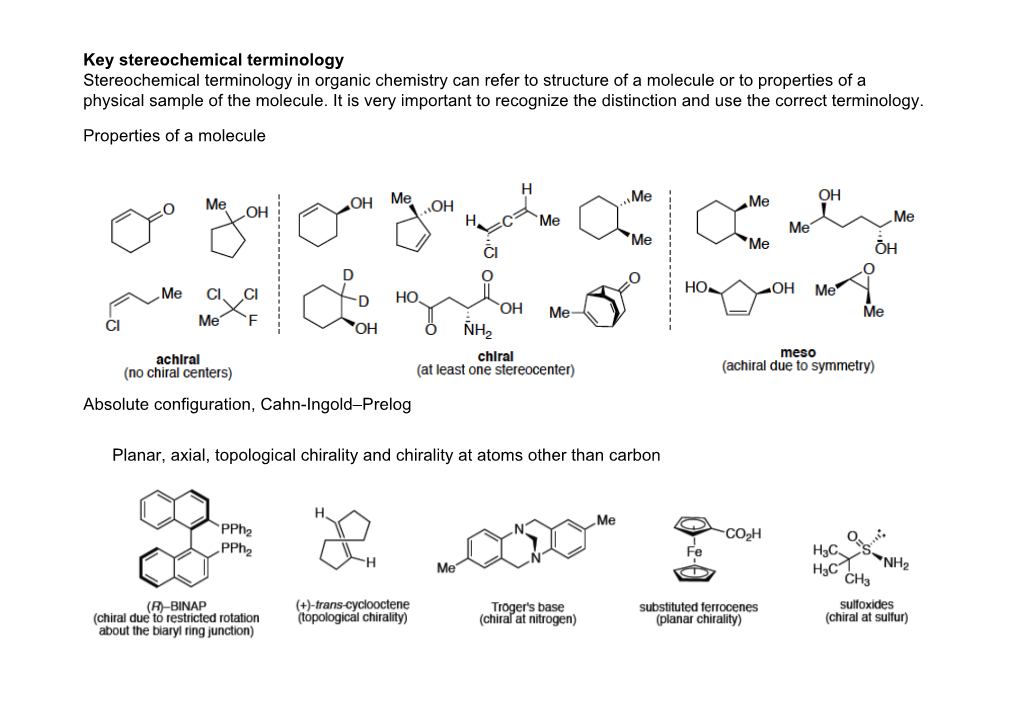
Stereoselective Reactions Substrate Controlled Diastereoselective Reactions Cyclic Stereocontrol, Acyclic Stereocontrol (I.E
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Chiral Separation of Rac-Propylene Oxide on Penicillamine Coated Gold Nps
nanomaterials Article Chiral Separation of rac-Propylene Oxide on Penicillamine Coated Gold NPs Nisha Shukla 1,2, Zachary Blonder 2 and Andrew J. Gellman 2,3,* 1 Institute of Complex Engineered Systems, Carnegie Mellon University, Pittsburgh, PA 15213, USA; [email protected] 2 Department of Chemical Engineering, Carnegie Mellon University, Pittsburgh, PA 15213, USA; [email protected] 3 W.E. Scott Institute for Energy Innovation, Carnegie Mellon University, Pittsburgh, PA 15213, USA * Correspondence: [email protected] Received: 16 July 2020; Accepted: 20 August 2020; Published: 30 August 2020 Abstract: The surfaces of chemically synthesized spherical gold NPs (Au-NPs) have been modified using chiral L- or D-penicillamine (Pen) in order to impart enantioselective adsorption properties. These chiral Au-NPs have been used to demonstrate enantioselective adsorption of racemic propylene oxide (PO) from aqueous solution. In the past we have studied enantioselective adsorption of racemic PO on L- or D-cysteine (Cys)-coated Au-NPs. This prior work suggested that adsorption of PO on Cys-coated Au-NPs equilibrates within an hour. In this work, we have studied the effect of time on the enantioselective adsorption of racemic PO from solution onto chiral Pen/Au-NPs. Enantioselective adsorption of PO on chiral Pen/Au-NPs is time-dependent but reaches a steady state after ~18 h at room temperature. More importantly, L- or D-Pen/Au-NPs are shown to adsorb R- or S-PO enantiospecifically and to separate the two PO enantiomers from racemic mixtures of RS-PO. Keywords: chiral; nanoparticles; enantioselective adsorption; penicillamine; cysteine 1. Introduction Chirality has attracted enormous interest in the field of chemistry due to the enantiospecific responses of living organisms to the two enantiomers of chiral compounds that they have ingested. -

