Clinical Management of Pain in Rodents
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
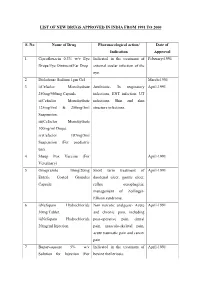
List of New Drugs Approved in India from 1991 to 2000
LIST OF NEW DRUGS APPROVED IN INDIA FROM 1991 TO 2000 S. No Name of Drug Pharmacological action/ Date of Indication Approval 1 Ciprofloxacin 0.3% w/v Eye Indicated in the treatment of February-1991 Drops/Eye Ointment/Ear Drop external ocular infection of the eye. 2 Diclofenac Sodium 1gm Gel March-1991 3 i)Cefaclor Monohydrate Antibiotic- In respiratory April-1991 250mg/500mg Capsule. infections, ENT infection, UT ii)Cefaclor Monohydrate infections, Skin and skin 125mg/5ml & 250mg/5ml structure infections. Suspension. iii)Cefaclor Monohydrate 100mg/ml Drops. iv)Cefaclor 187mg/5ml Suspension (For paediatric use). 4 Sheep Pox Vaccine (For April-1991 Veterinary) 5 Omeprazole 10mg/20mg Short term treatment of April-1991 Enteric Coated Granules duodenal ulcer, gastric ulcer, Capsule reflux oesophagitis, management of Zollinger- Ellison syndrome. 6 i)Nefopam Hydrochloride Non narcotic analgesic- Acute April-1991 30mg Tablet. and chronic pain, including ii)Nefopam Hydrochloride post-operative pain, dental 20mg/ml Injection. pain, musculo-skeletal pain, acute traumatic pain and cancer pain. 7 Buparvaquone 5% w/v Indicated in the treatment of April-1991 Solution for Injection (For bovine theileriosis. Veterinary) 8 i)Kitotifen Fumerate 1mg Anti asthmatic drug- Indicated May-1991 Tablet in prophylactic treatment of ii)Kitotifen Fumerate Syrup bronchial asthma, symptomatic iii)Ketotifen Fumerate Nasal improvement of allergic Drops conditions including rhinitis and conjunctivitis. 9 i)Pefloxacin Mesylate Antibacterial- In the treatment May-1991 Dihydrate 400mg Film Coated of severe infection in adults Tablet caused by sensitive ii)Pefloxacin Mesylate microorganism (gram -ve Dihydrate 400mg/5ml Injection pathogens and staphylococci). iii)Pefloxacin Mesylate Dihydrate 400mg I.V Bottles of 100ml/200ml 10 Ofloxacin 100mg/50ml & Indicated in RTI, UTI, May-1991 200mg/100ml vial Infusion gynaecological infection, skin/soft lesion infection. -

United States Patent (19) 11 Patent Number: 5,955,504 Wechter Et Al
USOO5955504A United States Patent (19) 11 Patent Number: 5,955,504 Wechter et al. (45) Date of Patent: Sep. 21, 1999 54 COLORECTAL CHEMOPROTECTIVE Marnett, “Aspirin and the Potential Role of Prostaglandins COMPOSITION AND METHOD OF in Colon Cancer, Cancer Research, 1992; 52:5575–89. PREVENTING COLORECTAL CANCER Welberg et al., “Proliferation Rate of Colonic Mucosa in Normal Subjects and Patients with Colonic Neoplasms: A 75 Inventors: William J. Wechter; John D. Refined Immunohistochemical Method.” J. Clin Pathol, McCracken, both of Redlands, Calif. 1990; 43:453-456. Thun et al., “Aspirin Use and Reduced Risk of Fatal Colon 73 Assignee: Loma Linda University Medical Cancer." N Engl J Med 1991; 325:1593-6. Center, Loma Linda, Calif. Peleg, et al., “Aspirin and Nonsteroidal Anti-inflammatory Drug Use and the Risk of Subsequent Colorectal Cancer.” 21 Appl. No.: 08/402,797 Arch Intern Med. 1994, 154:394–399. 22 Filed: Mar 13, 1995 Gridley, et al., “Incidence of Cancer among Patients With Rheumatoid Arthritis J. Natl Cancer Inst 1993 85:307-311. 51) Int. Cl. .......................... A61K 31/19; A61K 31/40; Labayle, et al., “Sulindac Causes Regression Of Rectal A61K 31/42 Polyps. In Familial Adenomatous Polyposis” Gastroenterol 52 U.S. Cl. .......................... 514/568; 514/569; 514/428; ogy 1991 101:635-639. 514/416; 514/375 Rigau, et al., “Effects Of Long-Term Sulindac Therapy On 58 Field of Search ..................................... 514/568, 570, Colonic Polyposis” Annals of Internal Medicine 1991 514/569, 428, 416, 375 11.5:952-954. Giardiello.et al., “Treatment Of Colonic and Rectal 56) References Cited Adenomas With Sulindac In Familial Adenomatous Poly U.S. -

