Oligodendrocyte Precursor Cells Prune Axons in the Mouse Neocortex
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-
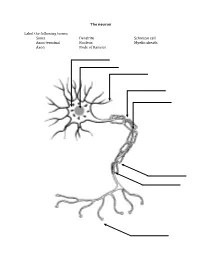
The Neuron Label the Following Terms: Soma Axon Terminal Axon Dendrite
The neuron Label the following terms: Soma Dendrite Schwaan cell Axon terminal Nucleus Myelin sheath Axon Node of Ranvier Neuron Vocabulary You must know the definitions of these terms 1. Synaptic Cleft 2. Neuron 3. Impulse 4. Sensory Neuron 5. Motor Neuron 6. Interneuron 7. Body (Soma) 8. Dendrite 9. Axon 10. Action Potential 11. Myelin Sheath (Myelin) 12. Afferent Neuron 13. Threshold 14. Neurotransmitter 15. Efferent Neurons 16. Axon Terminal 17. Stimulus 18. Refractory Period 19. Schwann 20. Nodes of Ranvier 21. Acetylcholine STEPS IN THE ACTION POTENTIAL 1. The presynaptic neuron sends neurotransmitters to postsynaptic neuron. 2. Neurotransmitters bind to receptors on the postsynaptic cell. - This action will either excite or inhibit the postsynaptic cell. - The soma becomes more positive. 3. The positive charge reaches the axon hillock. - Once the threshold of excitation is reached the neuron will fire an action potential. 4. Na+ channels open and Na+ is forced into the cell by the concentration gradient and the electrical gradient. - The neuron begins to depolarize. 5. The K+ channels open and K+ is forced out of the cell by the concentration gradient and the electrical gradient. - The neuron is depolarized. 6. The Na+ channels close at the peak of the action potential. - The neuron starts to repolarize. 7. The K+ channels close, but they close slowly and K+ leaks out. 8. The terminal buttons release neurotransmitter to the postsynaptic neuron. 9. The resting potential is overshot and the neuron falls to a -90mV (hyperpolarizes). - The neuron continues to repolarize. 10. The neuron returns to resting potential. The Synapse Label the following terms: Pre-synaptic membrane Neurotransmitters Post-synaptic membrane Synaptic cleft Vesicle Post-synaptic receptors . -

Resting and Active Properties of Pyramidal Neurons in Subiculum and CA1 of Rat Hippocampus
Resting and Active Properties of Pyramidal Neurons in Subiculum and CA1 of Rat Hippocampus NATHAN P. STAFF, HAE-YOON JUNG, TARA THIAGARAJAN, MICHAEL YAO, AND NELSON SPRUSTON Department of Neurobiology and Physiology, Institute for Neuroscience, Northwestern University, Evanston, Illinois 60208 Received 22 March 2000; accepted in final form 8 August 2000 Staff, Nathan P., Hae-Yoon Jung, Tara Thiagarajan, Michael region (Chrobak and Buzsaki 1996; Colling et al. 1998). Sub- Yao, and Nelson Spruston. Resting and active properties of pyrami- iculum has been shown to be the major output center of the dal neurons in subiculum and CA1 of rat hippocampus. J Neuro- hippocampus, to which it belongs, sending parallel projections physiol 84: 2398–2408, 2000. Action potentials are the end product of synaptic integration, a process influenced by resting and active neu- from various subicular regions to different cortical and subcor- ronal membrane properties. Diversity in these properties contributes tical areas (Naber and Witter 1998). Functionally, the principal to specialized mechanisms of synaptic integration and action potential neurons of the subiculum appear to be “place cells,” similar to firing, which are likely to be of functional significance within neural those of CA1 (Sharp and Green 1994); and in a human mag- circuits. In the hippocampus, the majority of subicular pyramidal netic resonance imaging (MRI) study, subiculum was shown to neurons fire high-frequency bursts of action potentials, whereas CA1 be an area involved in memory retrieval (Gabrieli et al. 1997). pyramidal neurons exhibit regular spiking behavior when subjected to Therefore both CA1 and subiculum have been implicated in direct somatic current injection. -

