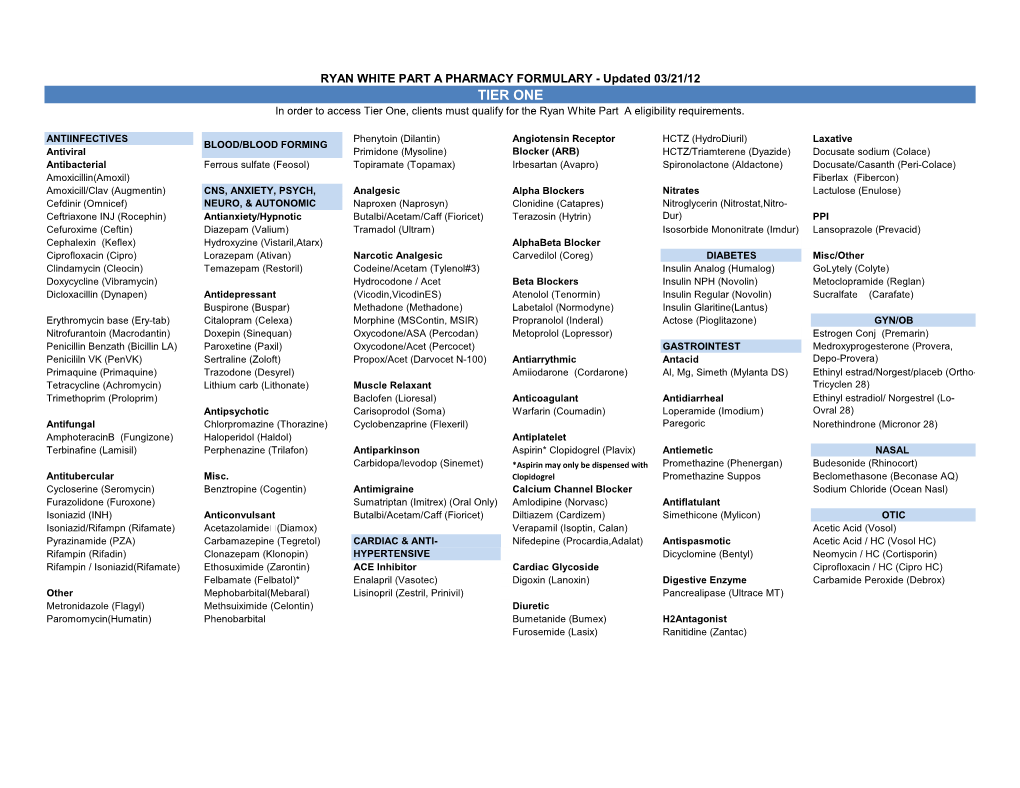
Pharmacy Formulary Tier 1
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Efficacy and Tolerability of Quinacrine Monotherapy and Albendazole Plus Chloroquine Combination Therapy in Nitroimidazole-Refractory Giardiasis: a Tropnet Study
Klinik für Infektiologie & Spitalhygiene Efficacy and tolerability of quinacrine monotherapy and albendazole plus chloroquine combination therapy in nitroimidazole-refractory giardiasis: a TropNet study Andreas Neumayr, Mirjam Schunk, Caroline Theunissen, Marjan Van Esbroeck, Matthieu Mechain, Manuel Jesús Soriano Pérez, Kristine Mørch, Peter Sothmann, Esther Künzli, Camilla Rothe, Emmanuel Bottieau Journal Club 01.03.21 Andreas Neumayr Background on giardia treatment: • 1st-line treatment: 5-nitroimidazoles: metronidazole (1957), tinidazole, ornidazole, secnidazole • cure rate of 5NIs in 1st-line treatment: ~90% • in the last decade, an increase of 5NI-refractory giardia cases has been observed in travel medicine clinics across Europe: Hospital for Tropical Diseases, London: 2008: 15% --> 2013: 40% 70% of 5NI-refractory cases imported from India • 2nd-line treatment: effectiveness of a 2nd round with a 5NI: ~17% alternative drugs: albendazole, mebendazole, nitazoxanide, quinacrine, furazolidone, chloroquine, paromomycin 2012 TropNet member survey: 53 centres use 39 different treatment regimens, consisting of 7 different drugs in mono- or combination-therapy in various dosages and durations JC 01.03.21 Nabarro LE et al. Clin Microbiol Infect. 2015;21:791-6. • by 2013, there were only 13 reports of 2nd-line therapy for giardiasis (8 case series, 5 individual case reports): n=110 Cure rates Albendazole 6/32 18.7% Paromomycin 5/17 29.4% Nitazoxanide 2/5 40.0% Albendazole + 5-NI 42/53 79.2% Quinacrine 19/21 90.5% Quinacrine + 5-NI 14/14 100% Quinacrine + Paromomycin 2/2 100% • 2013: TropNet "GiardiaREF" study kick-off: Study on efficacy and tolerability of two 2nd-line regimens in nitroimidazole-refractory giardiasis: Quinacrine JC 01.03.21 Meltzer E et al. -