Research Article RP-HPLC Method for Determination of Several Nsaids and Their Combination Drugs
Hindawi Publishing Corporation Chromatography Research International Volume 2013, Article ID 242868, 13 pages http://dx.doi.org/10.1155/2013/242868 Research Article RP-HPLC Method for Determination of Several NSAIDs and Their Combination Drugs Prinesh N. Patel, Gananadhamu Samanthula, Vishalkumar Shrigod, Sudipkumar C. Modh, and Jainishkumar R. Chaudhari Department of Pharmaceutical Analysis, National Institute of Pharmaceutical Education and Research (NIPER), Balanagar, Hyderabad, Andhra Pradesh 500037, India Correspondence should be addressed to Gananadhamu Samanthula; [email protected] Received 29 June 2013; Accepted 13 October 2013 Academic Editor: Andrew Shalliker Copyright © 2013 Prinesh N. Patel et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. An RP-HPLC method for simultaneous determination of 9 NSAIDs (paracetamol, salicylic acid, ibuprofen, naproxen, aceclofenac, diclofenac, ketorolac, etoricoxib, and aspirin) and their commonly prescribed combination drugs (thiocolchicoside, moxifloxacin, clopidogrel, chlorpheniramine maleate, dextromethorphan, and domperidone) was established. The separation was performed on ∘ Kromasil C18 (250 × 4.6 mm, 5 m) at 35 C using 15 mM phosphate buffer pH 3.25 and acetonitrile with gradient elution ata flow rate of 1.1 mL/min. The detection was performed by a diode array detector (DAD) at 230 nm with total run time of 30min. 2 Calibration curves were linear with correlation coefficients of determinationr ( ) > 0.999. Limit of detection (LOD) and Limit of quantification (LOQ) ranged from 0.04 to 0.97 g/mL and from 0.64 to 3.24 g/mL, respectively. -

Development of Macromolecular Prodrugs for the Treatment of Chronic Inflammatory Pain
University of Nebraska Medical Center DigitalCommons@UNMC Theses & Dissertations Graduate Studies Fall 12-14-2018 Development of Macromolecular Prodrugs for the Treatment of Chronic Inflammatory Pain Laura Weber University of Nebraska Medical Center Follow this and additional works at: https://digitalcommons.unmc.edu/etd Part of the Pharmacy and Pharmaceutical Sciences Commons Recommended Citation Weber, Laura, "Development of Macromolecular Prodrugs for the Treatment of Chronic Inflammatory Pain" (2018). Theses & Dissertations. 323. https://digitalcommons.unmc.edu/etd/323 This Dissertation is brought to you for free and open access by the Graduate Studies at DigitalCommons@UNMC. It has been accepted for inclusion in Theses & Dissertations by an authorized administrator of DigitalCommons@UNMC. For more information, please contact [email protected]. DEVELOPMENT OF MACROMOLECULAR PRODRUGS FOR THE TREATMENT OF CHRONIC INFLAMMATORY PAIN By Laura Weber A DISSERTATION Presented to the Faculty of The Graduate College of the University of NeBraska In Partial Fulfillment of the Requirements For the Degree of Doctor of Philosophy Department of Pharmaceutical Sciences Under the Supervision of Professor Dong Wang University of NeBraska Medical Center Omaha, NeBraska DecemBer 2018 TABLE OF CONTENTS ACKNOWLEDGEMENTS .............................................................................................. I ABSTRACT ................................................................................................................. IV LIST OF -

