Development of a Thyroperoxidase Inhibition Assay for High- Throughput Screening † ‡ ‡ ∥ § ∥ Katie B
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Peroxidase and NADPH-Cytochrome C Reductase Activity During Thyroid Hyperplasia and Involution
Peroxidase and NADPH-Cytochrome C Reductase Activity During Thyroid Hyperplasia and Involution KUNIHIRO YAMAMOTO AND LESLIE J. DEGROOT Thyroid Study Unit, Department of Medicine, University of Chicago, Chicago, Illinois 60637 ABSTRACT. The regulation of iodination was in- TSH injection, whether expressed per mg gland vestigated in male rats during physiological altera- weight, per mg protein, or per fxg DNA, suggesting tions in thyroid function. Thyroid hyperplasia was enzyme induction and cellular hypertrophy. Involu- Downloaded from https://academic.oup.com/endo/article/95/2/606/2618917 by guest on 30 September 2021 produced by giving 0.01% PTU in drinking water or tion by T4 administration caused a decrease in injection of TSH (2 USP U/day); involution was thyroid weight, DNA content, and enzyme activity per induced after PTU treatment by giving 3 fig L-T4/ml gland. The main reason for the decrease in enzyme in drinking water. Increase in activity of thyroid activity per gland was a diminution of cell numbers. peroxidase and NADPH-cytochrome c reductase per During thyroid hyperplasia and involution, perox- gland exceeded gains in thyroid weight and DNA idase and NADPH-cytochrome c reductase activity content early in hyperplasia, but increased essen- is regulated by TSH. During the onset of TSH tially in parallel manner during chronic PTU treat- action, peroxidase and NADPH-cytochrome c re- ment. Enzyme activity per /xg DNA increased to ductase increase to a greater extent than thyroid 155% of control after 4 days of PTU treatment, weight and DNA content, suggesting preferential decreased to 138% on the seventh day, and was at enzyme synthesis in addition to cell hypertrophy. -

Thyroid Peroxidase Antibody Is Associated with Plasma Homocysteine Levels in Patients with Graves’ Disease
Published online: 2018-07-02 Article Thieme Li Fang et al. Thyroid Peroxidase Antibody is … Exp Clin Endocrinol Diabetes 2018; 00: 00–00 Thyroid Peroxidase Antibody is Associated with Plasma Homocysteine Levels in Patients with Graves’ Disease Authors Fang Li1 * , Gulibositan Aji1 * , Yun Wang2, Zhiqiang Lu1, Yan Ling1 Affiliations ABSTRACT 1 Department of Endocrinology and Metabolism, Zhong- Purpose Homocysteine is associated with cardiovascular, shan Hospital, Fudan University, Shanghai, China inflammation and autoimmune diseases. Previous studies have 2 Department of Endocrinology and Metabolism, the shown that thyroid peroxidase antibody is associated with ho- Second Hospital of Shijiazhuang City, Shijiazhuang, mocysteine levels in hypothyroidism. The relationship between Hebei Province, China thyroid antibodies and homocysteine in hyperthyroidism re- mains unclear. In this study, we aimed to investigate the asso- Key words ciation of thyroid antibodies with homocysteine in patients human, cardiovascular risk, hyperthyroidism with Graves’ disease. Methods This was a cross-sectional study including 478 received 07.04.2018 Graves’ disease patients who were consecutively admitted and revised 10.05.2018 underwent radioiodine therapy. Homocysteine, thyroid hor- accepted 14.06.2018 mones, thyroid antibodies, glucose and lipids were measured. Results Patients with homocysteine levels above the median Bibliography were older and had unfavorable metabolic parameters com- DOI https://doi.org/10.1055/a-0643-4692 pared to patients with homocysteine levels below the median. Published online: 2.7.2018 Thyroglobulin antibody or thyroid peroxidase antibody was Exp Clin Endocrinol Diabetes 2020; 128: 8–14 associated with homocysteine levels (β = 0.56, 95 %CI 0.03- © J. A. Barth Verlag in Georg Thieme Verlag KG Stuttgart · 1.08, p = 0.04; β = 0.75, 95 %CI 0.23-1.27, p = 0.005). -

