Organophosphates
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

"U/^ ((*.U \ BIBLIOTHEEK STARINGGEBOUW Volatilization of Tri-Allate, Ethoprophos and Parathion Measured with Four Methods After Spraying on a Sandy Soil
"u/^ ((*.u \ BIBLIOTHEEK STARINGGEBOUW Volatilization of tri-allate, ethoprophos and parathion measured with four methods after spraying on a sandy soil G. Bor F. van den Berg J.H. Smelt R.A. Smidt A.E. van de Peppel-Groen M. Leistra Report 104 DLO Winand Staring Centre, Wageningen (The Netherlands), 1995 1 h FEB. 1998 0000' ABSTRACT Bor, G., F. van den Berg, J.H. Smelt, R.A. Smidt, A.E. van de Peppel-Groen, M. Leistra, 1995. Volatilization of tri-allate, ethoprophos and parathion measured withfour methods after spraying on a sandy soil. Wageningen (The Netherlands), DLO Winand Staring Centre. Report 104. 62 pp.; 9 Figs; 6 Tables; 14 Refs; 3 Annex. At about eleven times after application of tri-allate, ethoprophos and parathion to a sandy soil, their rates of volatilization were determined with the aerodynamic method (AD),th e Bowen-ratio method (BR), the theoretical-profile method (TP) and the Box method (B). The volatilization was highest for tri-allate and lowest for parathion. On the first day after application, the volatilization rate decreased sharply,bu tthereafte r the decreasewa smor egradual .Th edifference s involatilizatio n rate asdetermine d withth eAD ,B R andT Pmethod s werecomparativel y small.Th erate sdetermine d with the Box method were mostly lower than those determined with the other methods. Keywords: aerodynamic method, air quality, air sampling, Bowen ratio method, Box method, field experiment, gas chromatography, pesticide, polystyrene, theoretical-profile method, XAD ISSN 0927-4537 ©1995 DLO Winand Staring Centre for Integrated Land, Soil and Water Research (SC-DLO) P.O. -

Teratogenicity and Embryotoxicity of Organophosphorus Compounds in Animal Models - a Short Review
Mil. Med. Sci. Lett. (Voj. Zdrav. Listy) 2012, vol. 81(1), p. 16-26 ISSN 0372-7025 DOI: 10.31482/mmsl.2012.003 REVIEW ARTICLE TERATOGENICITY AND EMBRYOTOXICITY OF ORGANOPHOSPHORUS COMPOUNDS IN ANIMAL MODELS - A SHORT REVIEW Syed M Nurulain and M Shafiullah Department of Pharmacology and Therapeutic, Faculty of Medicine and Health Sciences, UAE University, AlAin, UAE. P.O.Box 17666, Alain, UAE Received 9 th December 2011. Revised 12 th February 2012. Published 9 th March 2012. Summary Organophosphorus compounds (OPCs) are a wide group of compounds both structurally and functionally. Each OPC has a unique toxicological profile. The exposure to this type of poison is not limited only to certain occupationally exposed people but also to children, women, pregnant women; all have chances to be exposed to this poison. During the recent past years it has been reported in many poison epidemiological studies and case reports that exposure of OPCs during pregnancy caused malformed fetuses, neural tube defect (NTD) and shortening of pregnancy. The literature for animal models reveals inconclusive evidence. The generalized view is that they are neither teratogenic nor embryotoxic. But it is not true. There is a lack of systematic study and scarcity of reports on the topic. The present study was undertaken to investigate the teratogenicity induced by organophosphorus compounds in different animal models by literature review. Literature was searched by Toxicology Data NetWork (TOXNET), Developmental and Reproductive Toxicology Database (DART), Toxicology Literature Online (TOXLINE), Hazardous Substances Data Bank (HSDB), Pubmed Central, Entrez-Pubmed, Science Direct, Directory Of Open Access Journal (DOAJ), Google Scholar and International Program on Chemical Safety (IPCS-INCHEM), Embase. -

