Rutherford and Radon
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Marie Curie and Her Time
Marie Curie and Her Time by Hélène Langevin-Joliot to pass our lives near each other hypnotized by our dreams, your patriotic dream, our humanitarian dream, arie Curie (1867–1934) belongs to that exclu- and our scientific dream.” sive group of women whose worldwide rec- Frederick Soddy wrote about Marie that she was Mognition and fame have endured for a century “the most beautiful discovery of Pierre Curie.” Of or more. She was indeed one of the major agents of course, it might also be said that Pierre Curie was the scientific revolution which allowed experimen- “the most beautiful discovery of Marie Skłodowska.” tal investigation to extend beyond the macroscopic It is difficult to imagine more contrasting personali- world. Her work placed the first stone in the founda- ties than those of Pierre and of Marie. In spite of that, tion of a new discipline: radiochemistry. And Curie’s or because of that, they complemented each other achievements are even more remarkable since they astonishingly well. Pierre was as dreamy as Marie was occurred in the field of science, an intellectual activ- organized. At the same time, they shared similar ideas ity traditionally forbidden to women. However, these about family and society. accomplishments alone don’t seem to fully explain the near mythic status of Marie Curie today. One hundred years ago, she was often considered to be just an assistant to her husband. Perhaps the reason her name still resonates is because of the compelling story of her life and her intriguing personality. The Most Beautiful Discovery of Pierre Curie The story of the young Maria Skłodowska leaving In this iconic photograph of participants at the Fifth her native Poland to pursue upper-level studies in Solvay Conference in 1927, Marie Curie is third from Paris sounds like something out of a novel. -

What Is Radon?
Biology Unit Radon Alert INVESTIGATION 3 WHAT IS RADON? INTRODUCTION Radon is a naturally occurring radioactive gas. It is formed by the radioactive breakdown of radium, and is found in soils just about everywhere. You cannot see it, taste it, or smell it. It is continuously formed in rocks and soils and escapes into the atmosphere. In some cases, it makes its way into homes, builds up to high concentrations in indoor air, and can become a health hazard. Although there are several different isotopes of radon, the one that is of greatest concern as a potential Radioactivity - the spontaneous human health threat is called radon-222. Radon-222 is emission of energy by certain (radio- formed naturally during a chain of radioactive disintegra- active) atoms, resulting in a change tion reactions (decay series). The decay series begins from one element to another or one when uranium-238 decays. Uranium is widely distrib- isotope to another. The energy can uted in rocks and soils throughout the earth’s crust. It has be in the form of alpha or beta par- a half-life of 4.5 billion years, which means a very slow ticles and gamma rays. breakdown. The decay series is shown schematically in Figure 1. There are eight different elements and 15 different isotopes in the series, beginning with uranium- 238 and ending with lead-206. New elements formed by radioactive disintegration reactions are called decay products. Thus, radium-226 is one of the decay products of uranium-238. Polonium-218 and lead-214 are decay products of radon-222. -

Case Studies in Environmental Medicine
ATSDR Case Studies in Environmental Medicine Radon Toxicity CS213021-A AGENCY FOR TOXIC SUBSTANCES AND DISEASE REGISTRY CASE STUDIES IN ENVIRONMENTAL MEDICINE (CSEM) Radon Toxicity Course : CB/WB1585 CE Original Date: 06/01/2010 CE Expiration Date: 06/01/2012 Key Concepts • The U. S. Environmental Protection Agency estimates that indoor radon exposure may result in 21,000 lung cancer deaths annually in the United States. • Radon may be second only to smoking as a cause of lung cancer. Increased use of medical radiation also contributes to the annual radiation dose. • The combination of smoking and radon exposure results in a higher health risk. • Current technology can easily decrease the concentration of radon in indoor air, and radon’s associated risk for producing lung cancer. About This This educational case study document is one in a series of and Other self-instructional modules designed to increase the primary Case Studies care provider’s knowledge of hazardous substances in the in environment and to promote the adoption of medical Environmental practices that aid in the evaluation and care of potentially Medicine exposed patients. The complete series of Case Studies in Environmental Medicine is on the ATSDR Web site at: http://www.atsdr.cdc.gov/emes/topics/. In addition, the downloadable PDF version of this educational series and other environmental medicine materials provide content in an electronic, printable format, especially for those who may lack adequate Internet service. How to Apply See Internet address: http://www2.cdc.gov/atsdrce/ for for and more information about continuing medical education Receive credits, continuing nursing education credits, and other Agency for Toxic Substances and Disease Registry Radon Toxicity Case Studies in Environmental Medicine (CSEM) Continuing continuing education units. -

