WHO Model List of Essential Medicines
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

National Antibiotic Consumption for Human Use in Sierra Leone (2017–2019): a Cross-Sectional Study
Tropical Medicine and Infectious Disease Article National Antibiotic Consumption for Human Use in Sierra Leone (2017–2019): A Cross-Sectional Study Joseph Sam Kanu 1,2,* , Mohammed Khogali 3, Katrina Hann 4 , Wenjing Tao 5, Shuwary Barlatt 6,7, James Komeh 6, Joy Johnson 6, Mohamed Sesay 6, Mohamed Alex Vandi 8, Hannock Tweya 9, Collins Timire 10, Onome Thomas Abiri 6,11 , Fawzi Thomas 6, Ahmed Sankoh-Hughes 12, Bailah Molleh 4, Anna Maruta 13 and Anthony D. Harries 10,14 1 National Disease Surveillance Programme, Sierra Leone National Public Health Emergency Operations Centre, Ministry of Health and Sanitation, Cockerill, Wilkinson Road, Freetown, Sierra Leone 2 Department of Community Health, Faculty of Clinical Sciences, College of Medicine and Allied Health Sciences, University of Sierra Leone, Freetown, Sierra Leone 3 Special Programme for Research and Training in Tropical Diseases (TDR), World Health Organization, 1211 Geneva, Switzerland; [email protected] 4 Sustainable Health Systems, Freetown, Sierra Leone; [email protected] (K.H.); [email protected] (B.M.) 5 Unit for Antibiotics and Infection Control, Public Health Agency of Sweden, Folkhalsomyndigheten, SE-171 82 Stockholm, Sweden; [email protected] 6 Pharmacy Board of Sierra Leone, Central Medical Stores, New England Ville, Freetown, Sierra Leone; [email protected] (S.B.); [email protected] (J.K.); [email protected] (J.J.); [email protected] (M.S.); [email protected] (O.T.A.); [email protected] (F.T.) Citation: Kanu, J.S.; Khogali, M.; 7 Department of Pharmaceutics and Clinical Pharmacy & Therapeutics, Faculty of Pharmaceutical Sciences, Hann, K.; Tao, W.; Barlatt, S.; Komeh, College of Medicine and Allied Health Sciences, University of Sierra Leone, Freetown 0000, Sierra Leone 8 J.; Johnson, J.; Sesay, M.; Vandi, M.A.; Directorate of Health Security & Emergencies, Ministry of Health and Sanitation, Sierra Leone National Tweya, H.; et al. -

AHFS Pharmacologic-Therapeutic Classification System
AHFS Pharmacologic-Therapeutic Classification System Abacavir 48:24 - Mucolytic Agents - 382638 8:18.08.20 - HIV Nucleoside and Nucleotide Reverse Acitretin 84:92 - Skin and Mucous Membrane Agents, Abaloparatide 68:24.08 - Parathyroid Agents - 317036 Aclidinium Abatacept 12:08.08 - Antimuscarinics/Antispasmodics - 313022 92:36 - Disease-modifying Antirheumatic Drugs - Acrivastine 92:20 - Immunomodulatory Agents - 306003 4:08 - Second Generation Antihistamines - 394040 Abciximab 48:04.08 - Second Generation Antihistamines - 394040 20:12.18 - Platelet-aggregation Inhibitors - 395014 Acyclovir Abemaciclib 8:18.32 - Nucleosides and Nucleotides - 381045 10:00 - Antineoplastic Agents - 317058 84:04.06 - Antivirals - 381036 Abiraterone Adalimumab; -adaz 10:00 - Antineoplastic Agents - 311027 92:36 - Disease-modifying Antirheumatic Drugs - AbobotulinumtoxinA 56:92 - GI Drugs, Miscellaneous - 302046 92:20 - Immunomodulatory Agents - 302046 92:92 - Other Miscellaneous Therapeutic Agents - 12:20.92 - Skeletal Muscle Relaxants, Miscellaneous - Adapalene 84:92 - Skin and Mucous Membrane Agents, Acalabrutinib 10:00 - Antineoplastic Agents - 317059 Adefovir Acamprosate 8:18.32 - Nucleosides and Nucleotides - 302036 28:92 - Central Nervous System Agents, Adenosine 24:04.04.24 - Class IV Antiarrhythmics - 304010 Acarbose Adenovirus Vaccine Live Oral 68:20.02 - alpha-Glucosidase Inhibitors - 396015 80:12 - Vaccines - 315016 Acebutolol Ado-Trastuzumab 24:24 - beta-Adrenergic Blocking Agents - 387003 10:00 - Antineoplastic Agents - 313041 12:16.08.08 - Selective -