Phytotoxicity of Some Organophosphate Insecticides to Oughly with Air Dry, Sieved Soil, to Pro Duce Rates of 37.5, 75 and ISO Mg Aj
Plant Protection Quarterly VoI.7(1} 1992 23 -------------------------------- ties of each pesticide were shaken thor Phytotoxicity of some organophosphate insecticides to oughly with air dry, sieved soil, to pro duce rates of 37.5, 75 and ISO mg aj. L-' onions and carrots during germination and emergence soil. Each pot constituted one plot. If it is assumed that in a field situation, P.J. Sinclair, New South Wales Agriculture, Horticultural Research and band-in-furrow treatment would treat a Advisory Station, Griffith, New South Wales 2680, Australia. strip 50 mm wide by 20 mm deep, the rates tested would correspond to 37.5, 75 R.J. Neeson and P.A. Williams, New South Wales Agriculture, Agricultural and 150 mg a.i. m" row, or 0.5,1.0 and 2.0 institute, Yanco, New South Wales 2703, Australia. kg a.i. ha" at a row spacing of 75 cm. The low and medium rates are then compara Summary ble to ra tes used in the field by Getzin The phytotoxicity of some commonly phytotoxicity from carbofuran applied to (1973), Thompson et al. (1981) and used insecticides to onions (Allium cepa) onions as a seed dressing. Chlorpyrifos is Goodyer et al. (1989) . and carrots (Daucus carota) during es generally non-phytotoxic at recom Daily counts of emerged seedlings were tablishment was assessed in pot trials. mended rates and methods of application, made at 7 to 18 days and at 21 days after Terbufos, ethoprophos, phoxirn and but some crops are especially sensitive to first watering. These data were used to de carbofuran (all 10% a.L granular fonnu it during the seedling stage or if the termine the total number of seedlings lations) and chlorpyrifos (25% a.i. -

Organophosphate Poisoning : a Review
120 Sinha and Sharma Med J Indones Organophosphate poisoning : A review Parmod K. Sinha, Ashok Sharma Abstrak Pestisida organofosfat digunakan secara luas di seluruh dunia. Keracunan oleh bahan ini merupakan masalah kesehatan masyarakat, terutama di negara berkembang. Zat neurotoksik organofosfat merupakan bahan yang dianggap mengancam dalam bidang militer dan terorisme. Mekanisme toksisitas bahan ini adalah dengan cara menghambat asetilkolinesterase yang mengakibatkan menumpuknya neurotransmitor asetilkolin dan terjadi rangsangan terus-menerus pada reseptor asetilkolin pada sistem saraf sentral maupun perifer. Selain krisis kolinergik, organofosfat dapat menimbulkan berbagai sindrom neurologis, baik akut maupun kronik. Sedangkan gejala peralihan ( intermediate) terjadi 1-4 hari setelah krisis kolinergik teratasi. Pengobatan standar terdiri dari reaktivasi asetilkolinesterase dengan antidot golongan oksim (prolidoksim, oksidoksime, HI-6 dan HLo7), dan pengendalian efek biokimia asetilkolin dengan menggunakan atropin. Golongan oksim yang baru HI-6 dan Hlo7 merupakan reaktivator asetilkolinesterase yang lebih cocok dan efektif untuk keracunan akut dan berat dibandingkan dengan prolidoksim dan obidoksim. Penderita yang mendapat pengobatan segera, biasanya dapat sembuh dari toksisitas akut, namun gejala neurologis ikutan dapat saja terjadi. (Med J Indones 2003; 12: 120-6) Abstract Organophosphate pesticides are used extensively worldwide, and poisoning by these agents, particularly in developing nations is a public health problem. Organophosphorous -