Table 6.12: Deaths from Poisoning, by Sex and Cause, Scotland, 2016
Table 6.12: Deaths from poisoning, by sex and cause, Scotland, 2016 ICD code(s), cause of death and substance(s) 1 Both Males Females ALL DEATHS FROM POISONING 2 1130 766 364 ACCIDENTS 850 607 243 X40 - X49 Accidental poisoning by and exposure to … X40 - Nonopioid analgesics, antipyretics and antirheumatics Paracetamol 2 1 1 Paracetamol, Cocaine, Amphetamine || 1 0 1 X41 - Antiepileptic, sedative-hypnotic, antiparkinsonism and psychotropic drugs, not elsewhere classified Alprazolam, MDMA, Cocaine || Cannabis, Alcohol 1 1 0 Alprazolam, Methadone || Pregabalin, Tramadol, Gabapentin, Cannabis 1 1 0 Alprazolam, Morphine, Heroin, Dihydrocodeine, Buprenorphine || Alcohol 1 1 0 Alprazolam, Oxycodone, Alcohol || Paracetamol 1 0 1 Amitriptyline, Cocaine, Etizolam || Paracetamol, Codeine, Hydrocodone, Alcohol 1 1 0 Amitriptyline, Dihydrocodeine || Diazepam, Paracetamol, Verapamil, Alcohol 1 1 0 Amitriptyline, Fluoxetine, Alcohol 1 0 1 Amitriptyline, Methadone, Diazepam || 1 1 0 Amitriptyline, Methadone, Morphine, Etizolam || Gabapentin, Cannabis, Alcohol 1 1 0 Amitriptyline, Venlafaxine 1 0 1 Amphetamine 1 1 0 Amphetamine || 1 1 0 Amphetamine || Alcohol 1 1 0 Amphetamine || Chlorpromazine 1 1 0 Amphetamine || Fluoxetine 1 1 0 Amphetamine, Dihydrocodeine, Alcohol || Procyclidine, Tramadol, Duloxetine, Haloperidol 1 0 1 Amphetamine, MDMA || Diclazepam, Cannabis, Alcohol 1 1 0 Amphetamine, Methadone || 1 1 0 Amphetamine, Oxycodone, Gabapentin, Zopiclone, Diazepam || Paracetamol, Alcohol 1 0 1 Amphetamine, Tramadol || Mirtazapine, Alcohol 1 1 0 Benzodiazepine -