Determination of Iodate in Iodised Salt by Redox Titration
College of Science Determination of Iodate in Iodised Salt by Redox Titration Safety • 0.6 M potassium iodide solution (10 g solid KI made up to 100 mL with distilled water) • 0.5% starch indicator solution Lab coats, safety glasses and enclosed footwear must (see below for preparation) be worn at all times in the laboratory. • 250 mL volumetric flask Introduction • 50 mL pipette (or 20 and 10 mL pipettes) • 250 mL conical flasks New Zealand soil is low in iodine and hence New Zealand food is low in iodine. Until iodised salt was • 10 mL measuring cylinder commonly used (starting in 1924), a large proportion • burette and stand of school children were reported as being affected • distilled water by iodine deficiency – as high as 60% in Canterbury schools, and averaging 20 − 40% overall. In the worst cases this deficiency can lead to disorders such as Method goitre, and impaired physical and mental development. 1. Preparation of 0.002 mol L−1 sodium thiosulfate In earlier times salt was “iodised” by the addition of solution: Accurately weigh about 2.5 g of solid potassium iodide; however, nowadays iodine is more sodium thiosulfate (NaS2O3•5H2O) and dissolve in commonly added in the form of potassium iodate 100 mL of distilled water in a volumetric flask. (This gives a 0.1 mol L−1 solution). Then use a pipette to (KIO3). The Australia New Zealand Food Standards Code specifies that iodised salt must contain: “equivalent to transfer 10 mL of this solution to a 500 mL volumetric no less than 25 mg/kg of iodine; and no more than 65 flask and dilute by adding distilled water up to the mg/kg of iodine”. -

WHO Model List of Essential Medicines
WHO Model List of Essential Medicines 15th list, March 2007 Status of this document This is a reprint of the text on the WHO Medicines web site http://www.who.int/medicines/publications/essentialmedicines/en/index.html 15th edition Essential Medicines WHO Model List (revised March 2007) Explanatory Notes The core list presents a list of minimum medicine needs for a basic health care system, listing the most efficacious, safe and cost‐effective medicines for priority conditions. Priority conditions are selected on the basis of current and estimated future public health relevance, and potential for safe and cost‐effective treatment. The complementary list presents essential medicines for priority diseases, for which specialized diagnostic or monitoring facilities, and/or specialist medical care, and/or specialist training are needed. In case of doubt medicines may also be listed as complementary on the basis of consistent higher costs or less attractive cost‐effectiveness in a variety of settings. The square box symbol () is primarily intended to indicate similar clinical performance within a pharmacological class. The listed medicine should be the example of the class for which there is the best evidence for effectiveness and safety. In some cases, this may be the first medicine that is licensed for marketing; in other instances, subsequently licensed compounds may be safer or more effective. Where there is no difference in terms of efficacy and safety data, the listed medicine should be the one that is generally available at the lowest price, based on international drug price information sources. Therapeutic equivalence is only indicated on the basis of reviews of efficacy and safety and when consistent with WHO clinical guidelines. -

United States Patent Office Patented Apr
3,505,222 United States Patent Office Patented Apr. 7, 1970 1. 2 3,505,222 product of a mercaptain with sulfur trioxide. Their metal LUBRICANT COMPOSITIONS salts are represented by the formula: Leonard M. Niebylski, Birmingham, Mich, assignor to O Ethyl Corporation, New York, N.Y., a corporation of Virginia (R-S-S-0--M No Drawing. Filed Mar. 29, 1967, Ser. No. 626,701 5 s (I) Int. C. C10m 5/14, 3/18, 7/36 wherein R is a hydrocarbon radical containing from 1 U.S. C. 252-17 2 Claims to about 30 carbon atoms, M is a metal, and n is the valence of metal M. For example, when M is the monova 0. lent sodium ion, n is 1. ABSTRACT OF THE DISCLOSURE The radical R can be an alkyl, cycloalkyl, aralkyl, The extreme pressure wear properties of base lubri alkaryl, or aryl radical. The radicals may contain other cants including water, hydrocarbons, polyesters, silicones, nonhydrocarbon substituents such as chloro, bromo, iodo, polyethers and halocarbons is enhanced by the addition fluoro, nitro, hydroxyl, nitrile, isocyanate, carboxyl, car of a synergistic mixture of a thiosulfate compound and 15 bonyl, and the like. a lead compound. The useful metals are all those capable of forming Bunte salts. Preferred metals are those previously listed as suitable for forming metal thiosulfates. Of these, the Background more preferred metals are sodium and lead, and lead is 20 the most preferred metal in the Bunte salts. This invention relates to improved lubricant composi Examples of useful Bunte salts include: tions. -