Approach to Site-Selective Protein Modification JUSTIN M
A “Tag-and-Modify” Approach to Site-Selective Protein Modification JUSTIN M. CHALKER, GONC-ALO J. L. BERNARDES, AND BENJAMIN G. DAVIS* Department of Chemistry, University of Oxford, Chemistry Research Laboratory, 12 Mansfield Road, Oxford OX1 3TA, United Kingdom RECEIVED ON FEBRUARY 27, 2011 CONSPECTUS ovalent modification can expand a protein's functional capacity. Fluorescent or radioactive labeling, for instance, allows C imaging of a protein in real time. Labeling with an affinity probe enables isolation of target proteins and other interacting molecules. At the other end of this functional spectrum, protein structures can be naturally altered by enzymatic action. ProteinÀprotein interactions, genetic regulation, and a range of cellular processes are under the purview of these post- translational modifications. The ability of protein chemists to install these covalent additions selectively has been critical for elucidating their roles in biology. Frequently the transformations must be applied in a site-specific manner, which demands the most selective chemistry. In this Account, we discuss the development and application of such chemistry in our laboratory. A centerpiece of our strategy is a “tag-and-modify” approach, which entails sequential installation of a uniquely reactive chemical group into the protein (the “tag”) and the selective or specific modification of this group. The chemical tag can be a natural or unnatural amino acid residue. Of the natural residues, cysteine is the most widely used as a tag. Early work in our program focused on selective disulfide formation in the synthesis of glycoproteins. For certain applications, the susceptibility of disulfides to reduction was a limitation and prompted the development of several methods for the synthesis of more stable thioether modifications. -

Guide No. 1 – October 2020 2/12 the CONCEPT and IMPLEMENTATION of CPA GUIDANCE RESIDUE LEVELS
Cooperation Centre for Scientific Research Relative to Tobacco CORESTA GUIDE N° 1 The Concept and Implementation of CPA Guidance Residue Levels October 2020 Agro-Chemical Advisory Committee CORESTA TECHNICAL GUIDE N° 1 Title: The Concept and Implementation of CPA Guidance Residue Levels Status: Valid Note: This document will be periodically reviewed by CORESTA Document history: Date of review Information July 2003 Version 1 GRL for Pyrethrins () and Terbufos corrected. December 2003 CPA terminology corrected. June 2008 Version 2 – GRLs revised and residue definitions added Provisional GRL of 2.00 ppm for Cyfluthrin to replace previous June 2010 GRL of 0.50 ppm July 2013 Version 3 – GRLs revised October 2013 Note for Maleic Hydrazide revised Version 4 – GRLs revised + clarification that scope of GRLs July 2016 applies predominantly to the production of traditional cigarette tobaccos and GAP associated with their cultivation. June 2018 Fluopyram GRL of 5 ppm added to GRL list Version 5 – Nine new CPAs with GRL added to list. November 2019 Revision of GRLs for Chlorantraniliprole and Indoxacarb. Updated web links. October 2020 Version 6 – Flupyradifurone GRL of 21 ppm added to GRL list. CORESTA Guide No. 1 – October 2020 2/12 THE CONCEPT AND IMPLEMENTATION OF CPA GUIDANCE RESIDUE LEVELS Executive Summary • Guidance Residue Levels (GRLs) are in the remit of the Agro-Chemical Advisory Committee (ACAC) of CORESTA. Their development is a joint activity of all ACAC members, who represent the leaf production, processing and manufacturing sectors of the Tobacco Industry. The concept of GRLs and their implementation are described in this guide. • GRLs provide guidance to tobacco growers and assist with interpretation and evaluation of results from analyses of residues of Crop Protection Agents (CPAs*). -

