P 10-14 Temperature Convers.Pdf
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

CNPEM – Campus Map
1 2 CNPEM – Campus Map 3 § SUMMARY 11 Presentation 12 Organizers | Scientific Committee 15 Program 17 Abstracts 18 Role of particle size, composition and structure of Co-Ni nanoparticles in the catalytic properties for steam reforming of ethanol addressed by X-ray spectroscopies Adriano H. Braga1, Daniela C. Oliveira2, D. Galante2, F. Rodrigues3, Frederico A. Lima2, Tulio R. Rocha2, 4 1 1 R. J. O. Mossanek , João B. O. Santos and José M. C. Bueno 19 Electro-oxidation of biomass derived molecules on PtxSny/C carbon supported nanoparticles A. S. Picco,3, C. R. Zanata,1 G. C. da Silva,2 M. E. Martins4, C. A. Martins5, G. A. Camara1 and P. S. Fernández6,* 20 3D Studies of Magnetic Stripe Domains in CoPd Multilayer Thin Films Alexandra Ovalle1, L. Nuñez1, S. Flewett1, J. Denardin2, J.Escrigr2, S. Oyarzún2, T. Mori3, J. Criginski3, T. Rocha3, D. Mishra4, M. Fohler4, D. Engel4, C. Guenther5, B. Pfau5 and S. Eisebitt6. 1 21 Insight into the activity of Au/Ti-KIT-6 catalysts studied by in situ spectroscopy during the epoxidation of propene reaction A. Talavera-López *, S.A. Gómez-Torres and G. Fuentes-Zurita 22 Nanosystems for nasal isoniazid delivery: small-angle x-ray scaterring (saxs) and rheology proprieties A. D. Lima1, K. R. B. Nascimento1 V. H. V. Sarmento2 and R. S. Nunes1 23 Assembly of Janus Gold Nanoparticles Investigated by Scattering Techniques Ana M. Percebom1,2,3, Juan J. Giner-Casares1, Watson Loh2 and Luis M. Liz-Marzán1 24 Study of the morphology exhibited by carbon nanotube from synchrotron small angle X-ray scattering 1 2 1 1 Ana Pacheli Heitmann , Iaci M. -

Having Regard to the Opinion of the European Chapter 1 of the Annex Binding Within Five Years of Parliament1 ; the Date of Entry Into Force of This Directive
878 Official Journal of the European Communities 29.10.71 Official Journal of the European Communities No L 243/29 COUNCIL DIRECTIVE of 18 October 1971 on the approximation of the laws of the Member States relating to units of measurement (71/354/EEC ) THE COUNCIL OF THE EUROPEAN COMMUNITIES, particular their names, symbols and use are not identical in the Member countries ; Having regard to the Treaty establishing the European Economic Community, and in particular HAS ADOPTED THIS DIRECTIVE : Article 100 thereof; Article 1 Having regard to the proposal from the Commission ; 1 . Member States shall make the provisions of Having regard to the Opinion of the European Chapter 1 of the Annex binding within five years of Parliament1 ; the date of entry into force of this Directive. 2 . Member States shall, with effect from 31 Having regard to the Opinion of the Economic and December 1977 at the latest, prohibit the use of the Social Committee2; units of measurement listed in Chapter III of the Annex. Whereas - the laws which regulate the use of units of measurement in the Member States differ from one 3 . The units of measurement temporarily" retained Member State to another and therefore hinder trade ; in accordance with the provisions of Chapter II or whereas application of the rules relating to measuring Chapter III of the Annex may not be brought into instruments is closely linked to the use of units of compulsory use by the Member States where they ' are measurement in the metrological system ; whereas, in not authorised at the date when this Directive enters into force . -

