Application of Multiple Omics and Network Projection Analyses to Drug
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Table 2. Significant
Table 2. Significant (Q < 0.05 and |d | > 0.5) transcripts from the meta-analysis Gene Chr Mb Gene Name Affy ProbeSet cDNA_IDs d HAP/LAP d HAP/LAP d d IS Average d Ztest P values Q-value Symbol ID (study #5) 1 2 STS B2m 2 122 beta-2 microglobulin 1452428_a_at AI848245 1.75334941 4 3.2 4 3.2316485 1.07398E-09 5.69E-08 Man2b1 8 84.4 mannosidase 2, alpha B1 1416340_a_at H4049B01 3.75722111 3.87309653 2.1 1.6 2.84852656 5.32443E-07 1.58E-05 1110032A03Rik 9 50.9 RIKEN cDNA 1110032A03 gene 1417211_a_at H4035E05 4 1.66015788 4 1.7 2.82772795 2.94266E-05 0.000527 NA 9 48.5 --- 1456111_at 3.43701477 1.85785922 4 2 2.8237185 9.97969E-08 3.48E-06 Scn4b 9 45.3 Sodium channel, type IV, beta 1434008_at AI844796 3.79536664 1.63774235 3.3 2.3 2.75319499 1.48057E-08 6.21E-07 polypeptide Gadd45gip1 8 84.1 RIKEN cDNA 2310040G17 gene 1417619_at 4 3.38875643 1.4 2 2.69163229 8.84279E-06 0.0001904 BC056474 15 12.1 Mus musculus cDNA clone 1424117_at H3030A06 3.95752801 2.42838452 1.9 2.2 2.62132809 1.3344E-08 5.66E-07 MGC:67360 IMAGE:6823629, complete cds NA 4 153 guanine nucleotide binding protein, 1454696_at -3.46081884 -4 -1.3 -1.6 -2.6026947 8.58458E-05 0.0012617 beta 1 Gnb1 4 153 guanine nucleotide binding protein, 1417432_a_at H3094D02 -3.13334396 -4 -1.6 -1.7 -2.5946297 1.04542E-05 0.0002202 beta 1 Gadd45gip1 8 84.1 RAD23a homolog (S. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