Determination of Organophosphorus Pesticide Residues in Vegetables Using Solid Phase Micro-Extraction Coupled with Gas Chromatography–flame Photometric Detector
Arabian Journal of Chemistry (2015) xxx, xxx–xxx King Saud University Arabian Journal of Chemistry www.ksu.edu.sa www.sciencedirect.com ORIGINAL ARTICLE Determination of organophosphorus pesticide residues in vegetables using solid phase micro-extraction coupled with gas chromatography–flame photometric detector Haizarul Aida Sapahin, Ahmad Makahleh *, Bahruddin Saad * School of Chemical Sciences, Universiti Sains Malaysia, 11800 Minden, Penang, Malaysia Received 17 July 2014; accepted 9 December 2014 KEYWORDS Abstract An adequate and simple analytical method based on solid-phase microextraction Organophosphorus pesticide; (SPME) followed by gas chromatography–flame photometric detection (GC–FPD) for the determi- Direct immersed-solid phase nation of eleven organophosphorus pesticide residues (i.e., ethoprophos, sulfotep, diazinon, tolclo- microextraction; fos-methyl, fenitrothion, chlorpyrifos, isofenphos, methidathion, ethion, triazophos, leptophos) in Gas chromatography–flame vegetables samples (cabbage, kale and mustard) was developed. Important parameters that influ- photometric detector; ence the extraction efficiency (i.e., fibre type, extraction modes, extraction time, salt addition, Vegetables desorption time and temperature) were systematically investigated. Four types of commercially available fibres (i.e., 50/30 lm divinylbenzene/carboxen/polydimethylsiloxane (DVB/CAR/PDMS), 65 lm polydimethylsiloxane/divinylbenzene (PDMS/DVB), 100 lm polydimethylsiloxane (PDMS), and 85 lm polyacrylate (PA)) were evaluated. PA fibre exhibited the best performance and was used for the rest of the studies. The optimised extraction conditions were: extraction time, 30 min at room temperature; stirring speed, 1275 rpm; salt content, 10% NaCl; desorption time and temper- ature, 11 min at 260 °C; and no pH adjustment of the sample extract. The method was validated over the range 0.1–100 lg/L. -

Chemical Name Federal P Code CAS Registry Number Acutely
Acutely / Extremely Hazardous Waste List Federal P CAS Registry Acutely / Extremely Chemical Name Code Number Hazardous 4,7-Methano-1H-indene, 1,4,5,6,7,8,8-heptachloro-3a,4,7,7a-tetrahydro- P059 76-44-8 Acutely Hazardous 6,9-Methano-2,4,3-benzodioxathiepin, 6,7,8,9,10,10- hexachloro-1,5,5a,6,9,9a-hexahydro-, 3-oxide P050 115-29-7 Acutely Hazardous Methanimidamide, N,N-dimethyl-N'-[2-methyl-4-[[(methylamino)carbonyl]oxy]phenyl]- P197 17702-57-7 Acutely Hazardous 1-(o-Chlorophenyl)thiourea P026 5344-82-1 Acutely Hazardous 1-(o-Chlorophenyl)thiourea 5344-82-1 Extremely Hazardous 1,1,1-Trichloro-2, -bis(p-methoxyphenyl)ethane Extremely Hazardous 1,1a,2,2,3,3a,4,5,5,5a,5b,6-Dodecachlorooctahydro-1,3,4-metheno-1H-cyclobuta (cd) pentalene, Dechlorane Extremely Hazardous 1,1a,3,3a,4,5,5,5a,5b,6-Decachloro--octahydro-1,2,4-metheno-2H-cyclobuta (cd) pentalen-2- one, chlorecone Extremely Hazardous 1,1-Dimethylhydrazine 57-14-7 Extremely Hazardous 1,2,3,4,10,10-Hexachloro-6,7-epoxy-1,4,4,4a,5,6,7,8,8a-octahydro-1,4-endo-endo-5,8- dimethanonaph-thalene Extremely Hazardous 1,2,3-Propanetriol, trinitrate P081 55-63-0 Acutely Hazardous 1,2,3-Propanetriol, trinitrate 55-63-0 Extremely Hazardous 1,2,4,5,6,7,8,8-Octachloro-4,7-methano-3a,4,7,7a-tetra- hydro- indane Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]- 51-43-4 Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]-, P042 51-43-4 Acutely Hazardous 1,2-Dibromo-3-chloropropane 96-12-8 Extremely Hazardous 1,2-Propylenimine P067 75-55-8 Acutely Hazardous 1,2-Propylenimine 75-55-8 Extremely Hazardous 1,3,4,5,6,7,8,8-Octachloro-1,3,3a,4,7,7a-hexahydro-4,7-methanoisobenzofuran Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime 26419-73-8 Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime. -

