Product Datasheet
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Investigation of the Immune Receptors CEACAM3 and CEACAM4
Investigation of the human immune receptors CEACAM3 and CEACAM4 Dissertation Zur Erlangung des akademischen Grades eines Doktors der Naturwissenschaften (Dr. rer. nat.) vorgelegt von Julia Delgado Tascón An der Universität Konstanz des Fachbereichs Biologie Konstanz, Oktober 2015 Konstanzer Online-Publikations-System (KOPS) URL: http://nbn-resolving.de/urn:nbn:de:bsz:352-0-306516 Tag der mündlichen Prüfung: 05.11.2015 Vorsitzender und mündlicher Prüfer: Herr Professor Dr. Bürkle 1. Referent und und mündlicher Prüfer: Herr Professor Dr. Hauck 2. Referent und und mündlicher Prüfer: Herr Professor Dr. Tschan, Universität Bern A mi familia Acknowledgements I would like to express my special gratitude to my advisor Prof. Dr. Christof Hauck. His patient guidance and enthusiastic encouragement during these four years of PhD were a crucial aid to my process. I’m very thankful for his willingness and for granting me with his time in search for valuable and constructive suggestions during the planning and development of this research work. This certainly allowed me to grow as a person and as a scientist. I would also like to thank my committee members: to Prof. Dr. Mario Tschan for giving me his academic support at this last phase of my PhD thesis, and to Prof. M.Dr. Alexander Bürkle for his wise advices accompanied with Spanish greetings along this time. My thanks are extended to every member of the AG Hauck as well. To Anne, Susana, Petra and Claudia: thank you very much for your technical and personal guidance during these years. I’m also thankful to my fellow colleagues for countless ‘Kaffeepausen’ full of jokes, nice discussions, and delicious vegan cakes. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

Supplementary Table 1: Adhesion Genes Data Set
Supplementary Table 1: Adhesion genes data set PROBE Entrez Gene ID Celera Gene ID Gene_Symbol Gene_Name 160832 1 hCG201364.3 A1BG alpha-1-B glycoprotein 223658 1 hCG201364.3 A1BG alpha-1-B glycoprotein 212988 102 hCG40040.3 ADAM10 ADAM metallopeptidase domain 10 133411 4185 hCG28232.2 ADAM11 ADAM metallopeptidase domain 11 110695 8038 hCG40937.4 ADAM12 ADAM metallopeptidase domain 12 (meltrin alpha) 195222 8038 hCG40937.4 ADAM12 ADAM metallopeptidase domain 12 (meltrin alpha) 165344 8751 hCG20021.3 ADAM15 ADAM metallopeptidase domain 15 (metargidin) 189065 6868 null ADAM17 ADAM metallopeptidase domain 17 (tumor necrosis factor, alpha, converting enzyme) 108119 8728 hCG15398.4 ADAM19 ADAM metallopeptidase domain 19 (meltrin beta) 117763 8748 hCG20675.3 ADAM20 ADAM metallopeptidase domain 20 126448 8747 hCG1785634.2 ADAM21 ADAM metallopeptidase domain 21 208981 8747 hCG1785634.2|hCG2042897 ADAM21 ADAM metallopeptidase domain 21 180903 53616 hCG17212.4 ADAM22 ADAM metallopeptidase domain 22 177272 8745 hCG1811623.1 ADAM23 ADAM metallopeptidase domain 23 102384 10863 hCG1818505.1 ADAM28 ADAM metallopeptidase domain 28 119968 11086 hCG1786734.2 ADAM29 ADAM metallopeptidase domain 29 205542 11085 hCG1997196.1 ADAM30 ADAM metallopeptidase domain 30 148417 80332 hCG39255.4 ADAM33 ADAM metallopeptidase domain 33 140492 8756 hCG1789002.2 ADAM7 ADAM metallopeptidase domain 7 122603 101 hCG1816947.1 ADAM8 ADAM metallopeptidase domain 8 183965 8754 hCG1996391 ADAM9 ADAM metallopeptidase domain 9 (meltrin gamma) 129974 27299 hCG15447.3 ADAMDEC1 ADAM-like, -