Crayfish Escape Behavior and Central Synapses
Crayfish Escape Behavior and Central Synapses. III. Electrical Junctions and Dendrite Spikes in Fast Flexor Motoneurons ROBERT S. ZUCKER Department of Biological Sciences and Program in the Neurological Sciences, Stanford University, Stanford, California 94305 THE LATERAL giant fiber is the decision fiber and fast flexor motoneurons are located in for a behavioral response in crayfish in- the dorsal neuropil of each abdominal gan- volving rapid tail flexion in response to glion. Dye injections and anatomical recon- abdominal tactile stimulation (27). Each structions of ‘identified motoneurons reveal lateral giant impulse is followed by a rapid that only those motoneurons which have tail flexion or tail flip, and when the giant dentritic branches in contact with a giant fiber does not fire in response to such stim- fiber are activated by that giant fiber (12, uli, the movement does not occur. 21). All of tl ie fast flexor motoneurons are The afferent limb of the reflex exciting excited by the ipsilateral giant; all but F4, the lateral giant has been described (28), F5 and F7 are also excited by the contra- and the habituation of the behavior has latkral giant (21 a). been explained in terms of the properties The excitation of these motoneurons, of this part of the neural circuit (29). seen as depolarizing potentials in their The properties of the neuromuscular somata, does not always reach threshold for junctions between the fast flexor motoneu- generating a spike which propagates out the rons and the phasic flexor muscles have also axon to the periphery (14). Indeed, only t,he been described (13). -

Long-Term Adult Human Brain Slice Cultures As a Model System to Study
TOOLS AND RESOURCES Long-term adult human brain slice cultures as a model system to study human CNS circuitry and disease Niklas Schwarz1, Betu¨ l Uysal1, Marc Welzer2,3, Jacqueline C Bahr1, Nikolas Layer1, Heidi Lo¨ ffler1, Kornelijus Stanaitis1, Harshad PA1, Yvonne G Weber1,4, Ulrike BS Hedrich1, Ju¨ rgen B Honegger4, Angelos Skodras2,3, Albert J Becker5, Thomas V Wuttke1,4†*, Henner Koch1†* 1Department of Neurology and Epileptology, Hertie-Institute for Clinical Brain Research, University of Tu¨ bingen, Tu¨ bingen, Germany; 2Department of Cellular Neurology, Hertie-Institute for Clinical Brain Research, University of Tu¨ bingen, Tu¨ bingen, Germany; 3German Center for Neurodegenerative Diseases (DZNE), Tu¨ bingen, Germany; 4Department of Neurosurgery, University of Tu¨ bingen, Tu¨ bingen, Germany; 5Department of Neuropathology, Section for Translational Epilepsy Research, University Bonn Medical Center, Bonn, Germany Abstract Most of our knowledge on human CNS circuitry and related disorders originates from model organisms. How well such data translate to the human CNS remains largely to be determined. Human brain slice cultures derived from neurosurgical resections may offer novel avenues to approach this translational gap. We now demonstrate robust preservation of the complex neuronal cytoarchitecture and electrophysiological properties of human pyramidal neurons *For correspondence: in long-term brain slice cultures. Further experiments delineate the optimal conditions for efficient [email protected] viral transduction of cultures, enabling ‘high throughput’ fluorescence-mediated 3D reconstruction tuebingen.de (TVW); of genetically targeted neurons at comparable quality to state-of-the-art biocytin fillings, and [email protected] demonstrate feasibility of long term live cell imaging of human cells in vitro. -

Activation of Autophagy Rescues Synaptic and Cognitive Deficits in Fragile X Mice
Activation of autophagy rescues synaptic and cognitive deficits in fragile X mice Jingqi Yana,1, Morgan W. Porcha,1, Brenda Court-Vazqueza, Michael V. L. Bennetta,2, and R. Suzanne Zukina,2 aDominick P. Purpura Department of Neuroscience, Albert Einstein College of Medicine, New York, NY 10461 Contributed by Michael V. L. Bennett, August 17, 2018 (sent for review May 29, 2018; reviewed by Claudia Bagni and Leonard K. Kaczmarek) Fragile X syndrome (FXS) is the most frequent form of heritable (17–19). Upon activation, mTOR phosphorylates Unc-51–like intellectual disability and autism. Fragile X (Fmr1-KO) mice exhibit autophagy-activating kinase 1 (ULK-1) at S757, a target of aberrant dendritic spine structure, synaptic plasticity, and cogni- mTORC1 and a well-established antiautophagy site (Fig. 1) tion. Autophagy is a catabolic process of programmed degradation (20). This, in turn, sequesters ULK-1 away from AMP kinase and recycling of proteins and cellular components via the lyso- (AMPK). AMPK phosphorylates and activates ULK-1 at S317. somal pathway. However, a role for autophagy in the pathophys- Upon activation, ULK-1 promotes phosphorylation and activa- iology of FXS is, as yet, unclear. Here we show that autophagic tion of Beclin-1 at S14, a critical step in the nucleation phase of flux, a functional readout of autophagy, and biochemical markers autophagy (21). Beclin-1 promotes lipidation of LC3-I to gen- of autophagy are down-regulated in hippocampal neurons of frag- erate its lipidated form LC3-II, enabling elongation of the lim- ile X mice. We further show that enhanced activity of mammalian iting membrane and the formation of autophagosomes (22). -