Guide for the Use of the International System of Units (SI)
Guide for the Use of the International System of Units (SI) m kg s cd SI mol K A NIST Special Publication 811 2008 Edition Ambler Thompson and Barry N. Taylor NIST Special Publication 811 2008 Edition Guide for the Use of the International System of Units (SI) Ambler Thompson Technology Services and Barry N. Taylor Physics Laboratory National Institute of Standards and Technology Gaithersburg, MD 20899 (Supersedes NIST Special Publication 811, 1995 Edition, April 1995) March 2008 U.S. Department of Commerce Carlos M. Gutierrez, Secretary National Institute of Standards and Technology James M. Turner, Acting Director National Institute of Standards and Technology Special Publication 811, 2008 Edition (Supersedes NIST Special Publication 811, April 1995 Edition) Natl. Inst. Stand. Technol. Spec. Publ. 811, 2008 Ed., 85 pages (March 2008; 2nd printing November 2008) CODEN: NSPUE3 Note on 2nd printing: This 2nd printing dated November 2008 of NIST SP811 corrects a number of minor typographical errors present in the 1st printing dated March 2008. Guide for the Use of the International System of Units (SI) Preface The International System of Units, universally abbreviated SI (from the French Le Système International d’Unités), is the modern metric system of measurement. Long the dominant measurement system used in science, the SI is becoming the dominant measurement system used in international commerce. The Omnibus Trade and Competitiveness Act of August 1988 [Public Law (PL) 100-418] changed the name of the National Bureau of Standards (NBS) to the National Institute of Standards and Technology (NIST) and gave to NIST the added task of helping U.S. -

Music Similarity: Learning Algorithms and Applications
UC San Diego UC San Diego Electronic Theses and Dissertations Title More like this : machine learning approaches to music similarity Permalink https://escholarship.org/uc/item/8s90q67r Author McFee, Brian Publication Date 2012 Peer reviewed|Thesis/dissertation eScholarship.org Powered by the California Digital Library University of California UNIVERSITY OF CALIFORNIA, SAN DIEGO More like this: machine learning approaches to music similarity A dissertation submitted in partial satisfaction of the requirements for the degree Doctor of Philosophy in Computer Science by Brian McFee Committee in charge: Professor Sanjoy Dasgupta, Co-Chair Professor Gert Lanckriet, Co-Chair Professor Serge Belongie Professor Lawrence Saul Professor Nuno Vasconcelos 2012 Copyright Brian McFee, 2012 All rights reserved. The dissertation of Brian McFee is approved, and it is ac- ceptable in quality and form for publication on microfilm and electronically: Co-Chair Co-Chair University of California, San Diego 2012 iii DEDICATION To my parents. Thanks for the genes, and everything since. iv EPIGRAPH I’m gonna hear my favorite song, if it takes all night.1 Frank Black, “If It Takes All Night.” 1Clearly, the author is lamenting the inefficiencies of broadcast radio programming. v TABLE OF CONTENTS Signature Page................................... iii Dedication...................................... iv Epigraph.......................................v Table of Contents.................................. vi List of Figures....................................x List of Tables................................... -

