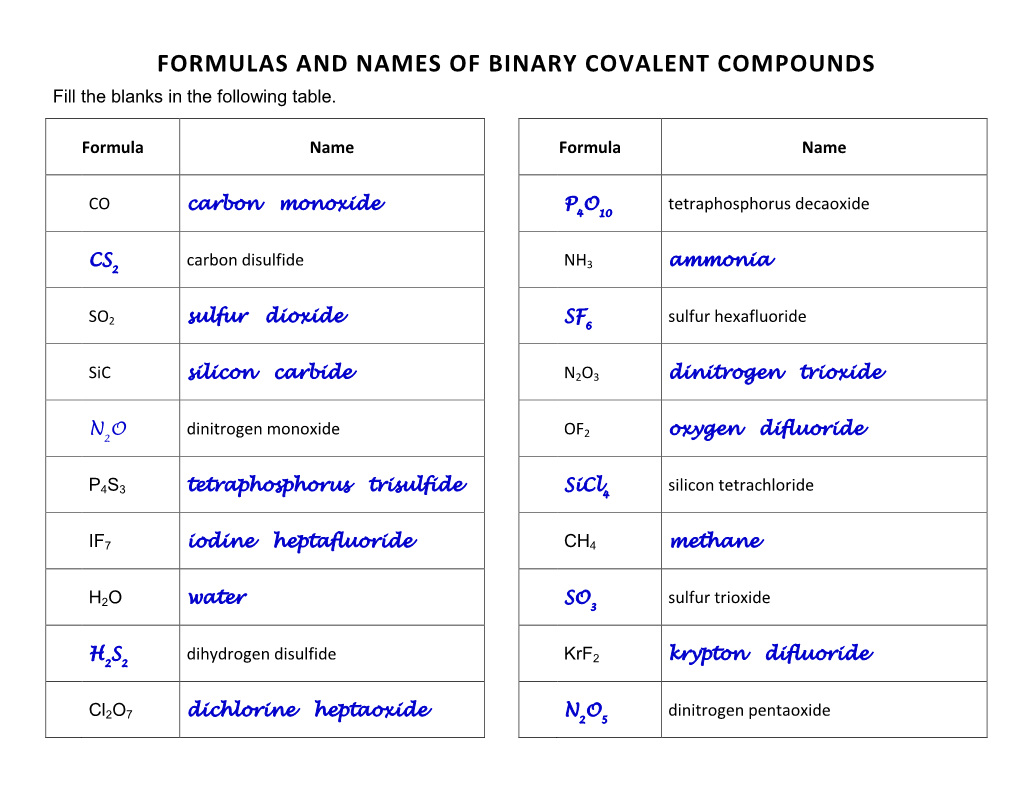
Formulas and Names of Binary Covalent Compounds
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Solutions to Exercises
Solutions to Exercises Note: There may be more than one correct path to a correct answer. Chapter 1. Problem Solving 1. If your answer doesn’tagree with the published answer,possibilities include a) your answer may be incorrect, b) the published answer may be incorrect, c) both may be incorrect, d) both may be correct (i.e. there may be more than one correct answer), e) typographical error (answer out of place). 2. Terminology: chemistry is a noun (object), but could refer to an object of study or prac- tice, etc. As an object of study,chemistry could refer to what is expected to be known by a novice, all that is known, all that may be known, etc; as a practice, chemistry could refer to practical applications, theoretical principles, processes where substances change called chemical reactions, etc. The word reasonable might connote logic, understand- ing, ethical practice, demand, etc. One might logically conclude that the subject of chemistry should be understandable, or that the field of chemistry should be non-productive (if it is reasonable to assume that chemistry is harmful to the environment, say), etc. This example is not meant to be totally trivial. Communication involves common under- standing of terminology,construction (syntax) and meaning (semantics). Forexample, the word "add" may mean different things depending on the context; e.g. calculators (add twointegers), travelagents (add the mileage, add a flight, total the cost). Clear definitions and logical thinking are essential to science. 3. There seems to be an implicit assumption that Alice wants to go somewhere. If she doesn’tspecify anydestination, the Cat may give anarbitrary route. -