Thyroid Surgery for Patients with Hashimoto's Disease
® Clinical Thyroidology for the Public VOLUME 12 | ISSUE 7 | JULY 2019 HYPOTHYROIDISM Thyroid surgery for patients with Hashimoto’s disease BACKGROUND SUMMARY OF THE STUDY Hypothyroidism, or an underactive thyroid, is a common This study enrolled patients with hypothyroidism due to problem. In the United States, the most common cause Hashimoto’s thyroiditis who received treatment with thy- of hypothyroidism is Hashimoto’s thyroiditis. This is an roidectomy and thyroid hormone replacement or thyroid autoimmune disorder where antibodies attack the thyroid, hormone replacement alone. The outcome of the study causing inflammation and destruction of the gland. Char- was a patient-reported health score on the generic Short acteristic of Hashimoto’s thyroiditis are high antibodies Form-36 Health Survey (SF-36) after 18 months. to thyroid peroxidase (TPO Ab) on blood tests. Hypo- thyroidism is treated by thyroid hormone and returning Patients were in the age group of 18 to 79 years. They all thyroid hormone levels to the normal range usually had a TPOAb titer >1000 IU/L and reported persistent resolves symptoms in most patients. symptoms despite having normal thyroid hormone levels based on blood tests. Typical symptoms included fatigue, However, in some patients, symptoms may persist despite increased need for sleep associated with reduced sleep what appears to be adequate treatment based on blood quality, joint and muscle tenderness, dry mouth, and dry tests of thyroid function. This raises the possibility that eyes. Follow up visits were done every 3 months for 18 some symptoms may be related to the autoimmune months and the thyroid hormone therapy was adjusted as condition itself. -

How Useful Are Autoantibodies in Diagnosing Thyroid Disorders?
Evidence Based Answers CLINIcAL INQUiRiES from the Family Physicians Inquiries Network Heather Downs, DO, How useful are autoantibodies and Albert A. Meyer, MD New Hanover Regional Medical in diagnosing thyroid disorders? Center, Wilmington, NC Donna Flake, MSLS, MSAS Coastal Area Health Education Evidence-based answer Center, Wilmington, NC They’re useful in diagnosing Graves’ increases or decreases the probability disease and, to a lesser extent, of autoimmune thyroid disease by only autoimmune thyroid disease; they can also a small to moderate degree (SOR: B, 3 help predict hypothyroidism. Thyrotropin cross-sectional studies). receptor antibodies (TRAb) may be mildly Thyroid-stimulating hormone (TSH) elevated in a variety of thyroid disorders, levels >2 mU/L, although still in the normal but a TRAb level >10 U/L increases range, can be followed up with TPOAb ® the probability of Graves’ disease by Dowdentesting to determine Health whether Mediathe patient a moderate to large degree (strength has an increased probability of developing of recommendation [SORCopyright]: B, cross- hypothyroidism (SOR: B, cohort study sectional study). A positive or negativeFor personalwith a vague hypothyroidism use only reference thyroid peroxidase antibody (TPOAb) test standard). Clinical commentary FAST TRACK In equivocal situations and pregnancy, infiltrative disorders, Reidel’s thyroiditis, antibodies may help or subacute granulomatous thyroiditis are Thyroid Under most circumstances, hypo- and suspected. TPOAb may help predict the autoantibodies hyperthyroid disorders can be diagnosed development of clinical hypothyroidism in can help diagnose by testing TSH and free T , without thyroid patients with TSH in the range of 5-10 mU/L. 4 Graves’ disease antibody testing. Radionuclide uptake and Pregnancy-related hyperthyroidism scan provide diagnostic information for requires antibody testing because and autoimmune hyperthyroid states. -

NNT Mutations: a Cause of Primary Adrenal Insufficiency, Oxidative Stress and Extra- Adrenal Defects
175:1 F Roucher-Boulez and others NNT, adrenal and extra-adrenal 175:1 73–84 Clinical Study defects NNT mutations: a cause of primary adrenal insufficiency, oxidative stress and extra- adrenal defects Florence Roucher-Boulez1,2, Delphine Mallet-Motak1, Dinane Samara-Boustani3, Houweyda Jilani1, Asmahane Ladjouze4, Pierre-François Souchon5, Dominique Simon6, Sylvie Nivot7, Claudine Heinrichs8, Maryline Ronze9, Xavier Bertagna10, Laure Groisne11, Bruno Leheup12, Catherine Naud-Saudreau13, Gilles Blondin13, Christine Lefevre14, Laetitia Lemarchand15 and Yves Morel1,2 1Molecular Endocrinology and Rare Diseases, Lyon University Hospital, Bron, France, 2Claude Bernard Lyon 1 University, Lyon, France, 3Pediatric Endocrinology, Gynecology and Diabetology, Necker University Hospital, Paris, France, 4Pediatric Department, Bab El Oued University Hospital, Alger, Algeria, 5Pediatric Endocrinology and Diabetology, American Memorial Hospital, Reims, France, 6Pediatric Endocrinology, Robert Debré Hospital, Paris, France, 7Department of Pediatrics, Rennes Teaching Hospital, Rennes, France, 8Pediatric Endocrinology, Queen Fabiola Children’s University Hospital, Brussels, Belgium, 9Endocrinology Department, L.-Hussel Hospital, Vienne, France, 10Endocrinology Department, Cochin University Hospital, Paris, France, 11Endocrinology Department, Lyon University Hospital, Bron-Lyon, France, 12Paediatric and Clinical Genetic Department, Correspondence Nancy University Hospital, Vandoeuvre les Nancy, France, 13Pediatric Endocrinology and Diabetology, should be -