Jos Journal 2
POST-OPERATIVE AUDIT OF G6PD-DEFICIENT MALE CHILDREN WITH OBSTRUCTIVE ADENOTONSILLAR ENLARGEMENT AT UNIVERSITY COLLEGE HOSPITAL, IBADAN, NIGERIA. John EN1, Totyen EL1, Jacob N2, Nwaorgu OGB1 1 .Department of ENT/Head and Neck Surgery, University College Hospital, Ibadan, Nigeria 2. Department of paediatrics, University College Hospital, Ibadan, Nigeria All correspondences and request for reprint to Dr John EN, Department of ENT/Head and Neck Surgery, University College Hospital, Ibadan, Nigeria Email: [email protected] Telephone: +2348036240109 Abstract Background: G6PD deficiency ranks among the commonest hereditary enzyme deficiency worldwide and notable as a predisposing condition to haemolyticcrises. The fear of possible untoward effects is often expressed by parents of G6PD deficient male children scheduled for surgery after obtaining an informed and understood consent. The parental perception of obstructive adenotonsillar enlargement in this condition was also appraised. Methods: A retrospective chart review of all G6PD deficient male children between ages 1 to 7years who had adenotonsillectomy over a 3year period at University college Hospital, Ibadan, Nigeria. Results: The patients comprised of 22 G6PD deficient male children diagnosed shortly after birth upon development of neonatal jaundice. Fifteen(68.2%) and 6(27.3%) of the patients subsequently developed episodes of drug- induced haemolysis and non-haemolytic drug reactions prior to undergoing adenotonsillectomy by the otolaryngologists. None of the patients was observed to develop haemolytic crises up to 2weeks post-adenotonsillectomy. From the parental perception and responses in the follow-up period,all 22(100%) patient had resolution of noisy breathing, 20(91%) had improvement of snoring and apnoeic spells. Only 15 (68%) were reported to stop mouth-breathing. -

PROVERA Medroxyprogesterone Acetate Tablets 2.5Mg, 5Mg, 10Mg, 100 Mg, 200 Mg Tablets
PROVERA Medroxyprogesterone acetate tablets 2.5mg, 5mg, 10mg, 100 mg, 200 mg tablets What is in this leaflet establish a regular menstrual breast cancer or breast lumps cycle not diagnosed by your doctor This leaflet answers some common bleeding or discharge from questions about PROVERA. It does certain types of cancer including your nipples not contain all the available cancer of the breast, kidney and information. It does not take the endometrium (the lining of the miscarriage place of talking to your doctor or womb). pharmacist. cancer of the womb or ovary PROVERA, in combination with an uncontrolled high blood estrogen containing medicine, is All medicines have risks and pressure. benefits. Your doctor has weighed used to relieve symptoms of the risks of you taking PROVERA menopause in women with an intact Do not take PROVERA if you are against the benefits it is expected to uterus. This is called hormone pregnant or intend to become have for you. replacement therapy (HRT). pregnant. PROVERA is used to protect the If you have any concerns about lining of the uterus while the PROVERA may affect your taking this medicine, ask your estrogens relieve the symptoms of developing baby if you take it doctor or pharmacist. Keep this menopause. PROVERA is not during pregnancy. leaflet with your medicine. suitable as a HRT treatment in women who have undergone a Do not take PROVERA if the You may need to read it again. hysterectomy. packaging is torn or shows signs of tampering. Do not take Your doctor may have prescribed PROVERA after the expiry date What PROVERA is PROVERA for another purpose. -