MODULE 11: GLOSSARY and CONVERSIONS Cell Engines
Hydrogen Fuel MODULE 11: GLOSSARY AND CONVERSIONS Cell Engines CONTENTS 11.1 GLOSSARY.......................................................................................................... 11-1 11.2 MEASUREMENT SYSTEMS .................................................................................. 11-31 11.3 CONVERSION TABLE .......................................................................................... 11-33 Hydrogen Fuel Cell Engines and Related Technologies: Rev 0, December 2001 Hydrogen Fuel MODULE 11: GLOSSARY AND CONVERSIONS Cell Engines OBJECTIVES This module is for reference only. Hydrogen Fuel Cell Engines and Related Technologies: Rev 0, December 2001 PAGE 11-1 Hydrogen Fuel Cell Engines MODULE 11: GLOSSARY AND CONVERSIONS 11.1 Glossary This glossary covers words, phrases, and acronyms that are used with fuel cell engines and hydrogen fueled vehicles. Some words may have different meanings when used in other contexts. There are variations in the use of periods and capitalization for abbrevia- tions, acronyms and standard measures. The terms in this glossary are pre- sented without periods. ABNORMAL COMBUSTION – Combustion in which knock, pre-ignition, run- on or surface ignition occurs; combustion that does not proceed in the nor- mal way (where the flame front is initiated by the spark and proceeds throughout the combustion chamber smoothly and without detonation). ABSOLUTE PRESSURE – Pressure shown on the pressure gauge plus at- mospheric pressure (psia). At sea level atmospheric pressure is 14.7 psia. Use absolute pressure in compressor calculations and when using the ideal gas law. See also psi and psig. ABSOLUTE TEMPERATURE – Temperature scale with absolute zero as the zero of the scale. In standard, the absolute temperature is the temperature in ºF plus 460, or in metric it is the temperature in ºC plus 273. Absolute zero is referred to as Rankine or r, and in metric as Kelvin or K. -
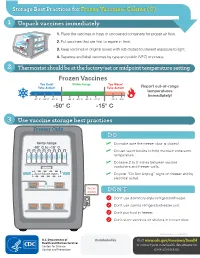
Storage Best Practices for Frozen Vaccines-Celsius
Storage Best Practices for Frozen Vaccines–Celsius (C) 1 Unpack vaccines immediately 1. Place the vaccines in trays or uncovered containers for proper air flow. 2. Put vaccines that are first to expire in front. HEP A - VFC 3. Keep vaccines in original boxes with lids closed to prevent exposure to light. 4. Separate and label vaccines by type and public (VFC) or private. 2 Thermostat should be at the factory-set or midpoint temperature setting Frozen Vaccines Too Cold! Within Range Too Warm! Take Action! Take Action! Report out-of-range temperatures immediately! -57° C -54° C -51° C -46° C -43° C -40° C -37° C -12° C -9°C -50° C -15° C 3 Use vaccine storage best practices Freezer Only DO temp range ✓ Do make sure the freezer door is closed! -50° C to -15° C ✓ Do use water bottles to help maintain consistent temperature. ✓ Do leave 2 to 3 inches between vaccine containers and freezer walls. don’t block vents ✓ Do post “Do Not Unplug” signs on freezer and by electrical outlet. do not unplug DON’T Don’t use dormitory-style refrigerator/freezer. Don’t use combo refrigerator/freezer unit. Don’t put food in freezer. Don’t store vaccines on shelves in freezer door. CS243541-D Revision %FDFNCFS 20 Distributed by Visit www.cdc.gov/vaccines/SandH or contact your state health department for more information. Test Your Knowledge 1 Which of the following units is the best for storing frozen vaccines? Freezer Freezer Freezer Freezer A. Full-size B. Full-size C. -
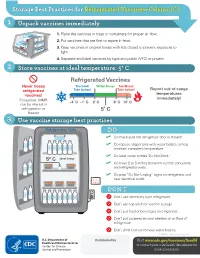
Storage Best Practices for Refrigerated Vaccines–Celsius (C)
Storage Best Practices for Refrigerated Vaccines–Celsius (C) 1 Unpack vaccines immediately 1. Place the vaccines in trays or containers for proper air flow. 2. Put vaccines that are first to expire in front. HEP A - VFC 3. Keep vaccines in original boxes with lids closed to prevent exposure to light. 4. Separate and label vaccines by type and public (VFC) or private. 2 Store vaccines at ideal temperature: 5° C Refrigerated Vaccines Never freeze Too Cold! Within Range Too Warm! refrigerated Take Action! Take Action! Report out-of-range vaccines! temperatures Exception: MMR immediately! -4˚ C -1˚ C 2˚ C 8˚ C 10˚ C can be stored in refrigerator or 5˚ C freezer 3 Use vaccine storage best practices Refrigerator Only DO ✓ Do make sure the refrigerator door is closed! ✓ Do replace crisper bins with water bottles to help maintain consistent temperature. ✓ Do label water bottles "Do Not Drink." 5° C ideal temp ✓ Do leave 2 to 3 inches between vaccine containers and refrigerator walls. ✓ Do post “Do Not Unplug” signs on refrigerator and do not near electrical outlet. unplug DON’T ; Don’t use dormitory-style refrigerator. ; Don’t use top shelf for vaccine storage. ; Don’t put food or beverages in refrigerator. ; Don’t put vaccines on door shelves or on floor of refrigerator. Don’t drink from or remove water bottles. ; CS243541-C Revision February 2018 Distributed by Visit www.cdc.gov/vaccines/SandH or contact your state health department for more information. Test Your Knowledge 1 Can you find at least 8 things that are wrong with vaccine storage in this refrigerator? 5° C ideal temp 2 When unpacking vaccines, why is it important to put the first-to-expire in the front? A. -