Uncovering Ubiquitin and Ubiquitin-Like Signaling Networks Alfred C
REVIEW pubs.acs.org/CR Uncovering Ubiquitin and Ubiquitin-like Signaling Networks Alfred C. O. Vertegaal* Department of Molecular Cell Biology, Leiden University Medical Center, Albinusdreef 2, 2333 ZA Leiden, The Netherlands CONTENTS 8. Crosstalk between Post-Translational Modifications 7934 1. Introduction 7923 8.1. Crosstalk between Phosphorylation and 1.1. Ubiquitin and Ubiquitin-like Proteins 7924 Ubiquitylation 7934 1.2. Quantitative Proteomics 7924 8.2. Phosphorylation-Dependent SUMOylation 7935 8.3. Competition between Different Lysine 1.3. Setting the Scenery: Mass Spectrometry Modifications 7935 Based Investigation of Phosphorylation 8.4. Crosstalk between SUMOylation and the and Acetylation 7925 UbiquitinÀProteasome System 7935 2. Ubiquitin and Ubiquitin-like Protein Purification 9. Conclusions and Future Perspectives 7935 Approaches 7925 Author Information 7935 2.1. Epitope-Tagged Ubiquitin and Ubiquitin-like Biography 7935 Proteins 7925 Acknowledgment 7936 2.2. Traps Based on Ubiquitin- and Ubiquitin-like References 7936 Binding Domains 7926 2.3. Antibody-Based Purification of Ubiquitin and Ubiquitin-like Proteins 7926 1. INTRODUCTION 2.4. Challenges and Pitfalls 7926 Proteomes are significantly more complex than genomes 2.5. Summary 7926 and transcriptomes due to protein processing and extensive 3. Ubiquitin Proteomics 7927 post-translational modification (PTM) of proteins. Hundreds ff fi 3.1. Proteomic Studies Employing Tagged of di erent modi cations exist. Release 66 of the RESID database1 (http://www.ebi.ac.uk/RESID/) contains 559 dif- Ubiquitin 7927 ferent modifications, including small chemical modifications 3.2. Ubiquitin Binding Domains 7927 such as phosphorylation, acetylation, and methylation and mod- 3.3. Anti-Ubiquitin Antibodies 7927 ification by small proteins, including ubiquitin and ubiquitin- 3.4. -
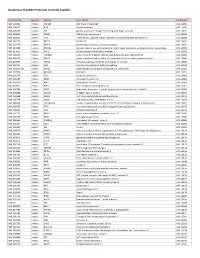
Quantigene Flowrna Probe Sets Currently Available
QuantiGene FlowRNA Probe Sets Currently Available Accession No. Species Symbol Gene Name Catalog No. NM_003452 Human ZNF189 zinc finger protein 189 VA1-10009 NM_000057 Human BLM Bloom syndrome VA1-10010 NM_005269 Human GLI glioma-associated oncogene homolog (zinc finger protein) VA1-10011 NM_002614 Human PDZK1 PDZ domain containing 1 VA1-10015 NM_003225 Human TFF1 Trefoil factor 1 (breast cancer, estrogen-inducible sequence expressed in) VA1-10016 NM_002276 Human KRT19 keratin 19 VA1-10022 NM_002659 Human PLAUR plasminogen activator, urokinase receptor VA1-10025 NM_017669 Human ERCC6L excision repair cross-complementing rodent repair deficiency, complementation group 6-like VA1-10029 NM_017699 Human SIDT1 SID1 transmembrane family, member 1 VA1-10032 NM_000077 Human CDKN2A cyclin-dependent kinase inhibitor 2A (melanoma, p16, inhibits CDK4) VA1-10040 NM_003150 Human STAT3 signal transducer and activator of transcripton 3 (acute-phase response factor) VA1-10046 NM_004707 Human ATG12 ATG12 autophagy related 12 homolog (S. cerevisiae) VA1-10047 NM_000737 Human CGB chorionic gonadotropin, beta polypeptide VA1-10048 NM_001017420 Human ESCO2 establishment of cohesion 1 homolog 2 (S. cerevisiae) VA1-10050 NM_197978 Human HEMGN hemogen VA1-10051 NM_001738 Human CA1 Carbonic anhydrase I VA1-10052 NM_000184 Human HBG2 Hemoglobin, gamma G VA1-10053 NM_005330 Human HBE1 Hemoglobin, epsilon 1 VA1-10054 NR_003367 Human PVT1 Pvt1 oncogene homolog (mouse) VA1-10061 NM_000454 Human SOD1 Superoxide dismutase 1, soluble (amyotrophic lateral sclerosis 1 (adult)) -

Heat Shock Factors: Integrators of Cell Stress, Development and Lifespan
REVIEWS Heat shock factors: integrators of cell stress, development and lifespan Malin Åkerfelt*‡, Richard I. Morimoto§ and Lea Sistonen*‡ Abstract | Heat shock factors (HSFs) are essential for all organisms to survive exposures to acute stress. They are best known as inducible transcriptional regulators of genes encoding molecular chaperones and other stress proteins. Four members of the HSF family are also important for normal development and lifespan-enhancing pathways, and the repertoire of HSF targets has thus expanded well beyond the heat shock genes. These unexpected observations have uncovered complex layers of post-translational regulation of HSFs that integrate the metabolic state of the cell with stress biology, and in doing so control fundamental aspects of the health of the proteome and ageing. Polytene chromosome In the early 1960s, Ritossa made the seminal discovery of shock response. Here, we present the recent discover- A chromosome that undergoes temperature-induced puffs in polytene chromosomes ies of novel target genes and physiological functions multiple rounds of DNA of Drosophila melanogaster larvae salivary glands1. of HSFs, which have changed the view that HSFs act replication, without cell A decade later, it was shown that the puffing pattern solely in the heat shock response. Based on the current division, and produces many sister chromatids that remain corresponded to a robust activation of genes encoding knowledge of small-molecule activators and inhibitors of synapsed together; for the heat shock proteins (HSPs), which function as HSFs, we also highlight the potential for pharmacologic example, in larval salivary molecular chaperones2. The heat shock response is a modulation of HSF-mediated gene regulation. -