Plp-Positive Progenitor Cells Give Rise to Bergmann Glia in the Cerebellum
Citation: Cell Death and Disease (2013) 4, e546; doi:10.1038/cddis.2013.74 OPEN & 2013 Macmillan Publishers Limited All rights reserved 2041-4889/13 www.nature.com/cddis Olig2/Plp-positive progenitor cells give rise to Bergmann glia in the cerebellum S-H Chung1, F Guo2, P Jiang1, DE Pleasure2,3 and W Deng*,1,3,4 NG2 (nerve/glial antigen2)-expressing cells represent the largest population of postnatal progenitors in the central nervous system and have been classified as oligodendroglial progenitor cells, but the fate and function of these cells remain incompletely characterized. Previous studies have focused on characterizing these progenitors in the postnatal and adult subventricular zone and on analyzing the cellular and physiological properties of these cells in white and gray matter regions in the forebrain. In the present study, we examine the types of neural progeny generated by NG2 progenitors in the cerebellum by employing genetic fate mapping techniques using inducible Cre–Lox systems in vivo with two different mouse lines, the Plp-Cre-ERT2/Rosa26-EYFP and Olig2-Cre-ERT2/Rosa26-EYFP double-transgenic mice. Our data indicate that Olig2/Plp-positive NG2 cells display multipotential properties, primarily give rise to oligodendroglia but, surprisingly, also generate Bergmann glia, which are specialized glial cells in the cerebellum. The NG2 þ cells also give rise to astrocytes, but not neurons. In addition, we show that glutamate signaling is involved in distinct NG2 þ cell-fate/differentiation pathways and plays a role in the normal development of Bergmann glia. We also show an increase of cerebellar oligodendroglial lineage cells in response to hypoxic–ischemic injury, but the ability of NG2 þ cells to give rise to Bergmann glia and astrocytes remains unchanged. -

Control of Synapse Number by Glia Erik M
R EPORTS RGCs (8). Similar results were obtained un- der both conditions. After 2 weeks in culture, Control of Synapse Number we used whole-cell patch-clamp recording to measure the significant enhancement of spon- by Glia taneous synaptic activity by astrocytes (Fig. 1, A and B) (9). In the absence of astroglia, Erik M. Ullian,* Stephanie K. Sapperstein, Karen S. Christopherson, little synaptic activity occurred even when Ben A. Barres RGCs were cultured with their tectal target cells (6). This difference in synaptic activity Although astrocytes constitute nearly half of the cells in our brain, their persisted even after a month in culture, indi- function is a long-standing neurobiological mystery. Here we show by quantal cating that it is not accounted for by a matu- analyses, FM1-43 imaging, immunostaining, and electron microscopy that few rational delay. To examine whether glia reg- synapses form in the absence of glial cells and that the few synapses that do ulate synaptic activity by enhancing postsyn- form are functionally immature. Astrocytes increase the number of mature, aptic responsiveness, we measured the re- functional synapses on central nervous system (CNS) neurons by sevenfold and sponse of RGCs to pulses of L-glutamate are required for synaptic maintenance in vitro. We also show that most syn- applied to the cell somas. Regardless of the apses are generated concurrently with the development of glia in vivo. These presence of glia, RGCs responded with in- data demonstrate a previously unknown function for glia in inducing and ward currents (Fig. 1C) that were blocked by stabilizing CNS synapses, show that CNS synapse number can be profoundly glutamate receptor antagonists (10). -