Independent Evolution of Four Heme Peroxidase Superfamilies
Archives of Biochemistry and Biophysics xxx (2015) xxx–xxx Contents lists available at ScienceDirect Archives of Biochemistry and Biophysics journal homepage: www.elsevier.com/locate/yabbi Independent evolution of four heme peroxidase superfamilies ⇑ Marcel Zámocky´ a,b, , Stefan Hofbauer a,c, Irene Schaffner a, Bernhard Gasselhuber a, Andrea Nicolussi a, Monika Soudi a, Katharina F. Pirker a, Paul G. Furtmüller a, Christian Obinger a a Department of Chemistry, Division of Biochemistry, VIBT – Vienna Institute of BioTechnology, University of Natural Resources and Life Sciences, Muthgasse 18, A-1190 Vienna, Austria b Institute of Molecular Biology, Slovak Academy of Sciences, Dúbravská cesta 21, SK-84551 Bratislava, Slovakia c Department for Structural and Computational Biology, Max F. Perutz Laboratories, University of Vienna, A-1030 Vienna, Austria article info abstract Article history: Four heme peroxidase superfamilies (peroxidase–catalase, peroxidase–cyclooxygenase, peroxidase–chlo- Received 26 November 2014 rite dismutase and peroxidase–peroxygenase superfamily) arose independently during evolution, which and in revised form 23 December 2014 differ in overall fold, active site architecture and enzymatic activities. The redox cofactor is heme b or Available online xxxx posttranslationally modified heme that is ligated by either histidine or cysteine. Heme peroxidases are found in all kingdoms of life and typically catalyze the one- and two-electron oxidation of a myriad of Keywords: organic and inorganic substrates. In addition to this peroxidatic activity distinct (sub)families show pro- Heme peroxidase nounced catalase, cyclooxygenase, chlorite dismutase or peroxygenase activities. Here we describe the Peroxidase–catalase superfamily phylogeny of these four superfamilies and present the most important sequence signatures and active Peroxidase–cyclooxygenase superfamily Peroxidase–chlorite dismutase superfamily site architectures. -