Marketing Authorisations Granted in December 2020
Marketing authorisations granted in December 2020 PL Number Grant Date MA Holder Licensed Name(s) Active Ingredient Quantity Units Legal Status Territory PL 14251/0100 01/12/2020 MANX HEALTHCARE LIMITED COLCHICINE 500 MICROGRAMS TABLETS COLCHICINE 0.500 MILLIGRAMS POM UK PL 34424/0050 02/12/2020 KEY PHARMACEUTICALS LIMITED SPIRONOLACTONE 25MG FILM-COATED TABLETS SPIRONOLACTONE 25 MILLIGRAMS POM UK PL 34424/0051 02/12/2020 KEY PHARMACEUTICALS LIMITED SPIRONOLACTONE 50MG FILM-COATED TABLETS SPIRONOLACTONE 50 MILLIGRAMS POM UK PL 34424/0052 02/12/2020 KEY PHARMACEUTICALS LIMITED SPIRONOLACTONE 100MG FILM-COATED TABLETS SPIRONOLACTONE 100 MILLIGRAMS POM UK PL 36282/0021 03/12/2020 RIA GENERICS LIMITED COLCHICINE 500 MICROGRAM TABLETS COLCHICINE 500 MICROGRAMS POM UK PL 39352/0439 03/12/2020 KOSEI PHARMA UK LIMITED FROVATRIPTAN 2.5 MG FILM-COATED TABLETS FROVATRIPTAN SUCCINATE MONOHYDRATE 2.5 MILLIGRAMS POM UK PL 31750/0174 04/12/2020 SUN PHARMACEUTICAL INDUSTRIES EUROPE BV CETRORELIX SUN 0.25 MG SOLUTION FOR INJECTION IN PRE-FILLED SYRINGE CETRORELIX ACETATE 0.25 MILLIGRAMS PER MILLILITRE POM UK PL 34424/0054 04/12/2020 KEY PHARMACEUTICALS LIMITED ALIMEMAZINE TARTRATE 10MG FILM COATED TABLETS ALIMEMAZINE TARTRATE 10.00 MILLIGRAMS POM UK PL 17780/0858 07/12/2020 ZENTIVA PHARMA UK LIMITED FINGOLIMOD ZENTIVA 0.5 MG HARD CAPSULES FINGOLIMOD HYDROCHLORIDE 0.56 MILLIGRAMS POM UK PL 01502/0113 08/12/2020 HAMELN PHARMA LTD AMIODARONE HYDROCHLORIDE 20 MG/ML SOLUTION FOR INFUSION AMIODARONE HYDROCHLORIDE 20 MILLIGRAMS POM UK PL 16786/0006 08/12/2020 -

Torolac Combined with Oxycodone in Patient- Controlled Analgesia After Gynecol
Int. J. Med. Sci. 2015, Vol. 12 644 Ivyspring International Publisher International Journal of Medical Sciences 2015; 12(8): 644-649. doi: 10.7150/ijms.11828 Research Paper A Randomized Clinical Trial of Nefopam versus Ke- torolac Combined With Oxycodone in Patient- Controlled Analgesia after Gynecologic Surgery Boo-Young Hwang, Jae-Young Kwon, Do-Won Lee, Eunsoo Kim, Tae-Kyun Kim, Hae-Kyu Kim Department of Anesthesia and Pain Medicine, Biomedical Research Institute, Pusan National University Hospital, Busan, Republic of Korea Corresponding author: Jae-Young Kwon, Department of Anesthesia and Pain Medicine, Biomedical Research Institute, Pusan National University Hospital, 179 Gudeok-ro, Seo-gu, Busan 602-793, Republic of Korea. Tel: +82-51-240-7399; Fax: +82-51-242-7466; E-mail: [email protected] © 2015 Ivyspring International Publisher. Reproduction is permitted for personal, noncommercial use, provided that the article is in whole, unmodified, and properly cited. See http://ivyspring.com/terms for terms and conditions. Received: 2015.02.09; Accepted: 2015.07.18; Published: 2015.07.30 Abstract Objectives: Nefopam is a centrally-acting non-opioid analgesic, which has no effect on bleeding time and platelet aggregation. There has been no study about nefopam and oxycodone combina- tion for postoperative analgesia. In this study, we present efficacy and side effects of nefo- pam/oxycodone compared with ketorolac/oxycodone in patient-controlled analgesia (PCA) after gynecologic surgery. Methods: 120 patients undergoing gynecologic surgery were divided randomly into two groups: Nefopam group treated with oxycodone 1 mg and nefopam 1 mg bolus; and Ketorolac group treated with oxycodone 1 mg and ketorolac 1.5 mg bolus. -