Liquid Spray Formulations for Buccal Delivery of Cannabinoids
(19) TZZ_¥__T (11) EP 1 361 864 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: A61K 9/08 (2006.01) A61K 9/107 (2006.01) 04.12.2013 Bulletin 2013/49 A61K 36/185 (2006.01) (21) Application number: 02712063.3 (86) International application number: PCT/GB2002/000620 (22) Date of filing: 14.02.2002 (87) International publication number: WO 2002/064109 (22.08.2002 Gazette 2002/34) (54) LIQUID SPRAY FORMULATIONS FOR BUCCAL DELIVERY OF CANNABINOIDS FLÜSSIGE SPRAY FORMULIERUNGEN ZUR BUCCALEN VERABREICHUNG VON CANNABINOIDEN PREPARATIONS DE SPRAY LIQUIDE POUR L’ADMINISTRATION BUCCALE DE CANNABINOIDES (84) Designated Contracting States: (74) Representative: Schiller, Dominic Christopher et AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU al MC NL PT SE TR Harrison Goddard Foote LLP Designated Extension States: 4th Floor, Merchant Exchange RO SI 17-19 Whitworth Street West Manchester M1 5WG (GB) (30) Priority: 14.02.2001 GB 0103638 30.03.2001 US 280044 P (56) References cited: 05.04.2001 US 827158 WO-A-00/25127 WO-A-01/66089 11.05.2001 GB 0111597 WO-A-95/25504 WO-A1-02/32420 07.09.2001 GB 0121715 WO-A1-02/069993 US-A- 3 560 625 12.09.2001 US 951022 • EITAN ALHANATY; ET AL.: "Osmotic fragility of (43) Date of publication of application: liposomes as affected by antihemolytic 19.11.2003 Bulletin 2003/47 compounds" BIOCHIMICA ET BIOPHYSICA ACTA, vol. 339, no. 1, 1974, pages 146-155, (60) Divisional application: XP008008726 10180611.5 / 2 286 793 • PATRICIA S. -

Student Members of the American Chemical Society Poster Presentation Event November 6, 2020 5:30 - 7:00 Pm Stonecipher Lecture Hall
Student Members of the American Chemical Society Poster Presentation Event November 6, 2020 5:30 - 7:00 pm Stonecipher Lecture Hall Author: Brooke Underwood*, Courtney LaPointe, Dr. Jeffrey Boles Contact information: [email protected] Title: Liquid-liquid extraction and ultraviolet visible spectroscopy methods for distinguishing between hemp and marijuana Abstract: In December 2018, cannabis containing less than 0.3% tetrahydrocannabinol (THC), otherwise known as hemp, became legalized due to passage of the Farm Bill (1). This creates problems for law enforcement since current presumptive test kits either 1) don’t work at all or 2) work somewhat in differentiating between legal and illegal hemp crops. This problem exists because most hemp crops and hemp products contain low levels of THC and the carboxylated form, THCA. Our approach involves the advancement of an efficient, mobile, liquid-liquid extraction (LLE) that provides presumptive, qualitative forensic evidence of the chemical extract of a bud or other plant material. This research is focused on developing a kit that functions in a similar manner to NIK kits, commonly used by law enforcement, where all components of the kit are contained within a bag. The current NIK kit for Marijuana provides a false positive when Hemp is placed in the bag, thus creating the need for a more reliable test (2). The evidence would later be sent to a crime lab for definitive analysis and quantitation of THC by ultraviolet-visible spectroscopy (UV-vis). This research has focused on the utilization of liquid-liquid extraction techniques and commercially available stains. The methods presented are rapid (requiring no more than five to six minutes to complete). -

Annex 4: Drug Dosages for Children (Formulary)
Medicines Dosage Form Dose according to body weight (calculate if weight is below or over) 3-6 kg 6-10 kg 10-15 kg 15-20 kg 20-29 kg albendazole 200 mg (half tablet) 12-24 months chewable tablet, 400mg 400 mg (one tablet) over 24 months amodiaquine 10 mg base/kg/3 days (total dose 30 mg base/kg) tablet, 200mg - - 1 1 1 amoxicillin 15 mg/kg/dose for 7 days tablet/capsule 250 mg ¼ ½ ¾ 1 1½ oral suspension, 125mg/5ml 2.5 ml 5 ml 7.5 ml 10 ml - non-severe pneumonia: 25 mg/kg 2 times per day for 3 days tablet/capsule 250 mg ½ 1 1½ 2 2½ oral suspension, 125mg/5ml 5 ml 10 ml 15 ml - - ampicillin IM 50 mg/kg/6 hours Vial of 500 mg mixed with 2.1 ml 1 ml 2 ml 3 ml 5 ml 6 ml sterile water to give 500 mg/2.5 ml artemether IM 3.2 mg/kg once on day 1 injection, 40mg/ml in 1ml ampoule then injection, 80mg/ml in 1ml ampoule see Chapter 5, management of the child with malaria IM 1.6 mg/kg daily until oral therapy is possible, total therapy one week artemether + fixed dose treatment (20+120 mg) twice daily for 3 days tablet 10+120 mg see Chapter 5, management of the child with malaria lumefantrine artesunate severe malaria: IV or IM 2.4 mg/kg over 3 minutes at 0, 12 and 24 vial of 60 mg in 0.6 ml with 3.4 ml hours on day 1. -