Oligodendrocytes in Development, Myelin Generation and Beyond
cells Review Oligodendrocytes in Development, Myelin Generation and Beyond Sarah Kuhn y, Laura Gritti y, Daniel Crooks and Yvonne Dombrowski * Wellcome-Wolfson Institute for Experimental Medicine, Queen’s University Belfast, Belfast BT9 7BL, UK; [email protected] (S.K.); [email protected] (L.G.); [email protected] (D.C.) * Correspondence: [email protected]; Tel.: +0044-28-9097-6127 These authors contributed equally. y Received: 15 October 2019; Accepted: 7 November 2019; Published: 12 November 2019 Abstract: Oligodendrocytes are the myelinating cells of the central nervous system (CNS) that are generated from oligodendrocyte progenitor cells (OPC). OPC are distributed throughout the CNS and represent a pool of migratory and proliferative adult progenitor cells that can differentiate into oligodendrocytes. The central function of oligodendrocytes is to generate myelin, which is an extended membrane from the cell that wraps tightly around axons. Due to this energy consuming process and the associated high metabolic turnover oligodendrocytes are vulnerable to cytotoxic and excitotoxic factors. Oligodendrocyte pathology is therefore evident in a range of disorders including multiple sclerosis, schizophrenia and Alzheimer’s disease. Deceased oligodendrocytes can be replenished from the adult OPC pool and lost myelin can be regenerated during remyelination, which can prevent axonal degeneration and can restore function. Cell population studies have recently identified novel immunomodulatory functions of oligodendrocytes, the implications of which, e.g., for diseases with primary oligodendrocyte pathology, are not yet clear. Here, we review the journey of oligodendrocytes from the embryonic stage to their role in homeostasis and their fate in disease. We will also discuss the most common models used to study oligodendrocytes and describe newly discovered functions of oligodendrocytes. -

Neuregulin 1–Erbb2 Signaling Is Required for the Establishment of Radial Glia and Their Transformation Into Astrocytes in Cerebral Cortex
Neuregulin 1–erbB2 signaling is required for the establishment of radial glia and their transformation into astrocytes in cerebral cortex Ralf S. Schmid*, Barbara McGrath*, Bridget E. Berechid†, Becky Boyles*, Mark Marchionni‡, Nenad Sˇ estan†, and Eva S. Anton*§ *University of North Carolina Neuroscience Center and Department of Cell and Molecular Physiology, University of North Carolina School of Medicine, Chapel Hill, NC 27599; †Department of Neurobiology, Yale University School of Medicine, New Haven, CT 06510; and ‡CeNes Pharamceuticals, Inc., Norwood, MA 02062 Communicated by Pasko Rakic, Yale University School of Medicine, New Haven, CT, January 27, 2003 (received for review December 12, 2002) Radial glial cells and astrocytes function to support the construction mine whether NRG-1-mediated signaling is involved in radial and maintenance, respectively, of the cerebral cortex. However, the glial cell development and differentiation in the cerebral cortex. mechanisms that determine how radial glial cells are established, We show that NRG-1 signaling, involving erbB2, may act in maintained, and transformed into astrocytes in the cerebral cortex are concert with Notch signaling to exert a critical influence in the not well understood. Here, we show that neuregulin-1 (NRG-1) exerts establishment, maintenance, and appropriate transformation of a critical role in the establishment of radial glial cells. Radial glial cell radial glial cells in cerebral cortex. generation is significantly impaired in NRG mutants, and this defect can be rescued by exogenous NRG-1. Down-regulation of expression Materials and Methods and activity of erbB2, a member of the NRG-1 receptor complex, leads Clonal Analysis to Study NRG’s Role in the Initial Establishment of to the transformation of radial glial cells into astrocytes. -

Chapter 8 Nervous System
Chapter 8 Nervous System I. Functions A. Sensory Input – stimuli interpreted as touch, taste, temperature, smell, sound, blood pressure, and body position. B. Integration – CNS processes sensory input and initiates responses categorizing into immediate response, memory, or ignore C. Homeostasis – maintains through sensory input and integration by stimulating or inhibiting other systems D. Mental Activity – consciousness, memory, thinking E. Control of Muscles & Glands – controls skeletal muscle and helps control/regulate smooth muscle, cardiac muscle, and glands II. Divisions of the Nervous system – 2 anatomical/main divisions A. CNS (Central Nervous System) – consists of the brain and spinal cord B. PNS (Peripheral Nervous System) – consists of ganglia and nerves outside the brain and spinal cord – has 2 subdivisions 1. Sensory Division (Afferent) – conducts action potentials from PNS toward the CNS (by way of the sensory neurons) for evaluation 2. Motor Division (Efferent) – conducts action potentials from CNS toward the PNS (by way of the motor neurons) creating a response from an effector organ – has 2 subdivisions a. Somatic Motor System – controls skeletal muscle only b. Autonomic System – controls/effects smooth muscle, cardiac muscle, and glands – 2 branches • Sympathetic – accelerator “fight or flight” • Parasympathetic – brake “resting and digesting” * 4 Types of Effector Organs: skeletal muscle, smooth muscle, cardiac muscle, and glands. III. Cells of the Nervous System A. Neurons – receive stimuli and transmit action potentials -