Host Cell Factors Necessary for Influenza a Infection: Meta-Analysis of Genome Wide Studies
Host Cell Factors Necessary for Influenza A Infection: Meta-Analysis of Genome Wide Studies Juliana S. Capitanio and Richard W. Wozniak Department of Cell Biology, Faculty of Medicine and Dentistry, University of Alberta Abstract: The Influenza A virus belongs to the Orthomyxoviridae family. Influenza virus infection occurs yearly in all countries of the world. It usually kills between 250,000 and 500,000 people and causes severe illness in millions more. Over the last century alone we have seen 3 global influenza pandemics. The great human and financial cost of this disease has made it the second most studied virus today, behind HIV. Recently, several genome-wide RNA interference studies have focused on identifying host molecules that participate in Influen- za infection. We used nine of these studies for this meta-analysis. Even though the overlap among genes identified in multiple screens was small, network analysis indicates that similar protein complexes and biological functions of the host were present. As a result, several host gene complexes important for the Influenza virus life cycle were identified. The biological function and the relevance of each identified protein complex in the Influenza virus life cycle is further detailed in this paper. Background and PA bound to the viral genome via nucleoprotein (NP). The viral core is enveloped by a lipid membrane derived from Influenza virus the host cell. The viral protein M1 underlies the membrane and anchors NEP/NS2. Hemagglutinin (HA), neuraminidase Viruses are the simplest life form on earth. They parasite host (NA), and M2 proteins are inserted into the envelope, facing organisms and subvert the host cellular machinery for differ- the viral exterior. -

Comparative Analysis of the Ubiquitin-Proteasome System in Homo Sapiens and Saccharomyces Cerevisiae
Comparative Analysis of the Ubiquitin-proteasome system in Homo sapiens and Saccharomyces cerevisiae Inaugural-Dissertation zur Erlangung des Doktorgrades der Mathematisch-Naturwissenschaftlichen Fakultät der Universität zu Köln vorgelegt von Hartmut Scheel aus Rheinbach Köln, 2005 Berichterstatter: Prof. Dr. R. Jürgen Dohmen Prof. Dr. Thomas Langer Dr. Kay Hofmann Tag der mündlichen Prüfung: 18.07.2005 Zusammenfassung I Zusammenfassung Das Ubiquitin-Proteasom System (UPS) stellt den wichtigsten Abbauweg für intrazelluläre Proteine in eukaryotischen Zellen dar. Das abzubauende Protein wird zunächst über eine Enzym-Kaskade mit einer kovalent gebundenen Ubiquitinkette markiert. Anschließend wird das konjugierte Substrat vom Proteasom erkannt und proteolytisch gespalten. Ubiquitin besitzt eine Reihe von Homologen, die ebenfalls posttranslational an Proteine gekoppelt werden können, wie z.B. SUMO und NEDD8. Die hierbei verwendeten Aktivierungs- und Konjugations-Kaskaden sind vollständig analog zu der des Ubiquitin- Systems. Es ist charakteristisch für das UPS, daß sich die Vielzahl der daran beteiligten Proteine aus nur wenigen Proteinfamilien rekrutiert, die durch gemeinsame, funktionale Homologiedomänen gekennzeichnet sind. Einige dieser funktionalen Domänen sind auch in den Modifikations-Systemen der Ubiquitin-Homologen zu finden, jedoch verfügen diese Systeme zusätzlich über spezifische Domänentypen. Homologiedomänen lassen sich als mathematische Modelle in Form von Domänen- deskriptoren (Profile) beschreiben. Diese Deskriptoren können wiederum dazu verwendet werden, mit Hilfe geeigneter Verfahren eine gegebene Proteinsequenz auf das Vorliegen von entsprechenden Homologiedomänen zu untersuchen. Da die im UPS involvierten Homologie- domänen fast ausschließlich auf dieses System und seine Analoga beschränkt sind, können domänen-spezifische Profile zur Katalogisierung der UPS-relevanten Proteine einer Spezies verwendet werden. Auf dieser Basis können dann die entsprechenden UPS-Repertoires verschiedener Spezies miteinander verglichen werden. -