Ncomms12703.Pdf
ARTICLE Received 17 Feb 2016 | Accepted 26 Jul 2016 | Published 2 Sep 2016 DOI: 10.1038/ncomms12703 OPEN Chemoselective synthesis and analysis of naturally occurring phosphorylated cysteine peptides Jordi Bertran-Vicente1, Martin Penkert1,2, Olaia Nieto-Garcia1,2, Jean-Marc Jeckelmann3, Peter Schmieder1, Eberhard Krause1 & Christian P. R. Hackenberger1,2 In contrast to protein O-phosphorylation, studying the function of the less frequent N- and S-phosphorylation events have lagged behind because they have chemical features that prevent their manipulation through standard synthetic and analytical methods. Here we report on the development of a chemoselective synthetic method to phosphorylate Cys side-chains in unprotected peptides. This approach makes use of a reaction between nucleophilic phosphites and electrophilic disulfides accessible by standard methods. We achieve the stereochemically defined phosphorylation of a Cys residue and verify the modification using electron-transfer higher-energy dissociation (EThcD) mass spectrometry. To demonstrate the use of the approach in resolving biological questions, we identify an endogenous Cys phosphorylation site in IICBGlc, which is known to be involved in the carbohydrate uptake from the bacterial phosphotransferase system (PTS). This new chemical and analytical approach finally allows further investigating the functions and significance of Cys phosphorylation in a wide range of crucial cellular processes. 1 Leibniz-Institut fu¨r Molekulare Pharmakologie (FMP), Robert-Ro¨ssle-Str. 10, D-13125 Berlin, Germany. 2 Humboldt-Universita¨t zu Berlin, Institut fu¨r Chemie, Brook-Taylor-Str. 2, D-12489 Berlin, Germany. 3 Institute of Biochemistry and Molecular Medicine, University of Bern, 3012 Bern, Switzerland. Correspondence and requests for materials should be addressed to C.P.R.H. -
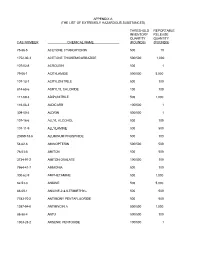
The List of Extremely Hazardous Substances)
APPENDIX A (THE LIST OF EXTREMELY HAZARDOUS SUBSTANCES) THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 75-86-5 ACETONE CYANOHYDRIN 500 10 1752-30-3 ACETONE THIOSEMICARBAZIDE 500/500 1,000 107-02-8 ACROLEIN 500 1 79-06-1 ACRYLAMIDE 500/500 5,000 107-13-1 ACRYLONITRILE 500 100 814-68-6 ACRYLYL CHLORIDE 100 100 111-69-3 ADIPONITRILE 500 1,000 116-06-3 ALDICARB 100/500 1 309-00-2 ALDRIN 500/500 1 107-18-6 ALLYL ALCOHOL 500 100 107-11-9 ALLYLAMINE 500 500 20859-73-8 ALUMINUM PHOSPHIDE 500 100 54-62-6 AMINOPTERIN 500/500 500 78-53-5 AMITON 500 500 3734-97-2 AMITON OXALATE 100/500 100 7664-41-7 AMMONIA 500 100 300-62-9 AMPHETAMINE 500 1,000 62-53-3 ANILINE 500 5,000 88-05-1 ANILINE,2,4,6-TRIMETHYL- 500 500 7783-70-2 ANTIMONY PENTAFLUORIDE 500 500 1397-94-0 ANTIMYCIN A 500/500 1,000 86-88-4 ANTU 500/500 100 1303-28-2 ARSENIC PENTOXIDE 100/500 1 THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 1327-53-3 ARSENOUS OXIDE 100/500 1 7784-34-1 ARSENOUS TRICHLORIDE 500 1 7784-42-1 ARSINE 100 100 2642-71-9 AZINPHOS-ETHYL 100/500 100 86-50-0 AZINPHOS-METHYL 10/500 1 98-87-3 BENZAL CHLORIDE 500 5,000 98-16-8 BENZENAMINE, 3-(TRIFLUOROMETHYL)- 500 500 100-14-1 BENZENE, 1-(CHLOROMETHYL)-4-NITRO- 500/500 500 98-05-5 BENZENEARSONIC ACID 10/500 10 3615-21-2 BENZIMIDAZOLE, 4,5-DICHLORO-2-(TRI- 500/500 500 FLUOROMETHYL)- 98-07-7 BENZOTRICHLORIDE 100 10 100-44-7 BENZYL CHLORIDE 500 100 140-29-4 BENZYL CYANIDE 500 500 15271-41-7 BICYCLO[2.2.1]HEPTANE-2-CARBONITRILE,5- -