Prebiological Evolution and the Metabolic Origins of Life
Prebiological Evolution and the Andrew J. Pratt* Metabolic Origins of Life University of Canterbury Keywords Abiogenesis, origin of life, metabolism, hydrothermal, iron Abstract The chemoton model of cells posits three subsystems: metabolism, compartmentalization, and information. A specific model for the prebiological evolution of a reproducing system with rudimentary versions of these three interdependent subsystems is presented. This is based on the initial emergence and reproduction of autocatalytic networks in hydrothermal microcompartments containing iron sulfide. The driving force for life was catalysis of the dissipation of the intrinsic redox gradient of the planet. The codependence of life on iron and phosphate provides chemical constraints on the ordering of prebiological evolution. The initial protometabolism was based on positive feedback loops associated with in situ carbon fixation in which the initial protometabolites modified the catalytic capacity and mobility of metal-based catalysts, especially iron-sulfur centers. A number of selection mechanisms, including catalytic efficiency and specificity, hydrolytic stability, and selective solubilization, are proposed as key determinants for autocatalytic reproduction exploited in protometabolic evolution. This evolutionary process led from autocatalytic networks within preexisting compartments to discrete, reproducing, mobile vesicular protocells with the capacity to use soluble sugar phosphates and hence the opportunity to develop nucleic acids. Fidelity of information transfer in the reproduction of these increasingly complex autocatalytic networks is a key selection pressure in prebiological evolution that eventually leads to the selection of nucleic acids as a digital information subsystem and hence the emergence of fully functional chemotons capable of Darwinian evolution. 1 Introduction: Chemoton Subsystems and Evolutionary Pathways Living cells are autocatalytic entities that harness redox energy via the selective catalysis of biochemical transformations. -

Experiment 4
Experiment Limiting Reactant 5 A limiting reactant is the reagent that is completely consumed during a chemical reaction. Once this reagent is consumed the reaction stops. An excess reagent is the reactant that is left over once the limiting reagent is consumed. The maximum theoretical yield of a chemical reaction is dependent upon the limiting reagent thus the one that produces the least amount of product is the limiting reagent. For example, if a 2.00 g sample of ammonia is mixed with 4.00 g of oxygen in the following reaction, use stoichiometry to determine the limiting reagent. 4NH3 + 5O2 4NO + 6H2O Since the 4.00 g of O2 produced the least amount of product, O2 is the limiting reagent. In this experiment you will be given a mixture of two ionic solids, AgNO3 and K2CrO4, that are both soluble in water. When the mixture is dissolved into water they will react to form an insoluble compound, Ag2CrO4, which can be collected and weighed. The reaction is the following: 2 AgNO3(aq) + K2CrO4(aq) Ag2CrO4(s) + 2 KNO3(aq) Colorless Yellow Red colorless Precipitate Precipitate is the term used for an insoluble solid which is produced by mixing two solutions. Often the solid particles are so fine that they will pass right through the pores of a filter. Heating the solution for a while causes the particles to clump together, a process called digesting. The precipitate coagulates upon cooling and eventually settles to the bottom of a solution container. The clear liquid above the precipitate is called the supernatant. -

Chapter 3 • Ratio: Cnh2n+2
Organic Chemistry, 5th Edition Alkane Formulas L. G. Wade, Jr. • All C-C single bonds • Saturated with hydrogens Chapter 3 • Ratio: CnH2n+2 Structure and Stereochemistry • Alkane homologs: CH3(CH2)nCH3 of Alkanes • Same ratio for branched alkanes H H H H H H CH H C C C C H H H H C CCH Jo Blackburn H H H H H H Richland College, Dallas, TX H => Butane, C4H10 Dallas County Community College District Chapter 3Isobutane, C H 2 © 2003, Prentice Hall 4 10 Common Names Pentanes H H H H H H H CH • Isobutane, “isomer of butane” H C C C C C H H H • Isopentane, isohexane, etc., methyl H H H H H H H C CCC H branch on next-to-last carbon in chain. H H H H • Neopentane, most highly branched n-pentane, C5H12 isopentane, C5H12 • Five possible isomers of hexane, CH3 18 isomers of octane and 75 for H3C C CH3 decane! CH3 => => neopentane, C5H12 Chapter 3 3 Chapter 3 4 Longest Chain IUPAC Names • The number of carbons in the longest • Find the longest continuous carbon chain determines the base name: chain. ethane, hexane. (Listed in Table 3.2, • Number the carbons, starting closest to page 81.) the first branch. • If there are two possible chains with the • Name the groups attached to the chain, same number of carbons, use the chain using the carbon number as the locator. with the most substituents. • Alphabetize substituents. H3C CH CH CH • Use di-, tri-, etc., for multiples of same 2 3 CH3 substituent. -