Sodium Nitrite and Sodium Thiosulfate
PATIENT & CAREGIVER EDUCATION Sodium Nitrite and Sodium Thiosulfate This information from Lexicomp® explains what you need to know about this medication, including what it’s used for, how to take it, its side effects, and when to call your healthcare provider. Brand Names: US Nithiodote Warning This drug may cause low blood pressure and a red blood cell problem called methemoglobinemia. These may be life-threatening. This drug is only for use when cyanide poisoning is life-threatening. This drug must be used with care if it is not known if cyanide poisoning has happened. Talk with the doctor. Tell the doctor if your child has inhaled a lot of smoke or if your child has any of these health problems: Anemia, heart problems, lack of a certain enzyme called congenital methemoglobin reductase deficiency, or lung problems. What is this drug used for? It is used to treat cyanide poisoning. What do I need to tell the doctor BEFORE my child takes this drug? If your child is allergic to this drug; any part of this drug; or any other drugs, foods, or substances. Tell the doctor about the allergy and what signs your child had. Sodium Nitrite and Sodium Thiosulfate 1/6 If your child is breast-feeding a baby: Be sure your child does not breast-feed a baby while taking this drug. This drug may interact with other drugs or health problems. Tell the doctor and pharmacist about all of your child’s drugs (prescription or OTC, natural products, vitamins) and health problems. You must check to make sure that it is safe to give this drug with all of your child’s other drugs and health problems. -

Managing Medicine Selection 17 Treatment Guidelines and Formulary Manuals Procurement Distribution Use
Part I: Policy and economic issues Part II: Pharmaceutical management Part III: Management support systems Selection 16 Managing medicine selection 17 Treatment guidelines and formulary manuals Procurement Distribution Use chapter 16 Managing medicine selection Summary 16.2 illustrations 16.1 Introduction 16.2 Figure 16-1 The essential medicines target 16.7 Figure 16-2 Common health problems guide selection, training, 16.2 Practical implications of the essential medicines supply, and medicine use 16.9 concept 16.2 Figure 16-3 Sample form for proposing revisions in essential 16.3 Selection criteria 16.4 medicines lists 16.12 16.4 Use of International Nonproprietary (generic) Table 16-1 Advantages of a limited list of essential Names 16.5 medicines 16.4 Table 16-2 Example of level-of-use categories, Ethiopia 16.5 Essential medicines lists in context 16.6 Essential Drugs List, fifth edition, 2007 16.6 Lists of registered medicines • Formulary manuals • Table 16-3 Key factors in successfully developing Treatment guidelines and implementing an essential medicines 16.6 Approaches to developing essential medicines lists, program 16.14 formularies, and treatment guidelines 16.8 box 16.7 Therapeutic classification systems 16.10 Box 16-1 WHO criteria for selection of essential 16.8 Sources of information 16.13 medicines 16.5 16.9 Implementing and updating essential medicines lists 16.14 country studies Reasons for failure • Gaining acceptance of essential CS 16-1 Approaches to updating essential medicines and medicines lists • Authority of essential medicines lists formulary lists 16.10 Assessment guide 16.15 CS 16-2 Updating the National Essential Medicines List of Kenya 16.11 References and further readings 16.15 Glossary 16.16 copyright © management sciences for health 2012 16.2 selection SUMMARY The rationale for selecting a limited number of essen- The process of selecting essential medicines begins with tial medicines is that it may lead to better supply, more defining a list of common diseases for each level of health rational use, and lower costs. -