Of the Periodic Table
of the Periodic Table teacher notes Give your students a visual introduction to the families of the periodic table! This product includes eight mini- posters, one for each of the element families on the main group of the periodic table: Alkali Metals, Alkaline Earth Metals, Boron/Aluminum Group (Icosagens), Carbon Group (Crystallogens), Nitrogen Group (Pnictogens), Oxygen Group (Chalcogens), Halogens, and Noble Gases. The mini-posters give overview information about the family as well as a visual of where on the periodic table the family is located and a diagram of an atom of that family highlighting the number of valence electrons. Also included is the student packet, which is broken into the eight families and asks for specific information that students will find on the mini-posters. The students are also directed to color each family with a specific color on the blank graphic organizer at the end of their packet and they go to the fantastic interactive table at www.periodictable.com to learn even more about the elements in each family. Furthermore, there is a section for students to conduct their own research on the element of hydrogen, which does not belong to a family. When I use this activity, I print two of each mini-poster in color (pages 8 through 15 of this file), laminate them, and lay them on a big table. I have students work in partners to read about each family, one at a time, and complete that section of the student packet (pages 16 through 21 of this file). When they finish, they bring the mini-poster back to the table for another group to use. -

Keeping Your Home Safe from RADON
Keeping Your Home Safe From RADON 800-662-9278 | Michigan.gov/radon 08/2019 What is Radon? Radon is a colorless and odorless gas that comes from the soil. The gas can accumulate in our home and in the air we breathe. Radon gas decays into fine particles that are radioactive. When inhaled, these fine particles can damage the lung. Exposure to radon over a long period of time can lead to lung cancer. It is estimated that 21,000 people die each year in the United States from lung cancer due to radon exposure. A radon test is the only way to know how much radon is in your home. Radon can be reduced with a mitigation system. The Michigan Department of Environment, Great Lakes, and Energy (EGLE) has created this guide to explain: • How radon accumulates in homes • The health risks of radon exposure • How to test your home for radon • What to do if your home has high radon • Radon policies C Keeping Your Home Safe From Radon Table of Contents Where Does Radon Come From? ............................................. 1 Radon in Michigan ....................................................................... 1 Percentage of Elevated Radon Test Results by County ......... 2 Is There a Safe Level of Radon? ............................................... 3 Radon Health Risks ..................................................................... 4 How Radon Enters the Home ..................................................... 6 Radon Pathways ........................................................................... 7 Radon Testing ............................................................................ -
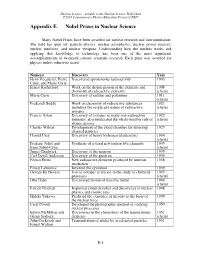
Appendix E Nobel Prizes in Nuclear Science
Nuclear Science—A Guide to the Nuclear Science Wall Chart ©2018 Contemporary Physics Education Project (CPEP) Appendix E Nobel Prizes in Nuclear Science Many Nobel Prizes have been awarded for nuclear research and instrumentation. The field has spun off: particle physics, nuclear astrophysics, nuclear power reactors, nuclear medicine, and nuclear weapons. Understanding how the nucleus works and applying that knowledge to technology has been one of the most significant accomplishments of twentieth century scientific research. Each prize was awarded for physics unless otherwise noted. Name(s) Discovery Year Henri Becquerel, Pierre Discovered spontaneous radioactivity 1903 Curie, and Marie Curie Ernest Rutherford Work on the disintegration of the elements and 1908 chemistry of radioactive elements (chem) Marie Curie Discovery of radium and polonium 1911 (chem) Frederick Soddy Work on chemistry of radioactive substances 1921 including the origin and nature of radioactive (chem) isotopes Francis Aston Discovery of isotopes in many non-radioactive 1922 elements, also enunciated the whole-number rule of (chem) atomic masses Charles Wilson Development of the cloud chamber for detecting 1927 charged particles Harold Urey Discovery of heavy hydrogen (deuterium) 1934 (chem) Frederic Joliot and Synthesis of several new radioactive elements 1935 Irene Joliot-Curie (chem) James Chadwick Discovery of the neutron 1935 Carl David Anderson Discovery of the positron 1936 Enrico Fermi New radioactive elements produced by neutron 1938 irradiation Ernest Lawrence -