Thermodynamics Introduction and Basic Concepts
Thermodynamics Introduction and Basic Concepts by Asst. Prof. Channarong Asavatesanupap Mechanical Engineering Department Faculty of Engineering Thammasat University 2 What is Thermodynamics? Thermodynamics is the study that concerns with the ways energy is stored within a body and how energy transformations, which involve heat and work, may take place. Conservation of energy principle , one of the most fundamental laws of nature, simply states that “energy cannot be created or destroyed” but energy can change from one form to another during an energy interaction, i.e. the total amount of energy remains constant. 3 Thermodynamic systems or simply system, is defined as a quantity of matter or a region in space chosen for study. Surroundings are physical space outside the system boundary. Surroundings System Boundary Boundary is the surface that separates the system from its surroundings 4 Closed, Open, and Isolated Systems The systems can be classified into (1) Closed system consists of a fixed amount of mass and no mass may cross the system boundary. The closed system boundary may move. 5 (2) Open system (control volume) has mass as well as energy crossing the boundary, called a control surface. Examples: pumps, compressors, and water heaters. 6 (3) Isolated system is a general system of fixed mass where no heat or work may cross the boundaries. mass No energy No An isolated system is normally a collection of a main system and its surroundings that are exchanging mass and energy among themselves and no other system. 7 Properties of a system Any characteristic of a system is called a property. -

Insights Into the Unification of Forces
Insights into the unification of forces John A. Macken Previously unknown simple equations are presented which show a close relationship between the gravitational force and the electromagnetic force. For example, the gravitational force can be expressed as the square of the electromagnetic force for a fundamental set of conditions. These equations also imply that the wave properties of particles are an important component in the generation of these forces. These insights contradict previously held assumptions about gravity. In response to the question “Which of our basic physical assumptions are wrong?” this article proposes that the following are erroneous assumptions about gravity: 1 Gravity is not a true force. For example, the standard model does not include gravity and general relativity characterizes gravity as resulting from the geometry of spacetime. Therefore general relativity does not consider gravity to be a true force. 2 Gravity was united with the other forces at the start of the Big Bang but today gravity is completely decoupled from the other forces. For example, at the start of the Big Bang fundamental particles could have energy close to Planck energy and the gravitational force between two such particles was comparable to the electrostatic force. However, today the gravitational force is vastly weaker than the other forces and is assumed to be completely different than the other forces. 3 The forces are transferred by messenger particles. For example, the electromagnetic force is believed to be transferred by virtual photons and the gravitational force is believed by many physicists to be transferred by gravitons. Gravity has always been the most mysterious of the forces 1 ‐ 3. -
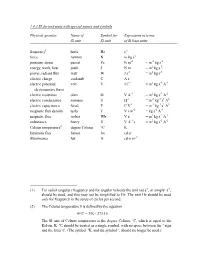
1.4.3 SI Derived Units with Special Names and Symbols
1.4.3 SI derived units with special names and symbols Physical quantity Name of Symbol for Expression in terms SI unit SI unit of SI base units frequency1 hertz Hz s-1 force newton N m kg s-2 pressure, stress pascal Pa N m-2 = m-1 kg s-2 energy, work, heat joule J N m = m2 kg s-2 power, radiant flux watt W J s-1 = m2 kg s-3 electric charge coulomb C A s electric potential, volt V J C-1 = m2 kg s-3 A-1 electromotive force electric resistance ohm Ω V A-1 = m2 kg s-3 A-2 electric conductance siemens S Ω-1 = m-2 kg-1 s3 A2 electric capacitance farad F C V-1 = m-2 kg-1 s4 A2 magnetic flux density tesla T V s m-2 = kg s-2 A-1 magnetic flux weber Wb V s = m2 kg s-2 A-1 inductance henry H V A-1 s = m2 kg s-2 A-2 Celsius temperature2 degree Celsius °C K luminous flux lumen lm cd sr illuminance lux lx cd sr m-2 (1) For radial (angular) frequency and for angular velocity the unit rad s-1, or simply s-1, should be used, and this may not be simplified to Hz. The unit Hz should be used only for frequency in the sense of cycles per second. (2) The Celsius temperature θ is defined by the equation θ/°C = T/K - 273.15 The SI unit of Celsius temperature is the degree Celsius, °C, which is equal to the Kelvin, K. -