Pre-Oligodendrocytes from Adult Human CNS
The Journal of Neuroscience, April 1992, 12(4): 1538-l 547 Pre-Oligodendrocytes from Adult Human CNS Regina C. Armstrong,lJ Henry H. Dorn, l,b Conrad V. Kufta,* Emily Friedman,3 and Monique E. Dubois-Dalcq’ ‘Laboratory of Viral and Molecular Pathogenesis, and %urgical Neurology Branch, National Institute of Neurological Disorders and Stroke, Bethesda, Maryland 20892 and 3Department of Neurosurgery, University of Pennsylvania, Philadelphia, Pennsylvania 19104-3246 CNS remyelination and functional recovery often occur after Rapid and efficient neurotransmission is dependent upon the experimental demyelination in adult rodents. This has been electrical insulating capacity of the myelin sheath around axons attributed to the ability of mature oligodendrocytes and/or (reviewed in Ritchie, 1984a,b). Nerve conduction is impaired their precursor cells to divide and regenerate in response after loss of the myelin sheath and results in severe neurological to signals in demyelinating lesions. To determine whether dysfunction in human demyelinating diseases such as multiple oligodendrocyte precursor cells exist in the adult human sclerosis (MS). Remyelination can occur in the CNS of MS CNS, we have cultured white matter from patients under- patients but appears to be limited (Perier and Gregoire, 1965; going partial temporal lobe resection for intractable epilep- Prineas et al., 1984). Studies of acute MS cases have revealed sy. These cultures contained a population of process-bear- that recent demyelinating lesions can exhibit remyelination that ing cells that expressed antigens recognized by the 04 appears to correlate with the generation of new oligodendrocytes monoclonal antibody, but these cells did not express galac- (Prineas et al., 1984; Raine et al., 1988). -

Area-Specific Synapse Structure in Branched Posterior Nucleus Axons Reveals a New Level of Complexity in Thalamocortical Networks
The Journal of Neuroscience, March 25, 2020 • 40(13):2663–2679 • 2663 Systems/Circuits Area-Specific Synapse Structure in Branched Posterior Nucleus Axons Reveals a New Level of Complexity in Thalamocortical Networks Javier Rodriguez-Moreno,1 Cesar Porrero,1 Astrid Rollenhagen,2 Mario Rubio-Teves,1 Diana Casas-Torremocha,1 X Lidia Alonso-Nanclares,3 Rachida Yakoubi,2 XAndrea Santuy,3 XAngel Merchan-Pe´rez,3,5,6 XJavier DeFelipe,3,4,5 Joachim H.R. Lu¨bke,2,7,8* and XFrancisco Clasca1* 1Department of Anatomy and Neuroscience, School of Medicine, Auto´noma de Madrid University, 28029 Madrid, Spain, 2Institute of Neuroscience and Medicine INM-10, Research Centre Ju¨lich GmbH, 52425 Ju¨lich, Germany, 3Laboratorio Cajal de Circuitos Corticales, Centro de Tecnología Biome´dica, Universidad Polite´cnica de Madrid, Pozuelo de Alarco´n, 28223 Madrid, Spain, 4Instituto Cajal, Consejo Superior de Investigaciones Científicas, Arce 37 28002, Madrid, Spain, 5CIBERNED, Centro de Investigacio´n Biome´dica en Red de Enfermedades Neurodegenerativas, 28031 Madrid, Spain, 6Departamento de Arquitectura y Tecnología de Sistemas Informa´ticos, Universidad Polite´cnica de Madrid. Boadilla del Monte, 28660 Madrid, Spain, 7Department of Psychiatry, Psychotherapy and Psychosomatics, Medical Faculty RWTH University Hospital Aachen, 52074 Aachen, Germany, and 8JARA-Translational Brain Medicine, 52425 Ju¨lich-Aachen, Germany Thalamocortical posterior nucleus (Po) axons innervating the vibrissal somatosensory (S1) and motor (MC) cortices are key links in the brain neuronal network that allows rodents to explore the environment whisking with their motile snout vibrissae. Here, using fine-scale high-end 3D electron microscopy, we demonstrate in adult male C57BL/6 wild-type mice marked differences between MC versus S1 Po synapses in (1) bouton and active zone size, (2) neurotransmitter vesicle pool size, (3) distribution of mitochondria around synapses, and (4) proportion of synapses established on dendritic spines and dendritic shafts.