ARIE SKLODOWSKA CURIE Opened up the Science of Radioactivity
ARIE SKLODOWSKA CURIE opened up the science of radioactivity. She is best known as the discoverer of the radioactive elements polonium and radium and as the first person to win two Nobel prizes. For scientists and the public, her radium was a key to a basic change in our understanding of matter and energy. Her work not only influenced the development of fundamental science but also ushered in a new era in medical research and treatment. This file contains most of the text of the Web exhibit “Marie Curie and the Science of Radioactivity” at http://www.aip.org/history/curie/contents.htm. You must visit the Web exhibit to explore hyperlinks within the exhibit and to other exhibits. Material in this document is copyright © American Institute of Physics and Naomi Pasachoff and is based on the book Marie Curie and the Science of Radioactivity by Naomi Pasachoff, Oxford University Press, copyright © 1996 by Naomi Pasachoff. Site created 2000, revised May 2005 http://www.aip.org/history/curie/contents.htm Page 1 of 79 Table of Contents Polish Girlhood (1867-1891) 3 Nation and Family 3 The Floating University 6 The Governess 6 The Periodic Table of Elements 10 Dmitri Ivanovich Mendeleev (1834-1907) 10 Elements and Their Properties 10 Classifying the Elements 12 A Student in Paris (1891-1897) 13 Years of Study 13 Love and Marriage 15 Working Wife and Mother 18 Work and Family 20 Pierre Curie (1859-1906) 21 Radioactivity: The Unstable Nucleus and its Uses 23 Uses of Radioactivity 25 Radium and Radioactivity 26 On a New, Strongly Radio-active Substance -

Radionuclides (Including Radon, Radium and Uranium)
Radionuclides (including Radon, Radium and Uranium) Hazard Summary Uranium, radium, and radon are naturally occurring radionuclides found in the environment. No information is available on the acute (short-term) noncancer effects of the radionuclides in humans. Animal studies have reported inflammatory reactions in the nasal passages and kidney damage from acute inhalation exposure to uranium. Chronic (long-term) inhalation exposure to uranium and radon in humans has been linked to respiratory effects, such as chronic lung disease, while radium exposure has resulted in acute leukopenia, anemia, necrosis of the jaw, and other effects. Cancer is the major effect of concern from the radionuclides. Radium, via oral exposure, is known to cause bone, head, and nasal passage tumors in humans, and radon, via inhalation exposure, causes lung cancer in humans. Uranium may cause lung cancer and tumors of the lymphatic and hematopoietic tissues. EPA has not classified uranium, radon or radium for carcinogenicity. Please Note: The main sources of information for this fact sheet are EPA's Integrated Risk Information System (IRIS) (5), which contains information on oral chronic toxicity and the RfD for uranium, and the Agency for Toxic Substances and Disease Registry's (ATSDR's) Toxicological Profiles for Uranium, Radium, and Radon. (1) Uses Uranium is used in nuclear power plants and nuclear weapons. Very small amounts are used in photography for toning, in the leather and wood industries for stains and dyes, and in the silk and wood industries. (2) Radium is used as a radiation source for treating neoplastic diseases, as a radon source, in radiography of metals, and as a neutron source for research. -

Chapter 1 the Biography of a Trafficking Material
Trafficking Materials and Maria Rentetzi Gendered Experimental Practices Chapter 1 The Biography of a Trafficking Material In 1904, an article titled "Radium and Radioactivity" appeared in Century 1 Magazine, a monthly popular magazine published from 1881 to 1930. The article presents Marie Curie's personal account of the discovery of radium and radioactivity. In the article, Curie discusses in depth her arduous attempts to study the radiation of the compounds of uranium and that of known chemical elements, hoping to discover more which are endowed with atomic radioactivity. She revels that it was the chemists who supplied her with the materials she needed. "As I desired to make a very thorough investigation, I had resource to different chemists, who put at my disposal specimens—in some cases the only ones in existence—containing very rare elements." Her next step was to examine different minerals, especially the oxide of uranium 2 ore (pitchblende). To her great surprise, this specimen was found to be four times more active than oxide of uranium itself. The explanation was more than obvious. "The ore must contain a substance more radioactive than uranium and thorium, and this substance must necessarily be a chemical element as yet unknown."1 Her attempts to isolate the new element would lead Marie and Pierre Curie, who joined her research shortly after, to the discovery of both polonium and radium, to the Nobel Prize in Physics in 1903, and to a second Nobel Prize in 1911, this time in chemistry. This chapter attempts to present a cultural -