Assessing the in Situ Efficacy of Tea Tree Oil As a Topical Antiseptic
Novasel Australia Pty Ltd Assessing the in situ efficacy of tea tree oil as a topical antiseptic A report for the Rural Industries Research and Development Corporation by S. Messager, K.A. Hammer & T.V. Riley August 2005 RIRDC Publication No 05/113 RIRDC Project No UWA-72A © 2005 Rural Industries Research and Development Corporation. All rights reserved. ISBN 1 74151 176 3 ISSN 1440-6845 Assessing the in situ efficacy of tea tree oil as a topical antiseptic Publication No. 05/113 Project No. UWA-72A The information contained in this publication is intended for general use to assist public knowledge and discussion and to help improve the development of sustainable industries. The information should not be relied upon for the purpose of a particular matter. Specialist and/or appropriate legal advice should be obtained before any action or decision is taken on the basis of any material in this document. The Commonwealth of Australia, Rural Industries Research and Development Corporation, the authors or contributors do not assume liability of any kind whatsoever resulting from any person's use or reliance upon the content of this document. This publication is copyright. However, RIRDC encourages wide dissemination of its research, providing the Corporation is clearly acknowledged. For any other enquiries concerning reproduction, contact the Publications Manager on phone 02 6272 3186. Researcher Contact Details Prof. T. V. Riley University of Western Australia School of Biomedical and Chemical Sciences Microbiology (M502) 35 Stirling Hwy CRAWLEY WA 6009 Phone: (08) 9346 3690 Fax: (08) 9346 2912 Email: [email protected] In submitting this report, the researcher has agreed to RIRDC publishing this material in its edited form. -

Chemical %Concentration CAS
Sebozole Shampoo Page 1of 5 Sebozole Shampoo SAFETY DATA SHEET (1) IDENTIFICATION Name: Sebozole Shampoo Use: veterinary skin care Supplier: Vetoquinol USA (Tomlyn Products) 4250 N. Sylvania Ave Fort Worth, TX 76137 Tel: ( 817)529-7500 Fax: (817)529-7506 (2) HAZARD(S) IDENTIFICATION Hazard Classification: Non Hazardous as defined by 29CFR Part 1910.1200 (3) COMPOSITION/INFORMATION ON INGREDIENTS Common Name: Sebozole Shampoo Composition: A blend of proprietary ingredients which make a veterinary dermatologic care product. Chemical %Concentration CAS Sodium Thiosulfate 3.13 10102-17-7 Miconazole Nitrate 2.0 22916-47-8 Salicylic Acid 2.0 69-72-7 Chloroxylenol 1.0 88-04-0 Sodium Olefin Sulfonate 25 6843-57-6 PEG 150 Pentaerythrityl Tetratstearate 5 130249-48-8 Lauramide DEA 5 92680-75-6 Propylene Glycol 5 57-55-6 Ultrez 21 1 9003-39-8 Sodium Hydroxide 1 1310-73-2 Fragrance 0.5 Kathon CG 0.1 6118-96-6 FD&C yellow #5 0.0013 84842-94-4 FD&C Blue #1 0.0013 3844-45-9 Purified Water 49 7732-18-5 Sebozole Shampoo Page 2 of 5 (4) First Aid Measures Ingestion • In case of ingestion call a physician and poison control. EYE • If product enters eye, rinse thouroughly with cool fresh water for 10 to 15 minutes. If irritation persists, seek medical attention. • a burning sensation, excessive tears, sensitivity to light, swelling and redness of the conjunctiva and increasedblinking. • Limited evidence or practical experience suggests, that the material may cause eye irritation in a substantial number of individuals.Prolonged eye contact may cause inflammation characterized by a redness of the conjunctiva (similar to windburn SKIN • none INHALED • None (5) FIRE-FIGHTING MEASURES Suitable Extinguisher: FOAM, DRY POWDER, WATER, CO2 Special Precautions: None Special Protective Equipment: None (6) ACCIDENTAL RELEASE PROCEDURES Personal Precautions: None. -
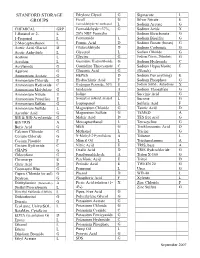
Stanford Storage Groups
STANFORD STORAGE Ethylene Glycol G Sigmacote L GROUPS Ficoll G Silver Nitrate E Formaldehyde w/ methanol L Sodium Acetate G CHEMICAL GRP Formaldehyde <37%, G Sodium Azide X 1-Butanol or 2- L 20% NBF Formalin Sodium Bicarbonate G 1-Propanol L Formamide L Sodium Bisulfite G 2-Mercaptoethanol L Formic Acid D Sodium Borate (borax) G Acetic Acid, Glacial D Glutaraldehyde D Sodium Carbonate G Acetic Anhydride L Glycerol L Sodium Chloride G Acetone L Glycine G Sodium Citrate, Dihydrate G Acrolein L Guanidine Hydrochloride G Sodium Hydroxide C Acrylamide G Guanidine Thiocyanate C Sodium Hypochlorite E Agarose G Gluconic Acid G (Bleach) Ammonium Acetate G HEPES D Sodium Per(anything) E Ammonium Chloride G Hydrochloric Acid F Sodium Phosphate G Ammonium Hydroxide C Hydrogen Peroxide, 30% E Sodium Sulfide, Anhydrous X Ammonium Molybdate G Imidazole A Sodium Thiosulfate G Ammonium Nitrate E Iodine E Succinic Acid G Ammonium Persulfate E Isoamyl or isobutyl alcohol L Sucrose G Ammonium Sulfate G Isopropanol L Sulfuric Acid F Ammonium Sulfide L Magnesium Chloride G Tannic Acid D Ascorbic Acid G Magnesium Sulfate G TEMED A BIS & BIS-Acrylamide G Maleic Acid D TES free acid G BIS TRIS A Mercaptoethanol L Tetracycline G Boric Acid G MES G Trichloroacetic Acid D Calcium Chloride G Methanol L Tricine G Cesium Chloride G N-Methyl-2-Pyrrolidone A Toluene L Cesium Fluoride F Mineral Oil L Triethanolamine A Cesium Hydroxide C Nitric Acid E TRIS, base A CHAPS G Oxalic Acid D TRIS Hydrochloride G Chloroform G Paraformaldehyde A Triton X-100 G Chromerge E Perchloric -