Illness-Induced Changes in Thyroid Hormone Metabolism: Focus on the Tissue Level
r e V i e W illness-induced changes in thyroid hormone metabolism: focus on the tissue level J. Kwakkel*, E. Fliers, A. Boelen Department of Endocrinology & Metabolism, Academic Medical Center, University of Amsterdam, the Netherlands, *corresponding author: tel.: +31 (0)20-566 67 01, fax: +31 (0)20-691 76 82, e-mail: [email protected] a b s t r a C t during illness changes in thyroid hormone metabolism ring and the outer (tyrosyl) ring of T4 can be deiodinated, occur, collectively known as the non-thyroidal illness ultimately leading to the formation of 3,3’-di-iodothyronine syndrome (NTIS). NTIS is characterised by low serum (T2) (figure 1). thyroid hormone levels without the expected rise in serum thyroid-stimulating hormone, indicating a major change in thyroid hormone feedback regulation. recent studies n o n - t H yroidal illness syndro M e have made clear that during NTIS differential changes in thyroid hormone metabolism occur in various tissues, the During illness many aspects of thyroid hormone net effect of which may be either activation or inhibition of metabolism change, collectively known as the thyroid hormone action. in this review we discuss systemic non-thyroidal illness syndrome (NTIS). The hallmark of and local changes in thyroid hormone metabolism during NTIS is decreased serum thyroid hormone levels without illness, highlighting their physiological implications in an increase in TSH and TRH expression, indicating terms of disease course. the absence of negative feedback regulation. This may represent a useful adaptation of the body to counteract excessive catabolism observed during illness and can be K e y W o r d s viewed as a part of the acute phase response.4 However, especially during prolonged critical illness in the ICU Deiodinase, inflammation, non-thyroidal illness syndrome, setting NTIS may be maladaptative.5 thyroid hormone figure 1. -
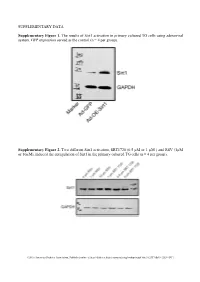
SUPPLEMENTARY DATA Supplementary Figure 1. The
SUPPLEMENTARY DATA Supplementary Figure 1. The results of Sirt1 activation in primary cultured TG cells using adenoviral system. GFP expression served as the control (n = 4 per group). Supplementary Figure 2. Two different Sirt1 activators, SRT1720 (0.5 µM or 1 µM ) and RSV (1µM or 10µM), induced the upregulation of Sirt1 in the primary cultured TG cells (n = 4 per group). ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. Primers used in qPCR Gene Name Primer Sequences Product Size (bp) Sirt1 F: tgccatcatgaagccagaga 241 (NM_001159589) R: aacatcgcagtctccaagga NOX4 F: tgtgcctttattgtgcggag 172 (NM_001285833.1) R: gctgatacactggggcaatg Supplementary Table 2. Antibodies used in Western blot or Immunofluorescence Antibody Company Cat. No Isotype Dilution Sirt1 Santa Cruz * sc-15404 Rabbit IgG 1/200 NF200 Sigma** N5389 Mouse IgG 1/500 Tubulin R&D# MAB1195 Mouse IgG 1/500 NOX4 Abcam† Ab133303 Rabbit IgG 1/500 NOX2 Abcam Ab129068 Rabbit IgG 1/500 phospho-AKT CST‡ #4060 Rabbit IgG 1/500 EGFR CST #4267 Rabbit IgG 1/500 Ki67 Santa Cruz sc-7846 Goat IgG 1/500 * Santa Cruz Biotechnology, Santa Cruz, CA, USA ** Sigma aldrich, Shanghai, China # R&D Systems Inc, Minneapolis, MN, USA † Abcam, Inc., Cambridge, MA, USA ‡ Cell Signaling Technology, Inc., Danvers, MA, USA ©2016 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-1283/-/DC1 SUPPLEMENTARY DATA Supplementary -

Type 3 Lodothyronine Deiodinase: Cloning, in Vitro Expression, and Functional Analysis of the Placental Selenoenzyme
Type 3 lodothyronine deiodinase: cloning, in vitro expression, and functional analysis of the placental selenoenzyme. D Salvatore, … , D L St Germain, P R Larsen J Clin Invest. 1995;96(5):2421-2430. https://doi.org/10.1172/JCI118299. Research Article Type 3 iodothyronine deiodinase (D3) catalyzes the conversion of T4 and T3 to inactive metabolites. It is highly expressed in placenta and thus can regulate circulating fetal thyroid hormone concentrations throughout gestation. We have cloned and expressed a 2.1-kb human placental D3 cDNA which encodes a 32-kD protein with a Km of 1.2 nM for 5 deiodination of T3 and 340 nM for 5' deiodination of reverse T3. The reaction requires DTT and is not inhibited by 6n- propylthiouracil. We quantitated transiently expressed D3 by specifically labeling the protein with bromoacetyl [125I]T3. The Kcat/Km ratio for 5 deiodination of T3 was over 1,000-fold that for 5' deiodination of reverse T3. Human D3 is a selenoenzyme as evidenced by (a) the presence of an in frame UGA codon at position 144, (b) the synthesis of a 32-kD 75Se-labeled protein in D3 cDNA transfected cells, and (c) the presence of a selenocysteine insertion sequence element in the 3' untranslated region of the mRNA which is required for its expression. The D3 selenocysteine insertion sequence element is more potent than that in the type 1 deiodinase or glutathione peroxidase gene, suggesting a high priority for selenocysteine incorporation into this enzyme. The conservation of this enzyme from Xenopus laevis tadpoles to humans implies an essential role for regulation of thyroid hormone inactivation during embryological development. -