ACUPAN Tablets
Consumer Medicine Information ACUPAN™ Nefopam hydrochloride 30 mg tablets What is in this leaflet This leaflet answers some common questions about ACUPAN tablets. However, it does not contain all the available information. It does not take the place of talking to your doctor or pharmacist. If you have any questions about ACUPAN that are not answered by this leaflet, ask your doctor or pharmacist. All medicines have risks and benefits. Your doctor has weighed the risks of you taking ACUPAN against the benefits before prescribing it to you. If you are worried about taking this medicine, ask your doctor or pharmacist. Keep this leaflet with your medicine as you may want to read it again. What ACUPAN is used for The name of your medicine is ACUPAN. ACUPAN belongs to a group of medicines called analgesics, commonly known as pain killers or pain relievers. It contains nefopam hydrochloride as the active ingredient. Nefopam hydrochloride interrupts the pain messages being sent to your brain, and it also acts in your brain to stop pain messages being felt. This means that ACUPAN does not stop the pain from happening, but you will not be able to feel the pain as much. ACUPAN is used to relieve both short term pain and long lasting pain (for example pain after an operation, dental pain, joint or muscle pain or after an injury). ACUPAN should not be used to treat the pain from a heart attack. Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed ACUPAN for another purpose. -

Full-Text (PDF)
Original Article J Babol Univ Med Sci Vol 18, Issu 12; Dec 2016. P:64-70 The Effect of Copper Chloride (CuCl2) on Pain and Inflammatory Paw Edema in Rats H. Nikbakhty (MSc)1, M. Fereidoni (PhD)1, A. Asadollahi (PhD)1 1. Department of Biology, Faculty of Science, Ferdowsi University of Mashhad, Mashhad, I.R.Iran J Babol Univ Med Sci; 18(12); Dec 2016; PP: 64-70 Received: Jul 12th 2016, Revised: Jul 27th 2016, Accepted: Sep 27th 2016. ABSTRACT BACKGROUND AND OBJECTIVE: As a crucial micronutrient, copper ion is engaged in various biochemical pathways and affects central nervous system, pain, and inflammation. Considering the significance of pain in physical and mental condition of patients, the present study aims to find new ways to reduce pain by investigating the effect of copper chloride on thermal and chemical pain and inflammatory edema through central and peripheral administrations. METHODS: In this empirical study, 77 male Wister rats (200–250g) were divided into 11 groups (n=7) including control group (with no treatment), sham 1 (saline, i.p) receiving 5, 10, 20 and 100 mg/kg CuCl2 intraperitoneally (i.p), sham 2 (saline, intrathecal (i.t)) receiving 0.002mg/10µl and 0.02mg/10µl CuCl2 intrathecally (i.t), sham 3 group receiving saline plus naloxone intraperitoneally (i.p) and the group receiving 10 mg/kg CuCl2 plus 2 mg/kg naloxone intraperitoneally (i.p). Thermal and chemical pain and the volume of inflammatory edema were assessed using tail flick, formalin and plethysmometery tests, respectively. Elevated plus maze and rotarod tests were used to examine the side effects of CuCl2. -

NIH Public Access Author Manuscript J Am Chem Soc
NIH Public Access Author Manuscript J Am Chem Soc. Author manuscript; available in PMC 2014 January 09. NIH-PA Author ManuscriptPublished NIH-PA Author Manuscript in final edited NIH-PA Author Manuscript form as: J Am Chem Soc. 2013 January 9; 135(1): 22–25. doi:10.1021/ja308733u. A binding site for non-steroidal anti-inflammatory drugs in FAAH Laura Bertolacci¶, Elisa Romeo¶, Marina Veronesi, Paola Magotti, Clara Albani, Mauro Dionisi, Chiara Lambruschini, Rita Scarpelli, Andrea Cavalli‡, Marco De Vivo, Daniele Piomelli*,†, and Gianpiero Garau* ‡Department of Pharmaceutical. Sciences, University of Bologna, Italy. †Department of Pharmacology, Univ. of California, Irvine, California, USA. Abstract In addition to inhibiting the cyclooxygenasemediated biosynthesis of prostanoids, various widely used non-steroidal anti-inflammatory drugs (NSAIDs) enhance endocannabinoid signaling by blocking the anandamidedegrading membrane enzyme, fatty acid amide hydrolase (FAAH). The X-ray structure of FAAH in complex with the NSAID carprofen, along with studies of site- directed mutagenesis, enzyme activity assays, and nuclear magnetic resonance, now reveal the molecular details of this interaction, providing information that may guide the design of dual FAAH-cyclooxygenase inhibitors with superior analgesic efficacy. Non-steroidal anti-inflammatory drugs (NSAIDs), one of the most widely used classes of therapeutic agents, alleviate pain and inflammation1 by inhibiting the enzymes cyclooxygenase-1 (COX-1) and COX-2,2 which catalyze the conversion of -