Review of Calculations for Organic Reactions (Assignment #1B)
Review of Calculations for Organic Reactions (Assignment #1b) In your experiments the quantities of many reactants are given in mass or volume, though chemicals react in mole ratios, because it is not possible to measure a quantity in moles easily. You will have to convert mass or volume (mass = volume x density) to moles before beginning an experiment. You will also have to recognize stoichiometric relationships between reactants and products, which is based on mole ratio, in order to be able to calculate the theoretical yield of product you expect to isolate. You will apply the knowledge from this session to complete the theoretical yield calculation in your prelab reports and postlab reports. In this lab you are expected to be able to differentiate between a limiting reagent and excess reagents, solvents and catalysts which are crucial for the reaction to occur and go to completion. A limiting reagent is a reactant that has the lowest number of moles of all reactants in the chemical reaction and once it is completely consumed the reaction terminates. An excess reagent is a reactant that has a higher number of moles and therefore is not used up when a reaction goes to completion. Solvents and catalysts are not involved in the determination of limiting reagent. An example of calculations that you will be expected to perform is shown using the reaction of phenol with nitric acid. Preparation of picric acid requires the nitration of phenol. Given 5.00 grams of phenol and 15.0 mL concentrated nitric acid (15.9 M), one can determine the MAXIMUM theoretical amount of picric acid formed. -

From Synthetic Chemistry and Stereoselective Biotransformations
PP Periodica Polytechnica From Synthetic Chemistry and Chemical Engineering Stereoselective Biotransformations to Enzyme Biochemistry – The Bioorganic Chemistry Group at the Budapest 59(1), pp. 59-71, 2015 University of Technology and Economics DOI: 10.3311/PPch.7390 Creative Commons Attribution b Zoltán BOROS1, Gábor HORNYÁNSZKY1, József NAGY1, László POPPE1 * research article Received 04 March 2014; accepted after revision 05 May 2014 Abstract 1 Introduction The activity of Bioorganic Chemistry Group (BCG) within 1.1 Scientific background of the Bioorganic Chemistry Department of Organic Chemistry and Technology at Budapest Research Group University of Technology and Economics is related to various The activity of Bioorganic Chemistry Group (BCG) of areas of synthetic chemistry, biotechnology and enzymology. Department of Organic Chemistry and Technology at Budapest This review gives an overview on the research activity of the University of Technology and Economics is related to various group covering development of synthetic organic chemistry areas of synthetic chemistry, selective biocatalysis [1] and methods; stereoselective biotransformations with lipases, enzymology with major emphasis on development of novel ammonia-lyases and further biocatalysts in batch and tools for stereoselective synthesis [2]. continuous-flow reactions; novel enzyme immobilization One of the main challenges facing organic chemistry is methods; and enzyme structural and mechanistic studies by the rational synthesis of an ever growing number of complex, experimental and computational techniques. optically active natural products and their analogues [3]. According to the regulation of FDA production of chiral Keywords drugs, agrochemicals, fine chemicals has been allowed in synthetic organic chemistry, stereoselective biotransformation, enantiomerically pure form, because it often happens that only continuous-flow reaction, lipase, ammonia-lyase, enzyme one of the two enantiomers shows the required therapeutical immobilization, enzyme structure, enzyme mechanism, QM/ effect [4]. -
Chiral Auxiliaries and Optical Resolving Agents
Chiral Auxiliaries and Optical Resolving Agents Most bioactive substances are optically active. For instance, if This brochure introduces a variety of chiral auxiliaries and a substance is synthesized as a racemic compound, its optical resolving agents. We hope that it will be useful for your enantiomer may show no activity or even undesired bioactivity. research of the synthesis of optically active compounds. Thus, methods to gain enantiopure compounds have been Additionally, TCI has some brochures introducing chiral developed. When synthesizing enantiopure compounds, the compounds for the chiral pool method in “Chiral Building Blocks”, methods are roughly divided into three methods. “Terpenes”, “Amino Acids” and other brochures. Sugar derivatives are also introduced in a catalog, “Reagents for Glyco Chemistry Chiral pool method: & Biology”, and category pages of sugar chains. Furthermore, The method using an easily available chiral compound as a TCI has many kinds of catalysts for asymmetric synthesis and starting material like an amino acid or sugar. introduce them in brochures such as “Asymmetric Synthesis” and Asymmetric synthesis: “Asymmetric Organocatalysts”, and other contents. The method to introduce an asymmetric point to compounds You can search our information through “asymmetric synthesis” without an asymmetric point. Syntheses using achiral as a keyword. auxiliaries are included here. Optical resolution: The method to separate a racemic compound into two ● Reactions with Chiral Auxiliaries enantiomers. The direct method using a chiral column and One of the most famous named reactions using chiral auxiliaries1) the indirect method to separate two enantiomers using is the Evans aldol reaction.2) This reaction is quite useful because optical resolving agents to convert into diastereomers are this reaction can efficiently introduce two asymmetric carbons into examples. -