Access to Essential Medicines As a Component of the Right to Health Stephen P
Health: A Human Rights Perspective 82 ACCESS TO ESSENTIAL MEDICINES AS A COMPONENT OF THE RIGHT TO HEALTH Stephen P. Marks Introduction In Human Rights Obligations of Non-State Actors, Andrew Clapham wrote, “Perhaps the most obvious threat to human rights has come from the in- ability of people to achieve access to expensive medicine, particularly in the context of HIV and AIDS.”1 He was referring to threats to human rights from intellectual property agreements under the World Trade Organiza- tion, which are often seen as obeying a different – and many would say ut- terly incompatible – logic than human rights. The right to health, in the interpretation of the Committee on Economic Social and Cultural Rights, means that “States Parties … have a duty to prevent unreasonably high costs for access to essential medicines.”2 This chapter will explain the significance and place of the human right to essential medicines as a derivative right within the broader right to the highest attainable standard of physical and mental health. As a component of the right to health, the right to essential medicines depends not only on the production, distribution, and pricing of medicines, but also on the in- centives for research and development of drugs needed to treat diseases in developing countries, functioning health systems so that drugs are part of a rational system of quality treatment and care, as well as on infrastructure, Access to Essential Medicines as a Component of the Right to Health 83 so that they can be delivered to all areas where they are needed. Consider- ing that these broader issues are examined in other chapters, this chap- ter will focus more on the impediment to the realization of the right to essential medicines caused by the protection of intellectual property. -

World Health Organization Model List of Essential Medicines, 21St List, 2019
World Health Organizatio n Model List of Essential Medicines 21st List 2019 World Health Organizatio n Model List of Essential Medicines 21st List 2019 WHO/MVP/EMP/IAU/2019.06 © World Health Organization 2019 Some rights reserved. This work is available under the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 IGO licence (CC BY-NC-SA 3.0 IGO; https://creativecommons.org/licenses/by-nc-sa/3.0/igo). Under the terms of this licence, you may copy, redistribute and adapt the work for non-commercial purposes, provided the work is appropriately cited, as indicated below. In any use of this work, there should be no suggestion that WHO endorses any specific organization, products or services. The use of the WHO logo is not permitted. If you adapt the work, then you must license your work under the same or equivalent Creative Commons licence. If you create a translation of this work, you should add the following disclaimer along with the suggested citation: “This translation was not created by the World Health Organization (WHO). WHO is not responsible for the content or accuracy of this translation. The original English edition shall be the binding and authentic edition”. Any mediation relating to disputes arising under the licence shall be conducted in accordance with the mediation rules of the World Intellectual Property Organization. Suggested citation. World Health Organization Model List of Essential Medicines, 21st List, 2019. Geneva: World Health Organization; 2019. Licence: CC BY-NC-SA 3.0 IGO. Cataloguing-in-Publication (CIP) data. CIP data are available at http://apps.who.int/iris.