Application of Dried Blood Spots for the Determination of Organophosphorus Pesticides by GC-MS/MS
UNIVERSIDADE DA BEIRA INTERIOR Ciências Application of dried blood spots for the determination of organophosphorus pesticides by GC-MS/MS Sofia Pires Seixo Soares Dissertação para obtenção do Grau de Mestre em Bioquímica (2º ciclo de estudos) Orientador: Doutor Mário Jorge Dinis Barroso Co-orientador: Professora Doutora Maria Eugenia Gallardo Alba Covilhã, junho de 2018 Agradecimentos A realização de uma dissertação de mestrado é o culminar de muitas horas de estudo, reflexão e trabalho pessoal, mas representa também uma etapa que não seria possível sem o contributo fundamental de várias pessoas. À Doutora Eugénia Gallardo e ao Doutor Mário Barroso, como excelentes mentores que são, quero agradecer toda a orientação, motivação, incentivo e confiança. Toda a ajuda e partilha de experiência e conhecimento durante todo este percurso. Ao Tiago Rosado, pela imprescindível ajuda e disponibilidade constante, bem como pela amizade. À Joana Gonçalves e ao Ângelo Luís um agradecimento especial pela amizade e carinho demonstrado ao longo deste ano. A todos, quero agradecer pelo bom ambiente proporcionado. Aos meus pais Ana Soares e José Soares, a quem dedico este feito, por me darem a possibilidade de concretizar conquistas como esta, pelo apoio incondicional, por sempre terem acreditado em mim e por fazerem parte deste momento. Aos meus avós e restante família, por todo o apoio e preocupação. Um agradecimento particular à Inês Ramos, à Rita Marques, à Maria Cunha e ao Pedro Batista pelo companheirismo, amizade e apoio de há tantos anos. E por último, mas nunca menos importante, um enorme obrigada ao meu namorado António Fernandes por toda a paciência, pelo incansável apoio, carinho e ajuda. -

Ambient Water Quality Criteria for Chlorpyrifos
United State. Office of Water EPA 440/5-86~05 Environmental Protection Regulations and Standards September 1986 Amy l. lea~erry Agency Criteria and Standards Division Washington DC 20460 Water &EPA Ambient Wat'er Quality Criteria for Chlorpyrifos - 1986 &~IENT AQUATIC LIFE WATER QUALITY CRITERIA FOR CHLORPYRIFOS U.S. ENVIRONMENTAL PROTECTION AGENCY OFFICE OF RESEARCH AND DEVELOPMENT ENVIRONMENTAL RESEARCH LABORATORIES DULUTH, MINNESOTA NARRAGANSETT, RHODE ISLAND NOTICES This document has been reviewed by the Criteria and Standards Division, Office of Water Regulations and Standards, U.S. Environmental Protection Agency, and approved for publication. Mention of trade names or commercial products does not constitute endorsement or recommendation for use. This document is availaryle to the public throu~h the National Technical Inf0rmation Service (NTIS), 5285 Port Royal Road, Springfield, VA 22161. 11 FOREWORD Section 304(a)(1) of the Clean Water Act of 1977 (P.L. 95-217) requires the Administrator of the Environmental Protection Agency to publish water quality criteria that accurately reflect the latest scientific knowledge on the kind and extent of all identifiable effects on health and welfare that might be expected from the presence of pollutants in any body of water~ including ground water. This document is a revision of proposed criteria based upon consideration of comments received from other Federal agencies~ State agencies~ special interest groups~ and individual scientists. Criteria contained in this document replace any previously published EPA aquatic life criteria for the same pollutant(s). The term "water quality criteria" is used in two sections of the Clean Water Act~ section 304(a)(1) and section 303(c)(2).