Separation of the Mixtures of Chiral Compounds by Crystallization
1 Separation of the Mixtures of Chiral Compounds by Crystallization Emese Pálovics2, Ferenc Faigl1,2 and Elemér Fogassy1* 1Department of Organic Chemistry and Technology, Budapest University of Technology and Economics, 2Research Group for Organic Chemical Technology, Hungarian Academy of Sciences, Budapest, Hungary 1. Introduction Reaction of a racemic acid or base with an optically active base or acid gives a pair of diastereomeric salts. Members of this pair exhibit different physicochemical properties (e.g., solubility, melting point, boiling point, adsorbtion, phase distribution) and can be separated owing to these differences. The most important method for the separation of enantiomers is the crystallization. This is the subject of this chapter. Preparation of enantiopure (ee~100%) compounds is one of the most important aims both for industrial practice and research. Actually, the resolution of racemic compounds (1:1 mixture of molecules having mirror-imagine relationship) still remains the most common method for producing pure enantiomers on a large scale. In these cases the enantiomeric mixtures or a sort of their derivatives are separated directly. This separation is based on the fact that the enantiomeric ratio in the crystallized phase differs from the initial composition. In this way, obtaining pure enantiomers requires one or more recrystallizations. (Figure 1). The results of these crystallizations (recrystallizations) of mixtures of chiral compounds differ from those observed at the achiral compounds. Expectedly, not only the stereoisomer in excess can be crystallized, because the mixture of enantiomers (with mirror image relationship) follows the regularities established from binary melting point phase diagrams, and ternary composition solubility diagrams, respectively, as a function of the starting enantiomer proportion. -

II. Stereochemistry 5
B.Sc.(H) Chemistry Semester - II Core Course - III (CC-III) Organic Chemistry - I II. Stereochemistry 5. Physical and Chemical Properties of Stereoisomers Dr. Rajeev Ranjan University Department of Chemistry Dr. Shyama Prasad Mukherjee University, Ranchi 1 Syllabus & Coverage Syllabus II Stereochemistry: Fischer Projection, Newmann and Sawhorse Projection formulae and their interconversions. Geometrical isomerism: cis–trans and syn-anti isomerism, E/Z notations with Cahn Ingold and Prelog (CIP) rules for determining absolute configuration. Optical Isomerism: Optical Activity, Specific Rotation, Chirality/Asymmetry, Enantiomers, Molecules with two or more chiral-centres, Distereoisomers, Meso structures, Racemic mixture. Resolution of Racemic mixtures. Relative and absolute configuration: D/L and R/S designations. Coverage: 1. Types of Isomers : Comparing Structures 2. Optical Activity 3. Racemic Mixtures : Separation of Racemic Mixtures 4. Enantiomeric Excess and Optical Purity 5. Relative and Absolute Configuration 6. Physical and Chemical Properties of Stereoisomers 2 Stereochemistry Types of Isomers Dr. Rajeev Ranjan 3 Stereochemistry Determining the Relationship Between Two Non-Identical Molecules Dr. Rajeev Ranjan 4 Stereochemistry Comparing Structures: Are the structures connected the same? yes no Are they mirror images? Constitutional Isomers yes no Enantiomers Enantiomers Is there a plane of symmetry? All chiral centers will be opposite between them. yes no Meso Diastereomers superimposable Dr. Rajeev Ranjan 5 Stereochemistry Optical Activity: • The chemical and physical properties of two enantiomers are identical except in their interaction with chiral substances. • The physical property that differs is the behavior when subjected to plane-polarized light ( this physical property is often called an optical property). • Plane-polarized (polarized) light is light that has an electric vector that oscillates in a single plane. -