Recalling Radon's Recognition
in your element Recalling radon’s recognition Brett F. Thornton and Shawn C. Burdette look back at the discovery — and the many different names — of element 86. n 1899, Pierre and Marie Curie noted an Thoron, unlike radon, requires no such “induced radioactivity” — left behind by clarification, and 220Rn is routinely called Iradium, distinct from its own radioactivity. thoron today. Thoron is far easier to say Ernest Rutherford and Robert B. Owens also than ‘radon-two-twenty’, perhaps explaining reported that year on a radioactive substance why the annual count of scientific papers 220 ( Rn, t1/2 = 55.6 s) emitted by thorium, which mentioning thoron has increased over twenty- they called emanation. In 1900, Friedrich fold since thoron was ‘disallowed’ in 1957. Dorn realized that the Curies had observed a Distinguishing between 222Rn (the 222 220 unique substance ( Rn, t1/2 = 3.8 d), similar isotope called ‘radon’) and Rn (thoron) is to emanation. In 1904, André-Louis Debierne not of idle linguistic and historic interest. 222 found a third radioactive particle; this one © SUPERSTOCK/ALAMY Rn can persist indoors, whereas the short- 219 produced from actinium ( Rn, t1/2 = 4 s). lived thoron cannot. Not all home radon These were at first regarded as elements and actinon (An) for the three isotopes; detectors (pictured) are sensitive to thoron, and became colloquially known as thorium names suggested by Elliott Q. Adams. and thoron-sensitive detectors must be emanation, radium emanation and actinium An official statement on a name for all placed with care because thoron does not emanation, but today we recognize them three isotopes — that is, a name for the travel far from its source. -

Dynamis-8 29.Indd
Isotope research before Isotopy: George Hevesy’s early radioactivity research in the Hungarian context Gábor Palló Institute for Philosophical Research, Hungarian Academy of Science. [email protected] Dynamis Fecha de recepción: 22 de febrero de 2008 [0211-9536] 2009; 29: 167-189 Fecha de aceptación: 12 de mayo de 2008 SUMMARY: 1.—Introduction. 2.—Two types of radioactivity research. 3.—The reception of radioactivity in Hungary. 4.—Starting migration: Béla Szilárd. 5.—George Hevesy’s early radioactivity research. 6.—Hevesy’s connection with the Hungarian scientific community in the 1910s. 7.—The origin of the radioactive tracer method. 8.—Delocalized research work. 9.—Budapest: The first applications of isotopes as indicators and tracers. 10.—Epilogue and concluding remarks. ABSTRACT: This paper presents a framework for the study of George Hevesy’s research in the 1910s by distinguishing two styles of radioactivity research: the analytical (as practices in Manchester and Vienna in some extent) and the natural historical styles (as practiced in Hungary). Georg Hevesy’s approach combined the two types. Indeed, by studying Hevesy’s research in context, I show that the earliest applications of isotopes were born in parallel with the establishment of the isotope theory of matter. PALABRAS CLAVE: Hungría, metodología de trazadores radioactivos, radioactividad, Béla Szilárd, George Hevesy, estilos de investigación científi ca. KEY WORDS: Hungary, radioactive tracer methodology, radioactivity, Béla Szilárd, George Hevesy, styles of scientifi c research. 1. Introduction George Hevesy was one of the earliest scientists to initiate and carry out research work applying radioactive isotopes. Frederick Soddy created the word «isotope» in February 18, 1913 1. -

The Noble Gases
INTERCHAPTER K The Noble Gases When an electric discharge is passed through a noble gas, light is emitted as electronically excited noble-gas atoms decay to lower energy levels. The tubes contain helium, neon, argon, krypton, and xenon. University Science Books, ©2011. All rights reserved. www.uscibooks.com Title General Chemistry - 4th ed Author McQuarrie/Gallogy Artist George Kelvin Figure # fig. K2 (965) Date 09/02/09 Check if revision Approved K. THE NOBLE GASES K1 2 0 Nitrogen and He Air P Mg(ClO ) NaOH 4 4 2 noble gases 4.002602 1s2 O removal H O removal CO removal 10 0 2 2 2 Ne Figure K.1 A schematic illustration of the removal of O2(g), H2O(g), and CO2(g) from air. First the oxygen is removed by allowing the air to pass over phosphorus, P (s) + 5 O (g) → P O (s). 20.1797 4 2 4 10 2s22p6 The residual air is passed through anhydrous magnesium perchlorate to remove the water vapor, Mg(ClO ) (s) + 6 H O(g) → Mg(ClO ) ∙6 H O(s), and then through sodium hydroxide to remove 18 0 4 2 2 4 2 2 the carbon dioxide, NaOH(s) + CO2(g) → NaHCO3(s). The gas that remains is primarily nitrogen Ar with about 1% noble gases. 39.948 3s23p6 36 0 The Group 18 elements—helium, K-1. The Noble Gases Were Kr neon, argon, krypton, xenon, and Not Discovered until 1893 83.798 radon—are called the noble gases 2 6 4s 4p and are noteworthy for their rela- In 1893, the English physicist Lord Rayleigh noticed 54 0 tive lack of chemical reactivity.