Effect of Restraint Stress on Nociceptive Responses in Rats: Role of the Histaminergic System
Niger. J. Physiol. Sci. 26(December 2011) 139 – 141 www.njps.com.ng Short communication Effect of restraint stress on nociceptive responses in rats: role of the histaminergic system *Ibironke G F and Mordi N E Department of Physiology, College of Medicine, University of Ibadan,Ibadan, Nigeria. Summary: Stress induced analgesia (SIA) is well known, but the reverse phenomenon, hyperalgesia is poorly documented. This study investigated the role of the histaminergic system in restraint stress hyperalgesia in rats, using thermal stimulation method (hot plate and tail flick tests). Paw licking and tail withdrawal latencies were taken before and after restraint for about one hour. Significant decreases (p< 0.05) were obtained in these latencies after the restraint in both tests. Administration of H1 and H2 receptor blockers, chlorpheniramine and cimetidine respectively 30 mins before the restraint still resulted in significant (p<0.05) reductions in these latencies, connoting the persistence of hyperalgesia, showing that histamine H1 and H2 receptors did not participate in the mechanism of restraint stress hyperalgesia. We therefore suggest a histaminergic independent mechanism for restraint stress induced hyperalgesia. Keywords: Restraint stress, Thermal stimulation, Hyperalgesia, Histamine. ©Physiological Society of Nigeria *Address for correspondence: [email protected] Manuscript Accepted: July, 2011 INTRODUCTION higher levels of suffering during stressful episodes (Conrad et al, 2007, Fishbain et al, 2006). Numerous Stress can have bilateral effects on pain related pre-clinical models have been developed to reproduce phenomena. Firstly, acute stress can produce various pain modalities that are encountered in the analgesia in humans and animals (Amit and Galina, clinic, however, animal studies assessing the 1986). -

The Anti-Nociceptive and Anti-Inflammatory Effect of Achyranthes Japonica Nakai
2004. Vol. 25. No. 4. 8-14 Korean Journal of Oriental Medicine Original Articles The Anti-nociceptive and Anti-inflammatory Effect of Achyranthes Japonica Nakai Hi-Joon Park, Ji-Suk Lee, Mi-Sook Hong, Chang-Ju Kim, Jin-Woo Kim, Hye-Jung Lee, Sabina Lim Research Group of Pain and Neuroscience, East-West Medical Research Institute, WHO Collaboration Center, Kyung Hee University, Seoul, Korea Objective : Achyranthes japonica Nakai (AJ) has been classified as a herb that activates blood flow and clears the stagnated blood. In this study, we evaluated its anti-nociceptive and anti-inflammatory activity in animals to clarify the effect of AJ on pain or inflammation. Methods : ICR mice and Sprague-Dawley rats were pretreated with an ethanolic extract of AJ with two dosages of 200 mg/kg (p.o.) and 400 mg/kg (p.o.). Nociceptive responses of acute pain were determined by hotplate and tail-flick tests. The effects of AJ on inflammation were evaluated by flexion/extention test and mechanical hyperalgesia test in models induced by both carrageenan and Complete Freund's Adjuvant (CFA). Results : AJ showed significant analgesic effects in both hotplate and tail-flick tests at the dose of 400 mg/kg. It also produced a significant inhibition of carrageenan-induced paw edema and CFA induced arthritis in rats at the dose of 400 mg/kg. Conclusion : We have demonstrated the analgesic and anti-inflammatory properties of an 80% ethanolic extract of AJ in animals. This suggests the application of AJ in relief of pain or inflammatory disease. Key Words: Analgesia; Inflammation; Achyranthes japonica Nakai Introduction stagnated blood.