The International System of Units (SI) - Conversion Factors For
NIST Special Publication 1038 The International System of Units (SI) – Conversion Factors for General Use Kenneth Butcher Linda Crown Elizabeth J. Gentry Weights and Measures Division Technology Services NIST Special Publication 1038 The International System of Units (SI) - Conversion Factors for General Use Editors: Kenneth S. Butcher Linda D. Crown Elizabeth J. Gentry Weights and Measures Division Carol Hockert, Chief Weights and Measures Division Technology Services National Institute of Standards and Technology May 2006 U.S. Department of Commerce Carlo M. Gutierrez, Secretary Technology Administration Robert Cresanti, Under Secretary of Commerce for Technology National Institute of Standards and Technology William Jeffrey, Director Certain commercial entities, equipment, or materials may be identified in this document in order to describe an experimental procedure or concept adequately. Such identification is not intended to imply recommendation or endorsement by the National Institute of Standards and Technology, nor is it intended to imply that the entities, materials, or equipment are necessarily the best available for the purpose. National Institute of Standards and Technology Special Publications 1038 Natl. Inst. Stand. Technol. Spec. Pub. 1038, 24 pages (May 2006) Available through NIST Weights and Measures Division STOP 2600 Gaithersburg, MD 20899-2600 Phone: (301) 975-4004 — Fax: (301) 926-0647 Internet: www.nist.gov/owm or www.nist.gov/metric TABLE OF CONTENTS FOREWORD.................................................................................................................................................................v -

Fast, Computer Supported Experimental Determination of Absolute Zero Temperature at School
European J of Physics Education Volume 5 Issue 1 2013 Bogacz & Pedziwiatr Fast, Computer Supported Experimental Determination of Absolute Zero Temperature at School Bogdan F. Bogacz Antoni T. Pędziwiatr M. Smoluchowski Institute of Physics Department of Methodics of Teaching and Methodology of Physics Jagiellonian University Reymonta 4, 30-059 Cracow, Poland E-mail: [email protected] (Received: 14.11. 2013, Accepted: 31.12.2013) Abstract A simple and fast experimental method of determining absolute zero temperature is presented. Air gas thermometer coupled with pressure sensor and data acquisition system COACH is applied in a wide range of temperature. By constructing a pressure vs temperature plot for air under constant volume it is possible to obtain - by extrapolation to zero pressure - a reasonable value of absolute zero temperature. The proposed way of conducting experiment allows students to "discover" intuitively the existence of minimal possible temperature and thus to understand the reason for introducing the concept of absolute zero temperature and absolute temperature Kelvin scale. It is a convincing and straightforward method to enhance understanding the general concept of temperature and support teaching thermodynamics and heat - mainly at secondary schools. This experiment was used with our university students who are being prepared to teach physics in secondary schools. They prepared physics lesson based on this material and then discussed didactic value of this material both for pupils and for teachers. Keywords: Physics education, absolute temperature, absolute zero determination, air gas thermometer. Introduction Temperature is a thermodynamic concept characterizing the state of thermal equilibrium of a macroscopic system. Equality of temperatures is a necessary and sufficient condition for reaching thermal equilibrium.