Integrating Text Mining, Data Mining, and Network Analysis for Identifying
Jurca et al. BMC Res Notes (2016) 9:236 DOI 10.1186/s13104-016-2023-5 BMC Research Notes RESEARCH ARTICLE Open Access Integrating text mining, data mining, and network analysis for identifying genetic breast cancer trends Gabriela Jurca1, Omar Addam1, Alper Aksac1, Shang Gao2, Tansel Özyer3, Douglas Demetrick4 and Reda Alhajj1,5* Abstract Background: Breast cancer is a serious disease which affects many women and may lead to death. It has received considerable attention from the research community. Thus, biomedical researchers aim to find genetic biomarkers indicative of the disease. Novel biomarkers can be elucidated from the existing literature. However, the vast amount of scientific publications on breast cancer make this a daunting task. This paper presents a framework which investigates existing literature data for informative discoveries. It integrates text mining and social network analysis in order to identify new potential biomarkers for breast cancer. Results: We utilized PubMed for the testing. We investigated gene–gene interactions, as well as novel interactions such as gene-year, gene-country, and abstract-country to find out how the discoveries varied over time and how overlapping/diverse are the discoveries and the interest of various research groups in different countries. Conclusions: Interesting trends have been identified and discussed, e.g., different genes are highlighted in relation- ship to different countries though the various genes were found to share functionality. Some text analysis based results have been validated against results from other tools that predict gene–gene relations and gene functions. Keywords: Breast cancer, Data mining, Text mining, Network analysis Background reducing the number of molecules to consider as poten- Introduction tial biomarkers. -

CD Markers Are Routinely Used for the Immunophenotyping of Cells
ptglab.com 1 CD MARKER ANTIBODIES www.ptglab.com Introduction The cluster of differentiation (abbreviated as CD) is a protocol used for the identification and investigation of cell surface molecules. So-called CD markers are routinely used for the immunophenotyping of cells. Despite this use, they are not limited to roles in the immune system and perform a variety of roles in cell differentiation, adhesion, migration, blood clotting, gamete fertilization, amino acid transport and apoptosis, among many others. As such, Proteintech’s mini catalog featuring its antibodies targeting CD markers is applicable to a wide range of research disciplines. PRODUCT FOCUS PECAM1 Platelet endothelial cell adhesion of blood vessels – making up a large portion molecule-1 (PECAM1), also known as cluster of its intracellular junctions. PECAM-1 is also CD Number of differentiation 31 (CD31), is a member of present on the surface of hematopoietic the immunoglobulin gene superfamily of cell cells and immune cells including platelets, CD31 adhesion molecules. It is highly expressed monocytes, neutrophils, natural killer cells, on the surface of the endothelium – the thin megakaryocytes and some types of T-cell. Catalog Number layer of endothelial cells lining the interior 11256-1-AP Type Rabbit Polyclonal Applications ELISA, FC, IF, IHC, IP, WB 16 Publications Immunohistochemical of paraffin-embedded Figure 1: Immunofluorescence staining human hepatocirrhosis using PECAM1, CD31 of PECAM1 (11256-1-AP), Alexa 488 goat antibody (11265-1-AP) at a dilution of 1:50 anti-rabbit (green), and smooth muscle KD/KO Validated (40x objective). alpha-actin (red), courtesy of Nicola Smart. PECAM1: Customer Testimonial Nicola Smart, a cardiovascular researcher “As you can see [the immunostaining] is and a group leader at the University of extremely clean and specific [and] displays Oxford, has said of the PECAM1 antibody strong intercellular junction expression, (11265-1-AP) that it “worked beautifully as expected for a cell adhesion molecule.” on every occasion I’ve tried it.” Proteintech thanks Dr. -
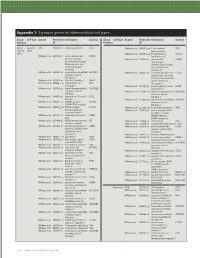
Technical Note, Appendix: an Analysis of Blood Processing Methods to Prepare Samples for Genechip® Expression Profiling (Pdf, 1
Appendix 1: Signature genes for different blood cell types. Blood Cell Type Source Probe Set Description Symbol Blood Cell Type Source Probe Set Description Symbol Fraction ID Fraction ID Mono- Lympho- GSK 203547_at CD4 antigen (p55) CD4 Whitney et al. 209813_x_at T cell receptor TRG nuclear cytes gamma locus cells Whitney et al. 209995_s_at T-cell leukemia/ TCL1A Whitney et al. 203104_at colony stimulating CSF1R lymphoma 1A factor 1 receptor, Whitney et al. 210164_at granzyme B GZMB formerly McDonough (granzyme 2, feline sarcoma viral cytotoxic T-lymphocyte- (v-fms) oncogene associated serine homolog esterase 1) Whitney et al. 203290_at major histocompatibility HLA-DQA1 Whitney et al. 210321_at similar to granzyme B CTLA1 complex, class II, (granzyme 2, cytotoxic DQ alpha 1 T-lymphocyte-associated Whitney et al. 203413_at NEL-like 2 (chicken) NELL2 serine esterase 1) Whitney et al. 203828_s_at natural killer cell NK4 (H. sapiens) transcript 4 Whitney et al. 212827_at immunoglobulin heavy IGHM Whitney et al. 203932_at major histocompatibility HLA-DMB constant mu complex, class II, Whitney et al. 212998_x_at major histocompatibility HLA-DQB1 DM beta complex, class II, Whitney et al. 204655_at chemokine (C-C motif) CCL5 DQ beta 1 ligand 5 Whitney et al. 212999_x_at major histocompatibility HLA-DQB Whitney et al. 204661_at CDW52 antigen CDW52 complex, class II, (CAMPATH-1 antigen) DQ beta 1 Whitney et al. 205049_s_at CD79A antigen CD79A Whitney et al. 213193_x_at T cell receptor beta locus TRB (immunoglobulin- Whitney et al. 213425_at Homo sapiens cDNA associated alpha) FLJ11441 fis, clone Whitney et al. 205291_at interleukin 2 receptor, IL2RB HEMBA1001323, beta mRNA sequence Whitney et al. -