Amount of Substance
www.CHEMSHEETS.co.uk AMOUNT OF SUBSTANCE © www.CHEMSHEETS.co.uk 28-July-2020 Chemsheets AS 1027 1 1 - FORMULAE If you are serious about doing A level Chemistry, you MUST be able to write a formula without a second thought. It is the single most essential skill for an A level chemist. You have to know and be able to use the information on this page – you should not be looking it up. There is no data sheet with ion charges at A level. If you can’t write a formula in an instant, DROP CHEMISTRY NOW and choose something else. Elements Monatomic Simple molecular Ionic Metallic Giant covalent helium hydrogen There are no ionic The formula is just the The formula is just the elements!! symbol, e.g. symbol neon nitrogen magnesium diamond argon oxygen iron graphite krypton fluorine sodium silicon xenon chlorine nickel radon bromine iodine phosphorus sulfur Compounds Monatomic Simple molecular Ionic Metallic Giant covalent There are no monatomic Some common These have to be There are no metallic silicon dioxide compounds!! molecular compounds: worked out using ion compounds!! charges – you have to carbon dioxide know these at AS/A carbon monoxide level! nitrogen monoxide LEARN them ASAP. nitrogen dioxide sulfur dioxide Note these acids: sulfur trioxide hydrochloric acid ammonia sulfuric acid methane nitric acid hydrogen sulfide phosphoric acid Positive ions Negative ions Group 1 ions: Group 3 ions: Group 7 ions: Other common ions lithium aluminium fluoride nitrate sodium chloride sulfate Other common ions potassium bromide carbonate silver iodide hydrogencarbonate Group 2 ions: zinc hydroxide magnesium Group 6 ions: ammonium hydride calcium oxide hydrogen phosphate barium sulfide © www.CHEMSHEETS.co.uk 28-July-2020 Chemsheets AS 1027 2 TASK 1 – WRITING FORMULAS OF IONIC COMPOUNDS 1) silver bromide …………………………. -

Product Stewardship Summary Hydrobromic Acid
1801 U.S. Highway 52 West West Lafayette, IN 47906 +1-765-497-6100 +1-800-428-7947 Product Stewardship Summary Hydrobromic Acid Hydrobromic acid is a versatile chemical that is used by a number of different industries. Among its many uses, hydrobromic acid is utilized as a flux material in soldering materials, as an activator to make certain plastics, and as a chemical intermediate. It is also a raw material for the manufacture of oil field salts, useful in food safety applications, and a reactant in the manufacture of pharmaceuticals. Hydrobromic acid is a liquid made by dissolving hydrogen bromide gas in water. Because the hydrogen bromide used to make hydrobromic acid is a pressurized gas, it has different hazards than this product and has its own Product Stewardship Summary. (For more information see the Product Stewardship Summary for Anhydrous Hydrogen Bromide). Hydrobromic acid offered by Great Lakes Solutions can be produced in various concentrations, but the most common contains 48% hydrogen bromide in water. It is a clear, near water-white to yellow colored liquid with a characteristic acrid odor. Chemical Identity: Hydrobromic acid is identified by several names, all of them referring to the same chemical product. These names include: • HBr (commonly 48% HBr) • CAS Number [10035-10-6] • Aqueous hydrogen bromide, aqHBr • Hydrogen bromide aqueous solution • Example: Hydrogen bromide, 48%; • Hydrobromic acid Production: The gaseous hydrogen bromide used to make hydrobromic acid is derived from either the burning of bromine and hydrogen in a specially designed furnace or as an off-gas from a bromination reaction where it is a co-product. -