Folding Status Is Determinant Over Traffic-Competence in Defining
cells Article Folding Status Is Determinant over Traffic-Competence in Defining CFTR Interactors in the Endoplasmic Reticulum 1, 1, 2 1 3 João D. Santos y, Sara Canato y, Ana S. Carvalho , Hugo M. Botelho , Kerman Aloria , Margarida D. Amaral 1 , Rune Matthiesen 2, Andre O. Falcao 1,4 and Carlos M. Farinha 1,* 1 BioISI—Biosystems & Integrative Sciences Institute, Faculty of Sciences, University of Lisboa, Campo Grande C8, 1749-016 Lisboa, Portugal; [email protected] (J.D.S.); [email protected] (S.C.); [email protected] (H.M.B.); [email protected] (M.D.A.); [email protected] (A.O.F.) 2 CEDOC-Chronic Diseases Research Centre, Nova Medical School, Faculdade de Ciências Médicas, Universidade Nova de Lisboa, Rua Câmara Pestana, 1150-082 Lisboa, Portugal; [email protected] (A.S.C.); [email protected] (R.M.) 3 Proteomics Core Facility-SGIKER, University of the Basque Country UPV/EHU, Barrio Sariena, 48940 Vizcaya, Spain; [email protected] 4 LASIGE, Faculty of Sciences, University of Lisboa, Campo Grande, 1749-016 Lisbon, Portugal * Correspondence: [email protected]; Tel.: +351-217500864 These authors contributed equally to this work. y Received: 28 February 2019; Accepted: 12 April 2019; Published: 14 April 2019 Abstract: The most common cystic fibrosis-causing mutation (F508del, present in ~85% of CF patients) leads to CFTR misfolding, which is recognized by the endoplasmic reticulum (ER) quality control (ERQC), resulting in ER retention and early degradation. It is known that CFTR exit from the ER is mediated by specific retention/sorting signals that include four arginine-framed tripeptide (AFT) retention motifs and a diacidic (DAD) exit code that controls the interaction with the COPII machinery. -

Signal Transducer and Activator of Transcription 3-Mediated Regulation
European Heart Journal (2011) 32, 1287–1297 BASIC SCIENCE doi:10.1093/eurheartj/ehq369 Signal transducer and activator of transcription 3-mediated regulation of miR-199a-5p links cardiomyocyte and endothelial cell function in the heart: a key role for ubiquitin-conjugating Downloaded from https://academic.oup.com/eurheartj/article/32/10/1287/563017 by guest on 26 September 2021 enzymes Arash Haghikia 1, Ewa Missol-Kolka 1, Dimitrios Tsikas 2, Letizia Venturini 3, Stephanie Brundiers 3, Mirko Castoldi 4, Martina U. Muckenthaler 4, Matthias Eder 3, Britta Stapel 1, Thomas Thum 5, Aiden Haghikia 6, Elisabeth Petrasch-Parwez 7, Helmut Drexler 1†, Denise Hilfiker-Kleiner 1*‡, and Michaela Scherr 3‡ 1Department of Cardiology and Angiology, Medical School Hannover, 30625 Hannover, Germany; 2Department of Clinical Pharmacology, Medical School Hannover, 30625 Hannover, Germany; 3Department of Hematology, Hemostasis, Oncology and Stem Cell Transplantation, Medical School Hannover, 30625 Hannover, Germany; 4Department of Pediatric Oncology, Hematology and Immunology, University of Heidelberg, 69120 Heidelberg, Germany; 5Institute of Molecular and Translational Therapeutic Strategies, Medical School Hannover, 30625 Hannover, Germany; 6Department of Neurology, St. Josef-Hospital, Ruhr-University, 44791 Bochum, Germany; and 7Department of Neuroanatomy and Molecular Brain Research, Ruhr-University, 44801 Bochum, Germany Received 27 April 2010; revised 16 July 2010; accepted 18 August 2010; online publish-ahead-of-print 21 October 2010 Aims Mice with -

(12) Patent Application Publication (10) Pub. No.: US 2004/0009477 A1 Fernandez Et Al
US 20040009477A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2004/0009477 A1 Fernandez et al. (43) Pub. Date: Jan. 15, 2004 (54) METHODS FOR PRODUCING LIBRARIES ation of application No. 09/647,651, now abandoned, OF EXPRESSIBLE GENE SEQUENCES filed as 371 of international application No. PCT/ US99/07270, filed on Apr. 2, 1999, which is a con (75) Inventors: Joseph M. Fernandez, Carlsbad, CA tinuation-in-part of application No. 09/054,936, filed (US); John A. Heyman, Rixensart on Apr. 3, 1998, now abandoned. (BE); James P. Hoeffler, Anchorage, AK (US); Heather L. Marks-Hull, (30) Foreign Application Priority Data Oceanside, CA (US); Michelle L. Sindici, San Diego, CA (US) Apr. 2, 1999 (US)....................................... US99/07270 Correspondence Address: Publication Classification LISA A. HAILE, Ph.D. GRAY CARY WARE & FREDENRICH LLP 51)1) Int. Cl.Cl." .............................. C12O 1/68 ; C12P 19/34 Suite 1100 (52) U.S. Cl. ............................................... 435/6; 435/91.2 43.65 Executive Drive (57) ABSTRACT San Diego, CA 92.121-2133 (US) The present invention comprises a method for producing (73) Assignee: INVITROGEN CORPORATION libraries of expressible gene Sequences. The method of the invention allows for the Simultaneous manipulation of mul (21) Appl. No.: 09/990,091 tiple gene Sequences and thus allows libraries to be created in an efficient and high throughput manner. The expression (22) Filed: Nov. 21, 2001 vectors containing verified gene Sequences can be used to Related U.S. Application Data transfect cells for the production of recombinant proteins. The invention further comprises libraries of expressible gene (63) Continuation of application No. -