Supplementary Table S4. FGA Co-Expressed Gene List in LUAD
Supplementary Table S4. FGA co-expressed gene list in LUAD tumors Symbol R Locus Description FGG 0.919 4q28 fibrinogen gamma chain FGL1 0.635 8p22 fibrinogen-like 1 SLC7A2 0.536 8p22 solute carrier family 7 (cationic amino acid transporter, y+ system), member 2 DUSP4 0.521 8p12-p11 dual specificity phosphatase 4 HAL 0.51 12q22-q24.1histidine ammonia-lyase PDE4D 0.499 5q12 phosphodiesterase 4D, cAMP-specific FURIN 0.497 15q26.1 furin (paired basic amino acid cleaving enzyme) CPS1 0.49 2q35 carbamoyl-phosphate synthase 1, mitochondrial TESC 0.478 12q24.22 tescalcin INHA 0.465 2q35 inhibin, alpha S100P 0.461 4p16 S100 calcium binding protein P VPS37A 0.447 8p22 vacuolar protein sorting 37 homolog A (S. cerevisiae) SLC16A14 0.447 2q36.3 solute carrier family 16, member 14 PPARGC1A 0.443 4p15.1 peroxisome proliferator-activated receptor gamma, coactivator 1 alpha SIK1 0.435 21q22.3 salt-inducible kinase 1 IRS2 0.434 13q34 insulin receptor substrate 2 RND1 0.433 12q12 Rho family GTPase 1 HGD 0.433 3q13.33 homogentisate 1,2-dioxygenase PTP4A1 0.432 6q12 protein tyrosine phosphatase type IVA, member 1 C8orf4 0.428 8p11.2 chromosome 8 open reading frame 4 DDC 0.427 7p12.2 dopa decarboxylase (aromatic L-amino acid decarboxylase) TACC2 0.427 10q26 transforming, acidic coiled-coil containing protein 2 MUC13 0.422 3q21.2 mucin 13, cell surface associated C5 0.412 9q33-q34 complement component 5 NR4A2 0.412 2q22-q23 nuclear receptor subfamily 4, group A, member 2 EYS 0.411 6q12 eyes shut homolog (Drosophila) GPX2 0.406 14q24.1 glutathione peroxidase -