Chapter 2, Section
Naming Simple Compounds Ionic Compounds Ionic compounds • consist of positive and negative ions. • have attractions called ionic bonds between positively and negatively charged ions. • have high melting and boiling points. • are solid at room temperature. Copyright © Houghton Mifflin Company. All rights reserved. 2–2 Salt is An Ionic Compound Sodium chloride or “table salt” is an example of an ionic compound. Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings Copyright © Houghton Mifflin Company. All rights reserved. 2–3 Naming Ionic Compounds with Two Elements To name a compound that contains two elements, • identify the cation and anion. • name the cation first followed by the name of the anion. Copyright © Houghton Mifflin Company. All rights reserved. 2–4 Binary Ionic Compounds Type (I) An ionic formula • consists of positively and negatively charged ions. • is neutral. • has charge balance. total positive charge = total negative charge The symbol of the metal is written first followed by the symbol of the nonmetal. Copyright © Houghton Mifflin Company. All rights reserved. 2–5 Charge Balance In MgCl2 In MgCl2, • a Mg atom loses two valence electrons. • two Cl atoms each gain one electron. • subscripts indicate the number of ions needed to give charge balance. Copyright © 2005 by Pearson Education, Inc. Publishing as Benjamin Cummings Copyright © Houghton Mifflin Company. All rights reserved. 2–6 Charge Balance in Na2S In Na2S. • two Na atoms lose one valence electron each. • one S atom gains two electrons. • subscripts show the number of ions needed to give Copyright © 2005 by Pearson Education, Inc. charge balance. Publishing as Benjamin Cummings Copyright © Houghton Mifflin Company. -

AP Chemistry Free Response
AP Chemistry Exam Reactions: Questions and Answers With the new format of the exam in 2007 and the availability of both questions and answers on the web at AP Central (http://apcentral.collegeboard.com:80/apc/public/courses/4606.html), I have determined not to update this page any longer. Please create an account as a teacher at AP Central and navigate to the full exams and scoring rubrics which are available back to 2003 Beginning in 2007, question 4 is no longer 5 out of 8 responses but rather three required responses. Also, in addition to writing the reactants and products, the equation must be balanced and there is a question about the chemical reaction. 2007 (a) A solution of sodium hydroxide is added to a solution of lead(II) nitrate. If 1.0 L volumes of 1.0 M solutions of sodium hydroxide and lead(II) nitrate are mixed together, now many moles of product(s) will be produced? Assume the reaction goes to completion. (b) Excess nitric acid is added to solid calcium carbonate. Briefly explain why statues made of marble (calcium carbonate) displayed outdoors in urban areas are deteriorating. (c) A solution containing silver(I) ion (an oxidixing agent) is mixed with a; solution containing iron(II) ion (a reducing agent). If the contents of the reaction mixture described above are filtered, what substance(s), if any, would remain on the filter paper? - 2+ → (a) (i) Balanced equation: 2OH + Pb Pb(OH)2 (s) (ii) The moles of each reactant are obtained by multiplying the volume times the molarity. -

Nitrogen Oxide - Wikipedia, the Free Encyclopedia Page 1 of 3
Nitrogen oxide - Wikipedia, the free encyclopedia Page 1 of 3 Nitrogen oxide From Wikipedia, the free encyclopedia Nitrogen oxide can refer to a binary compound of oxygen and nitrogen, or a mixture of such compounds: Contents ■ Nitric oxide (NO), nitrogen(II) oxide ■ Nitrogen dioxide (NO2), nitrogen(IV) oxide ■ 1 NOx ■ Nitrous oxide (N2O), nitrogen(I) oxide ■ 2 Derivatives ■ Nitrate radical (NO3), nitrogen(VI) oxide ■ 3 See also ■ Dinitrogen trioxide (N2O3), nitrogen(II,IV) oxide ■ 4 References ■ Dinitrogen tetroxide (N2O4), nitrogen(IV) oxide ■ Dinitrogen pentoxide (N2O5), nitrogen(V) oxide In atmospheric chemistry and air pollution and related fields, nitrogen oxides refers specifically to NOx [1][2] (NO and NO2). Only the first three of these compounds can be isolated at room temperature. N2O3, N2O4, and N2O5 all decompose rapidly at room temperature. Nitrate radical is very reactive. N2O is stable and rather unreactive at room temperature, while NO and NO2 are quite reactive but nevertheless quite stable when isolated. Dinitrogen trioxide, Nitric oxide, NO Nitrogen dioxide, NO2 Nitrous oxide, N2O N2O3 Dinitrogen tetroxide, Dinitrogen pentoxide, N2O4 N2O5 http://en.wikipedia.org/wiki/Nitrogen_oxide 11/2/2010 Nitrogen oxide - Wikipedia, the free encyclopedia Page 2 of 3 NOx Main article: NOx NOx (often written NOx) refers to NO and NO2. They are produced during combustion, especially at high temperature. These two chemicals are important trace species in Earth's atmosphere. In the troposphere, during daylight, NO reacts with partly oxidized organic species (or the peroxy radical) to form NO2, which is then photolyzed by sunlight to reform NO: NO + CH3O2 → NO2 + CH3O NO2 + sunlight → NO + O The oxygen atom formed in the second reaction then goes on to form ozone; this series of reactions is the main source of tropospheric ozone. -