Lactoperoxidase Antibacterial System: Natural Occurrence, Biological Functions and Practical Applications
724 Journal of Food Protection, Vol. 47. No. 9, Pages 724-732 (September 1984) Copyright®, International Association of Milk, Food, and Environmental Sanitarians Lactoperoxidase Antibacterial System: Natural Occurrence, Biological Functions and Practical Applications BRUNO REITER1 and GORAN HARNULV2* Downloaded from http://meridian.allenpress.com/jfp/article-pdf/47/9/724/1650811/0362-028x-47_9_724.pdf by guest on 29 September 2021 National Institute for Research in Dairying, Shinfield, Reading, RG2 9AT, England and Alfa-Laval Agri International AS, P.O. Box 39, S-I47 00 Tumba, Sweden (Received for publication January 30, 1984) ABSTRACT (37,80). The various biological functions of the LP sys tem, which have now been established, will be discussed In the present review dealing with the antibacterial lac below. toperoxidase (LP) system, it is shown that the two reactants In recent years, much work has been devoted to practi thiocyanate (SCN~) and hydrogen peroxide (H202) as well as cal applications of the LP system. The effect against oral the catalytic enzyme lactoperoxidase (LP) are widely distributed streptococci (5,19,33,101,105,107) has led to the de in nature and that evidence for the activity of the LP system velopment and marketing of a toothpaste containing nec in animals, including man, is accumulating. The in vitro effects on bacterial and animal cells are discussed and the unique ac essary ingredients to activate the LP system. Promising tion of the LP system on the bacterial cytoplasmic membrane results have also been obtained when including activating is pointed out. Some practical applications are also presented, components in the feed to calves (81,83,86) with the aim with particular emphses on the possibility of utilizing the LP of potentiating the LP system in the intestinal tract. -

Thyroxine Binding to Type III Iodothyronine Deiodinase Craig A
www.nature.com/scientificreports OPEN Thyroxine binding to type III iodothyronine deiodinase Craig A. Bayse*, Eric S. Marsan, Jenna R. Garcia & Alexis T. Tran‑Thompson Iodothyronine deiodinases (Dios) are important selenoproteins that control the concentration of the active thyroid hormone (TH) triiodothyronine through regioselective deiodination. The X‑ray structure of a truncated monomer of Type III Dio (Dio3), which deiodinates TH inner rings through a selenocysteine (Sec) residue, revealed a thioredoxin-fold catalytic domain supplemented with an unstructured Ω-loop. Loop dynamics are driven by interactions of the conserved Trp207 with solvent in multi-microsecond molecular dynamics simulations of the Dio3 thioredoxin(Trx)-fold domain. Hydrogen bonding interactions of Glu200 with residues conserved across the Dio family anchor the loop’s n‑terminus to the active site Ser‑cys-Thr‑Sec sequence. A key long‑lived loop conformation coincides with the opening of a cryptic pocket that accommodates thyroxine (T4) through an I⋯Se halogen bond to Sec170 and the amino acid group with a polar cleft. The Dio3-T4 complex is stabilized by an I⋯O halogen bond between an outer ring iodine and Asp211, consistent with Dio3 selectivity for inner ring deiodination. Non-conservation of residues, such as Asp211, in other Dio types in the fexible portion of the loop sequence suggests a mechanism for regioselectivity through Dio type- specifc loop conformations. Cys168 is proposed to attack the selenenyl iodide intermediate to regenerate Dio3 based upon structural comparison with related Trx-fold proteins. Iodothyronine deiodinase (Dio) membrane selenoproteins regulate thyroid hormone (TH) activity through regioselective deiodination (Fig. 1a)1–10. -

The Interactions Between Selenium and Iodine Deficiencies in Man And
Nutrition Research Reviews (1999), 12, 55±73 55 The interactions between selenium and iodine de®ciencies in man and animals John R. Arthur1*, Geoffrey J. Beckett2 and Julie H. Mitchell1 1Division of Micronutrient and Lipid Metabolism, Rowett Research Institute, Greenburn Road, Bucksburn, Aberdeen AB21 9SB, UK 2University Department of Clinical Biochemistry, The Royal In®rmary, Edinburgh EH3 9YW, UK Abstract Up to one billion people live in areas where they may be at risk from I de®ciency. Many of the debilitating effects of the de®ciency may be irreversible, consequently it is essential to understand the mechanisms whereby lack of I can cause disease 0 through decreased thyroxine and 3,3 ,5-triiodothyronine (T3) synthesis. Since Se has an essential role in thyroid hormone metabolism, it has the potential to play a major part in the outcome of I de®ciency. These effects of Se derive from two aspects of its biological function. First, three Se-containing deiodinases regulate the synthesis and degradation of the biologically active thyroid hormone, T3. Second, selenoperoxidases and possibly thioredoxin reductase (EC 1.6.4.5) protect the thyroid gland from H2O2 produced during the synthesis of thyroid hormones. The mechanisms whereby Se de®ciency exacerbates the hypothyroidism due to I de®ciency have been elucidated in animals. In contrast to these adverse effects, concurrent Se de®ciency may also cause changes in deiodinase activities which can protect the brain from low T3 concentrations in I de®ciency. Animals with Se and I de®ciency have changes in serum thyroid hormone concentrations that are similar to those observed in patients with I de®ciency disease.