CEACAM3: an Innate Immune Receptor Directed Against Human-Restricted Bacterial Pathogens Stefan Pilsa, Dave T
ARTICLE IN PRESS International Journal of Medical Microbiology 298 (2008) 553–560 www.elsevier.de/ijmm CEACAM3: An innate immune receptor directed against human-restricted bacterial pathogens Stefan Pilsa, Dave T. Gerrardb,1, Axel Meyerb, Christof R. Haucka,Ã aLehrstuhl Zellbiologie, Fachbereich Biologie X908, Universita¨t Konstanz, Universita¨tsstrasse 10, 78457 Konstanz, Germany bLehrstuhl fu¨r Zoologie und Evolutionsbiologie, Fachbereich Biologie, Universita¨t Konstanz, Germany Received 15 January 2008; accepted 4 April 2008 Abstract Carcinoembryonic antigen-related cell adhesion molecule 3 (CEACAM3) is an immunoglobulin-related glycoprotein exclusively expressed on granulocytes. In contrast to other members of the CEACAM family, CEACAM3 does not support cell–cell adhesion, but rather mediates the opsonin-independent recognition and elimination of a restricted set of human-specific Gram-negative bacterial pathogens including Neisseria gonorrhoeae, Haemophilus influenzae,andMoraxella catarrhalis. Within the last 4 years, molecular determinants of CEACAM3 function and CEACAM3-initiated signaling pathways have been elucidated. Sequence comparison between CEACAM3 and other CEACAM family members points to a chimeric origin of this receptor with the bacteria- binding extracellular domain and the function-promoting intracellular domain derived from different genes. This review summarizes the current knowledge about the structure–function relationship of CEACAM3 and tries to combine these molecular aspects with a plausible scenario concerning the evolutionary origin of this phagocyte receptor in the light of host–pathogen adaptation. r 2008 Published by Elsevier GmbH. Keywords: ITAM; Signal transduction; Phagocytosis; Host–pathogen coevolution Introduction main (Beauchemin et al., 1999; Kuespert et al., 2006). In humans, the CEACAM family comprises 12 closely CEACAM3 belongs to the family of carcinoembry- related proteins, with several family members expressed onic antigen (CEA)-related cell adhesion molecules by epithelial cells (e.g. -

Supplementary Material DNA Methylation in Inflammatory Pathways Modifies the Association Between BMI and Adult-Onset Non- Atopic
Supplementary Material DNA Methylation in Inflammatory Pathways Modifies the Association between BMI and Adult-Onset Non- Atopic Asthma Ayoung Jeong 1,2, Medea Imboden 1,2, Akram Ghantous 3, Alexei Novoloaca 3, Anne-Elie Carsin 4,5,6, Manolis Kogevinas 4,5,6, Christian Schindler 1,2, Gianfranco Lovison 7, Zdenko Herceg 3, Cyrille Cuenin 3, Roel Vermeulen 8, Deborah Jarvis 9, André F. S. Amaral 9, Florian Kronenberg 10, Paolo Vineis 11,12 and Nicole Probst-Hensch 1,2,* 1 Swiss Tropical and Public Health Institute, 4051 Basel, Switzerland; [email protected] (A.J.); [email protected] (M.I.); [email protected] (C.S.) 2 Department of Public Health, University of Basel, 4001 Basel, Switzerland 3 International Agency for Research on Cancer, 69372 Lyon, France; [email protected] (A.G.); [email protected] (A.N.); [email protected] (Z.H.); [email protected] (C.C.) 4 ISGlobal, Barcelona Institute for Global Health, 08003 Barcelona, Spain; [email protected] (A.-E.C.); [email protected] (M.K.) 5 Universitat Pompeu Fabra (UPF), 08002 Barcelona, Spain 6 CIBER Epidemiología y Salud Pública (CIBERESP), 08005 Barcelona, Spain 7 Department of Economics, Business and Statistics, University of Palermo, 90128 Palermo, Italy; [email protected] 8 Environmental Epidemiology Division, Utrecht University, Institute for Risk Assessment Sciences, 3584CM Utrecht, Netherlands; [email protected] 9 Population Health and Occupational Disease, National Heart and Lung Institute, Imperial College, SW3 6LR London, UK; [email protected] (D.J.); [email protected] (A.F.S.A.) 10 Division of Genetic Epidemiology, Medical University of Innsbruck, 6020 Innsbruck, Austria; [email protected] 11 MRC-PHE Centre for Environment and Health, School of Public Health, Imperial College London, W2 1PG London, UK; [email protected] 12 Italian Institute for Genomic Medicine (IIGM), 10126 Turin, Italy * Correspondence: [email protected]; Tel.: +41-61-284-8378 Int. -