S1303, Sodium Hydroxide, Pellets, FCC
Scientific Documentation S1303, Sodium Hydroxide, Pellets, FCC Not appropriate for regulatory submission. Please visit www.spectrumchemical.com or contact Tech Services for the most up‐to‐date information contained in this information package. Spectrum Chemical Mfg Corp 769 Jersey Avenue New Brunswick, NJ 08901 Phone 732.214.1300 Ver4.06 29.September.2021 S1303, Sodium Hydroxide, Pellets, FCC Table of Contents Product Specification Certificate of Analysis Sample(s) Safety Data Sheet (SDS) Certification of Current Good Manufacturing Practices (cGMP) Manufacturing Process Flowchart Source Statement BSE/TSE Statement Allergen Statement GMO Statement Melamine Statement Nitrosamine Statement Animal Testing Statement Organic Compliance Statement Shelf Life Statement Other Chemicals Statement General Label Information – Sample Label General Lot Numbering System Guidance Kosher Certificate Halal Certificate Specification for Sodium Hydroxide, Pellets, FCC (S1303) Item Number S1303 Item Sodium Hydroxide, Pellets, FCC CAS Number 1310-73-2 Molecular Formula NaOH Molecular Weight 40.00 MDL Number Synonyms Caustic Soda ; Soda Lye Test Specification Min Max ASSAY (TOTAL ALKALI as NaOH) 95.0 100.5 % ARSENIC (As) 3 mg/kg CARBONATE (as Na2CO3) 3.0 % INSOLUBLE SUBSTANCES & ORGANIC MATTER TO PASS TEST LEAD (Pb) 2 mg/kg MERCURY 0.1 mg/kg IDENTIFICATION TO PASS TEST CERTIFIED KOSHER CERTIFIED HALAL APPEARANCE EXPIRATION DATE DATE OF MANUFACTURE MONOGRAPH EDITION Certificate Of Analysis Item Number S1303 Lot Number 1JG0041 Item Sodium Hydroxide, -

Chemical Name Federal P Code CAS Registry Number Acutely
Acutely / Extremely Hazardous Waste List Federal P CAS Registry Acutely / Extremely Chemical Name Code Number Hazardous 4,7-Methano-1H-indene, 1,4,5,6,7,8,8-heptachloro-3a,4,7,7a-tetrahydro- P059 76-44-8 Acutely Hazardous 6,9-Methano-2,4,3-benzodioxathiepin, 6,7,8,9,10,10- hexachloro-1,5,5a,6,9,9a-hexahydro-, 3-oxide P050 115-29-7 Acutely Hazardous Methanimidamide, N,N-dimethyl-N'-[2-methyl-4-[[(methylamino)carbonyl]oxy]phenyl]- P197 17702-57-7 Acutely Hazardous 1-(o-Chlorophenyl)thiourea P026 5344-82-1 Acutely Hazardous 1-(o-Chlorophenyl)thiourea 5344-82-1 Extremely Hazardous 1,1,1-Trichloro-2, -bis(p-methoxyphenyl)ethane Extremely Hazardous 1,1a,2,2,3,3a,4,5,5,5a,5b,6-Dodecachlorooctahydro-1,3,4-metheno-1H-cyclobuta (cd) pentalene, Dechlorane Extremely Hazardous 1,1a,3,3a,4,5,5,5a,5b,6-Decachloro--octahydro-1,2,4-metheno-2H-cyclobuta (cd) pentalen-2- one, chlorecone Extremely Hazardous 1,1-Dimethylhydrazine 57-14-7 Extremely Hazardous 1,2,3,4,10,10-Hexachloro-6,7-epoxy-1,4,4,4a,5,6,7,8,8a-octahydro-1,4-endo-endo-5,8- dimethanonaph-thalene Extremely Hazardous 1,2,3-Propanetriol, trinitrate P081 55-63-0 Acutely Hazardous 1,2,3-Propanetriol, trinitrate 55-63-0 Extremely Hazardous 1,2,4,5,6,7,8,8-Octachloro-4,7-methano-3a,4,7,7a-tetra- hydro- indane Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]- 51-43-4 Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]-, P042 51-43-4 Acutely Hazardous 1,2-Dibromo-3-chloropropane 96-12-8 Extremely Hazardous 1,2-Propylenimine P067 75-55-8 Acutely Hazardous 1,2-Propylenimine 75-55-8 Extremely Hazardous 1,3,4,5,6,7,8,8-Octachloro-1,3,3a,4,7,7a-hexahydro-4,7-methanoisobenzofuran Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime 26419-73-8 Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime. -