Bacillus Anthracis' Lethal Toxin Induces Broad Transcriptional Responses In
Chauncey et al. BMC Immunology 2012, 13:33 http://www.biomedcentral.com/1471-2172/13/33 RESEARCH ARTICLE Open Access Bacillus anthracis’ lethal toxin induces broad transcriptional responses in human peripheral monocytes Kassidy M Chauncey1, M Cecilia Lopez2, Gurjit Sidhu1, Sarah E Szarowicz1, Henry V Baker2, Conrad Quinn3 and Frederick S Southwick1* Abstract Background: Anthrax lethal toxin (LT), produced by the Gram-positive bacterium Bacillus anthracis, is a highly effective zinc dependent metalloprotease that cleaves the N-terminus of mitogen-activated protein kinase kinases (MAPKK or MEKs) and is known to play a role in impairing the host immune system during an inhalation anthrax infection. Here, we present the transcriptional responses of LT treated human monocytes in order to further elucidate the mechanisms of LT inhibition on the host immune system. Results: Western Blot analysis demonstrated cleavage of endogenous MEK1 and MEK3 when human monocytes were treated with 500 ng/mL LT for four hours, proving their susceptibility to anthrax lethal toxin. Furthermore, staining with annexin V and propidium iodide revealed that LT treatment did not induce human peripheral monocyte apoptosis or necrosis. Using Affymetrix Human Genome U133 Plus 2.0 Arrays, we identified over 820 probe sets differentially regulated after LT treatment at the p <0.001 significance level, interrupting the normal transduction of over 60 known pathways. As expected, the MAPKK signaling pathway was most drastically affected by LT, but numerous genes outside the well-recognized pathways were also influenced by LT including the IL-18 signaling pathway, Toll-like receptor pathway and the IFN alpha signaling pathway. -

CDKN2A Antibody (Pab)
CDKN2A antibody (pAb) Catalog Nos: 61619, 61620 Volumes: 100 µl, 10 µl RRID: AB_2793705 Purification: Affinity Purified Isotype: IgG Host: Rabbit Application(s): IP, WB Molecular Weight: 16 kDa Reactivity: Human Background: CDKN2A (Cyclin-Dependent Kinase Inhibitor 2A) is capable of inducing cell cycle arrest in G1 and G2 phases. Acts as a tumor suppressor. Binds to MDM2 and blocks its nucleocytoplasmic shuttling by sequestering it in the nucleolus. This inhibits the oncogenic action of MDM2 by blocking MDM2-induced degradation of p53 and enhancing p53-dependent transactivation and apoptosis. Also induces G2 arrest and apoptosis in a p53-independent manner by preventing the activation of cyclin B1/CDC2 complexes. Binds to BCL6 and down-regulates BCL6-induced transcriptional repression. Binds to E2F1 and MYC and blocks their transcriptional activator activity but has no effect on MYC transcriptional repression. Binds to TOP1/TOPOI and stimulates its activity. This complex binds to rRNA gene promoters and may play a role in rRNA transcription and/or maturation. Interacts with NPM1/B23 and promotes its polyubiquitination and degradation, thus inhibiting rRNA processing. CDKN2A antibody (pAb) tested by Interacts with COMMD1 and promotes its 'Lys63'-linked polyubiquitination. Interacts Immunoprecipitation. 10 µl of CDKN2A antibody was used to with UBE2I/UBC9 and enhances sumoylation of a number of its binding partners immunoprecipitate CDKN2A from 400 µg of including MDM2 and E2F1. Binds to HUWE1 and represses its ubiquitin ligase HeLa whole cell extract (lane 2). 10 µl of rabbit activity. May play a role in controlling cell proliferation and apoptosis during mammary IgG was used as a negative control (lane 1).