPN- AdipoR1 signalling has the potential to alleviate neurodegenerative diseases such as Alzheimer’s diseases. Neurodegeneration is a term describing a pathological phenotype observed in the central nervous system, espe- cially the brain1. Many etiological models of neurodegeneration, such as that in Alzheimer’s disease (AD) and Parkinson’s disease (PD), are based on abnormal protein aggregation and sequentially entail chronic infamma- tion, generation of reactive oxygen species (ROS) and apoptosis2–4.