The Energy Landscape Concept and Its Implications for Synthesis Planning
Pure Appl. Chem. 2014; 86(6): 883–898 Conference paper Martin Jansen* The energy landscape concept and its implications for synthesis planning Abstract: Synthesis of novel solids, in a chemical sense, is one of the spearheads of innovation in materials research. However, such an undertaking is substantially impaired by lack of control and predictability. We present a concept that points the way towards rational planning of syntheses in solid state and materials chemistry. The foundation of our approach is the representation of the whole material world, i.e., the known and not-yet-known chemical compounds, on an energy landscape, which implies information about the free energies of these configurations. From this it follows at once that all chemical compounds capable of existence (both thermodynamically stable and metastable ones) are already present in virtuo in this landscape. For the first step of synthesis planning, i.e., the identification of candidates that are capable of existence, we computa- tionally search the respective potential energy landscapes for (meta)stable structure candidates. Recently we have extended our techniques to finite temperatures and pressures and calculated phase diagrams, including metastable manifestations of matter, without resorting to any experimental pre-information. The conception developed is physically consistent, and its feasibility has been proven. Applying appropriate experimental tools has enabled us to realize, e.g., elusive Na3N, including almost all of its predicted polymorphs, many years after the predictions were published. Keywords: calculation of phase diagrams; global searches; inorganic materials synthesis; IUPAC Congress-44; local ergodicity; solid state chemistry; structure prediction; synthesis from atoms. DOI 10.1515/pac-2014-0212 Introduction The lines of action called “analysis” and “synthesis” continue to constitute the foundation of chemistry, providing it with genuine and distinct characteristics in a time of increasing overlap among the various dis- ciplines of natural sciences. -

A Review of the Toxicity and Environmental Behaviour of Hydrogen Bromide In
w w w.environment-agency.gov.uk A Review of the Toxicity and Environmental Behaviour of Hydrogen Bromide in Air The Environment Agency is the leading public body protecting and improving the environment in England and Wales. It’s our job to make sure that air, land and water are looked after by everyone in today’s society, so that tomorrow’s generations inherit a cleaner, healthier world. Our work includes tackling flooding and pollution incidents, reducing industry’s impacts on the environment, cleaning up rivers, coastal waters and contaminated land, and improving wildlife habitats. This report is the result of research commissioned and funded by the Environment Agency’s Science Programme. Published by: Author(s): Environment Agency, Rio House, Waterside Drive, Aztec West, P Coleman, R Mascarenhas and P Rumsby Almondsbury, Bristol, BS32 4UD Tel: 01454 624400 Fax: 01454 624409 Dissemination Status: www.environment-agency.gov.uk Publicly available ISBN: 1844323552 Keywords: hydrogen bromide, inhalation toxicity, air quality, exposure, © Environment Agency January 2005 human health, air pollutant All rights reserved. This document may be reproduced with prior Research Contractor: permission of the Environment Agency. Netcen, Culham Science Centre, Culham, Abingdon, Oxfordshire, OX14 3ED The views expressed in this document are not necessarily WRc-NSF Ltd, Henley Road, Medmenham, Marlow, Bucks, those of the Environment Agency. SL7 2HD Tel: 0870 190 6437 Fax: 0870 190 6608 This report is printed on Cyclus Print, a 100% recycled stock, Website: www.netcen.co.uk. which is 100% post consumer waste and is totally chlorine free. Water used is treated and in most cases returned to source in Environment Agency’s Project Manager: better condition than removed. -