Data Sheet Anti-Human Ceacam1, 5 Monoclonal Antibody
PRODUCT DATA SHEET ANTI-HUMAN CEACAM1, 5 MONOCLONAL ANTIBODY PRODUCT INFORMATION Catalog Number: GM-0511 Clone: 4/3/17 Description: purified monoclonal mouse antibody Specificity: anti-human CEACAM1, 5 (BGP, CEA, CD66a,e) Isotype: IgG1/kappa Purification: Protein G Storage: short term: 2°C – 8°C; Buffer: phosphate buffered saline, pH 7.2 long term: –20°C (avoid repeated freezing and thawing) Immunogen: immunization with extracted protein Selection: based on recognition of the complete of CEACAM5 native protein expressed on transfected mammalian cells WORKING DILUTIONS Flow cytometry: 1.2 µg/106 cells ELISA: 1:200 – 1:400 CELISA: 1:200 Western blot: 4µg/ml Immunohistology: 1-2 µg/106 cells (on cryosections) For each application a titration should be performed to determine the optimal concentration. SPECIFICITY TESTING BY FLOW CYTOMETRY Fig. 1: FACS analysis of BOSC23 cells using 4/3/17 Cat.# GM-0511. Fig. 2: FACS analysis of BOSC23 cells using 4/3/17 Cat.# GM-0511. BOSC23 cells were transiently transfected with an expression vector encoding BOSC23 cells were transiently transfected with an expression vector encoding either CEACAM1 (red curve) or an irrelevant protein (control transfectant). either CEACAM5 (red curve) or an irrelevant protein (control transfectant). Binding of 4/3/17 was detected with a PE-conjugated secondary antibody. Binding of 4/3/17 was detected with a PE conjugated secondary antibody. A positive signal was obtained only with CEACAM1 transfected cells. A positive signal was obtained only with CEACAM5 transfected cells. CONFIDENTIAL Genovac GmbH Waltershofener Str. 17 [email protected] For research use only. Not for diagnostic or therapeutic use. -

Granulocyte CEACAM3 Is a Phagocytic Receptor of the Innate
Granulocyte CEACAM3 Is a Phagocytic Receptor of the Innate Immune System that Mediates Recognition and Elimination of Human-specific Pathogens Tim Schmitter, Franziska Agerer, Lisa Peterson, Petra Münzner, and Christof R. Hauck Zentrum für Infektionsforschung, Universität Würzburg, 97070 Würzburg, Germany Abstract Carcinoembryonic antigen-related cell adhesion molecules (CEACAMs) are used by several human pathogens to anchor themselves to or invade host cells. Interestingly, human granulocytes express a specific isoform, CEACAM3, that participates together with CEACAM1 and CEACAM6 in the recognition of CEACAM-binding microorganisms. Here we show that CEACAM3 can direct efficient, opsonin-independent phagocytosis of CEACAM-binding Neisseria, Moraxella, Downloaded from and Haemophilus species. CEACAM3- but not CEACAM6-mediated uptake is blocked by dominant-negative versions of the small GTPase Rac. Moreover, CEACAM3 engagement triggers membrane recruitment and increased GTP loading of Rac that are not observed upon bacterial binding to CEACAM6. Internalization and Rac stimulation are also inhibited by com- promising the integrity of an immunoreceptor tyrosine-based activation motif (ITAM)–like www.jem.org sequence in the cytoplasmic tail of CEACAM3 or by interference with Src family protein tyro- sine kinases that phosphorylate CEACAM3. In contrast to interfering with CEACAM6, blockage of CEACAM3-mediated events reduces the ability of primary human granulocytes to internalize and eliminate CEACAM-binding bacteria, indicating an important role of CEACAM3 in the on September 4, 2006 control of human-specific pathogens by the innate immune system. Key words: Neisseria gonorrhoeae • Opa protein • Haemophilus influenzae • Moraxella catarrhalis • innate immunity Introduction The gram-negative pathogens Neisseria gonorrhoeae, N. The four pathogenic species share several common adap- meningitidis, Moraxella catarrhalis, and Haemophilus influenzae tations such as phase or antigenic variation of surface struc- are highly adapted to the human as their sole natural host. -

In This Issue
IN THIS ISSUE J Immunol 2007; 178:3343-3344; ; doi: 10.4049/jimmunol.178.6.3343 This information is current as http://www.jimmunol.org/content/178/6/3343 of September 24, 2021. Downloaded from Why The JI? Submit online. • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists http://www.jimmunol.org/ • Fast Publication! 4 weeks from acceptance to publication *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription by guest on September 24, 2021 Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2007 by The American Association of Immunologists All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. THE JOURNAL OF IMMUNOLOGY IN THIS ISSUE Phagocytosing Neisseria with wild-type controls. A src family kinase inhibitor had effects on resting or fMLP-stimulated wild-type mouse or Ϫ Ϫ Ϫ Ϫ ram-negative Neisseria human neutrophils similar to those seen in hck / fgr / species infect mucosal mouse cells. Akt phosphorylation and calcium mobilization G surfaces by attachment were unaffected in mutant mouse cells or human cells treated with to carcinoembryonic Ag-related the inhibitor after fMLP stimulation. The authors conclude that cellular adhesion molecule 3 src family kinases hck and fgr play an essential role in NADPH (CEACAM3).