Effect of the Enantiomeric Ratio of Eutectics on the Results and Products of the Reactions Proceeding with the Participation Of
Perspective Effect of the Enantiomeric Ratio of Eutectics on the Results and Products of the Reactions Proceeding with the Participation of Enantiomers and Enantiomeric Mixtures Emese Pálovics *, Dorottya Fruzsina Bánhegyi and Elemér Fogassy Department of Organic Chemistry and Technology, Budapest University of Technology and Economics, 1521 Budapest, Hungary; [email protected] (D.F.B.); [email protected] (E.F.) * Correspondence: [email protected]; Tel.: +36-1-4632101 Received: 3 August 2020; Accepted: 14 September 2020; Published: 21 September 2020 Abstract: This perspective is focused on the main parameters determining the results of crystallization of enantiomers or enantiomeric mixtures. It was shown that the ratio of supramolecular and helical associations depends on the eutectic composition of the corresponding enantiomeric mixture. The M and P ratios together with the self-disproportionation (SDE) of enantiomers define the reaction of the racemic compound with the resolving agent. Eventually, each chiral molecule reacts with at least two conformers with different degrees of M and P helicity. The combined effect of the configuration, charge distribution, constituent atoms, bonds, flexibility, and asymmetry of the molecules influencing their behavior was also summarized. Keywords: chiral molecules; enantiomeric separation; self-disproportionation of enantiomers; supramolecular associations; helical structure; eutectic composition 1. Introduction The preparation of asymmetric (chiral) compounds (enantiomers) is one of the main goals of research, industry and medicine. The preparation of a given enantiomer is possible in several ways, for example by selective synthesis, which requires the separation of the chiral catalyst or the enantiomers of the racemic compound, and in which case a resolving agent (like a chiral catalyst) may be required. -

Enantioselective Synthesis of the (1S,5R)-Enantiomer of Litseaverticillols a and B
Biosci. Biotechnol. Biochem., 70 (10), 2564–2566, 2006 Note Enantioselective Synthesis of the (1S,5R)-Enantiomer of Litseaverticillols A and B y Akira MORITA, Hiromasa KIYOTA, and Shigefumi KUWAHARA Laboratory of Applied Bioorganic Chemistry, Graduate School of Agricultural Science, Tohoku University, Tsutsumidori-Amamiyamachi, Aoba-ku, Sendai 981-8555, Japan Received May 8, 2006; Accepted May 31, 2006; Online Publication, October 7, 2006 [doi:10.1271/bbb.60253] An enantioselective synthesis of the (1S,5R)-enan- which have recently culminated in the first enantiose- tiomer of litseaverticillols A and B was accomplished in lective total synthesis of (1R,5S)-(À)-1 and (1R,5S)-(À)- line with our previously reported synthetic pathway for 2.4) In this note, we describe the synthesis of their their (1R,5S)-enantiomer. The use of ‘‘EtSCeCl2’’ pre- enantiomers directed toward an evaluation of the differ- pared from EtSLi and CeCl3, instead of previously ence in biological activity between the enantiomers of employed EtSLi itself, for the formation of thiol ester litseaverticillols A and B. intermediates prevented any undesirable epimerization Our synthesis of (1S,5R)-1 and (1S,5R)-2 basically occurring in the process. followed our previous synthesis of their corresponding enantiomers, (1R,5S)-1 and (1R,5S)-2, respectively,4) Key words: litseaverticillol; enantioselective synthesis; except that (R)-4-benzyloxazolidin-2-one was employed sesquiterpenoid; anti-HIV as the source of chirality, instead of its (S)-form used in the previous synthesis. Homogeranic -

Lecture 3: Stereochemistry and Drugs Key Objectives: 1
Lecture 3: Stereochemistry and drugs Key objectives: 1. Be able to explain the role of stereochemistry in drug action or metabolism 2. Be able to identify a chiral center (or centers) in a drug 3. Be able to explain the difference between enantiomers and diastereomers Value of chirality and stereochemistry: Chirality as expressed through stereoisomers increases the specificity of molecule recognition and signaling. Like a key in a lock, or a hand in a glove. Some useful definitions: Chirality (and chiral centers): An object (such as a molecule) that is asymmetric and therefore not superimposable on its own image. Chiral centers are the most common features within molecules that give rise to chirality. Stereoisomer: A molecule that possesses at least one chiral center (or center of chirality). Enantiomers: A type of stereoisomer that exists in mirror image forms. See Figure 1. Diastereomers: A type of stereoisomer that possesses more than one chiral center. Stereospecific: A term used to describe a stereochemical property that one isomer possesses but the other does not. For example, the stereospecific metabolism of the S-enantiomer of a compound by an enzyme but the R-enantiomer is not metabolized by that enzyme. Stereoselective: A term used to describe a stereochemical property that is shared by both stereoisomers, but the property is greater in one isomer than the other. For example, the stereoselective binding of the R-enantiomer for a receptor indicates that the S-enantiomer also binds but not as avidly as the R-enantiomer. Enantiomers Figure 1: For enantiomers, many times we use the example of our left and right hands to demonstrate their asymmetry.