Acutely / Extremely Hazardous Waste List
Acutely / Extremely Hazardous Waste List Federal P CAS Registry Acutely / Extremely Chemical Name Code Number Hazardous 4,7-Methano-1H-indene, 1,4,5,6,7,8,8-heptachloro-3a,4,7,7a-tetrahydro- P059 76-44-8 Acutely Hazardous 6,9-Methano-2,4,3-benzodioxathiepin, 6,7,8,9,10,10- hexachloro-1,5,5a,6,9,9a-hexahydro-, 3-oxide P050 115-29-7 Acutely Hazardous Methanimidamide, N,N-dimethyl-N'-[2-methyl-4-[[(methylamino)carbonyl]oxy]phenyl]- P197 17702-57-7 Acutely Hazardous 1-(o-Chlorophenyl)thiourea P026 5344-82-1 Acutely Hazardous 1-(o-Chlorophenyl)thiourea 5344-82-1 Extemely Hazardous 1,1,1-Trichloro-2, -bis(p-methoxyphenyl)ethane Extemely Hazardous 1,1a,2,2,3,3a,4,5,5,5a,5b,6-Dodecachlorooctahydro-1,3,4-metheno-1H-cyclobuta (cd) pentalene, Dechlorane Extemely Hazardous 1,1a,3,3a,4,5,5,5a,5b,6-Decachloro--octahydro-1,2,4-metheno-2H-cyclobuta (cd) pentalen-2- one, chlorecone Extemely Hazardous 1,1-Dimethylhydrazine 57-14-7 Extemely Hazardous 1,2,3,4,10,10-Hexachloro-6,7-epoxy-1,4,4,4a,5,6,7,8,8a-octahydro-1,4-endo-endo-5,8- dimethanonaph-thalene Extemely Hazardous 1,2,3-Propanetriol, trinitrate P081 55-63-0 Acutely Hazardous 1,2,3-Propanetriol, trinitrate 55-63-0 Extemely Hazardous 1,2,4,5,6,7,8,8-Octachloro-4,7-methano-3a,4,7,7a-tetra- hydro- indane Extemely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]- 51-43-4 Extemely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]-, P042 51-43-4 Acutely Hazardous 1,2-Dibromo-3-chloropropane 96-12-8 Extemely Hazardous 1,2-Propylenimine P067 75-55-8 Acutely Hazardous 1,2-Propylenimine 75-55-8 Extemely Hazardous 1,3,4,5,6,7,8,8-Octachloro-1,3,3a,4,7,7a-hexahydro-4,7-methanoisobenzofuran Extemely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime 26419-73-8 Extemely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime. -

Hydrogen Bromide (CAS #10035-10-6) Information for the Public
Revised June 2005 Hydrogen Bromide (CAS #10035-10-6) Information for the Public What is hydrogen bromide? Hydrogen bromide is a colorless, or sometimes faint yellow, highly toxic gas with a sharp, irritating odor. It can also be found as a liquid, either as hydrobromic acid (hydrogen bromide dissolved in water) or as a compressed gas under pressure (anhydrous hydrogen bromide). It is an extremely dangerous substance and must be handled with caution as it can cause severe health effects and death. Hydrogen bromide can react violently when mixed with some chemicals, metals or water, forming a flammable, explosive gas. When hydrogen bromide is released into indoor air, dangerous levels will be reached very quickly. The gas is heavier than air and can travel to low- lying or confined areas. Containers of hydrogen bromide may explode when heated. How is hydrogen bromide used? Hydrogen bromide is used to make chemicals and drugs, as a solvent and as a veterinary drug. Before working with hydrogen bromide, employee training should be provided on proper safe handling and storage procedures. How can people be exposed to hydrogen bromide? Significant exposure usually occurs in the industries where hydrogen bromide is produced or used. Heating, pouring, spraying, spills and evaporation create conditions that increase the risk of employee exposure. Though unlikely, the general population may be exposed by breathing contaminated air or by drinking contaminated water from a facility using or storing hydrogen bromide, by skin or eye contact with the gas or liquid, or by eating food that has been contaminated with hydrogen bromide.