INTEGRIS Formulary July 2017
INTEGRIS Formulary July 2017 Foreword FORMULARY EXCLUDED THERAPEUTIC DRUG This document represents the efforts of the MedImpact Healthcare Systems THERAPEUTIC DRUGS CLASS Pharmacy and Therapeutics (P & T) and Formulary Committees to provide ALTERNATIVES physicians and pharmacists with a method to evaluate the safety, efficacy and cost- clindamycin/tretinoin, ACNE AGENTS, effectiveness of commercially available drug products. A structured approach to the VELTIN drug selection process is essential in ensuring continuing patient access to rational ZIANA TOPICAL drug therapies. The ultimate goal of the MedPerform Formulary is to provide a morphine sulfate ER process and framework to support the dynamic evolution of this document to guide tablets, oxycodone ANALGESICS, KADIAN prescribing decisions that reflect the most current clinical consensus associated ER, NUCYNTA, NARCOTICS with drug therapy decisions. NUCYNTA ER ANALGESICS, This is accomplished through the auspices of the MedImpact P & T and Formulary BELBUCA BUTRANS PATCH Committees. These committees meet quarterly and more often as warranted to NARCOTICS ensure clinical relevancy of the Formulary. To accommodate changes to this ABSTRAL, document, updates are made accessible as necessary. FENTORA, fentanyl citrate ANALGESICS, LAZANDA, lozenge NARCOTICS As you use this Formulary, you are encouraged to review the information and ONSOLIS, provide your input and comments to the MedImpact P & T and Formulary SUBSYS Committees. immediate-release GRALISE ANTICONVULSANTS The MedImpact P & T -
Medperform Low Formulary July 2017
MedPerform Low Formulary July 2017 Foreword FORMULARY This document represents the efforts of the MedImpact Healthcare Systems EXCLUDED THERAPEUTIC THERAPEUTIC DRUG CLASS Pharmacy and Therapeutics (P & T) and Formulary Committees to provide DRUGS physicians and pharmacists with a method to evaluate the safety, efficacy and cost- ALTERNATIVES effectiveness of commercially available drug products. A structured approach to the clindamycin/tretinoin, VELTIN ACNE AGENTS, TOPICAL drug selection process is essential in ensuring continuing patient access to rational ZIANA drug therapies. The ultimate goal of the MedPerform Formulary is to provide a morphine sulfate ER process and framework to support the dynamic evolution of this document to guide tablets, oxycodone ER, ANALGESICS, prescribing decisions that reflect the most current clinical consensus associated KADIAN with drug therapy decisions. NUCYNTA, NUCYNTA NARCOTICS ER This is accomplished through the auspices of the MedImpact P & T and Formulary ANALGESICS, BELBUCA BUTRANS PATCH Committees. These committees meet quarterly and more often as warranted to NARCOTICS ensure clinical relevancy of the Formulary. To accommodate changes to this ABSTRAL, document, updates are made accessible as necessary. FENTORA, ANALGESICS, LAZANDA, fentanyl citrate lozenge As you use this Formulary, you are encouraged to review the information and NARCOTICS provide your input and comments to the MedImpact P & T and Formulary ONSOLIS, Committees. SUBSYS immediate-release GRALISE ANTICONVULSANTS The MedImpact -

Vaginal Administration of Contraceptives
Scientia Pharmaceutica Review Vaginal Administration of Contraceptives Esmat Jalalvandi 1,*, Hafez Jafari 2 , Christiani A. Amorim 3 , Denise Freitas Siqueira Petri 4 , Lei Nie 5,* and Amin Shavandi 2,* 1 School of Engineering and Physical Sciences, Heriot-Watt University, Edinburgh EH14 4AS, UK 2 BioMatter Unit, École Polytechnique de Bruxelles, Université Libre de Bruxelles, Avenue F.D. Roosevelt, 50-CP 165/61, 1050 Brussels, Belgium; [email protected] 3 Pôle de Recherche en Gynécologie, Institut de Recherche Expérimentale et Clinique, Université Catholique de Louvain, 1200 Brussels, Belgium; [email protected] 4 Fundamental Chemistry Department, Institute of Chemistry, University of São Paulo, Av. Prof. Lineu Prestes 748, São Paulo 05508-000, Brazil; [email protected] 5 College of Life Sciences, Xinyang Normal University, Xinyang 464000, China * Correspondence: [email protected] (E.J.); [email protected] (L.N.); [email protected] (A.S.); Tel.: +32-2-650-3681 (A.S.) Abstract: While contraceptive drugs have enabled many people to decide when they want to have a baby, more than 100 million unintended pregnancies each year in the world may indicate the contraceptive requirement of many people has not been well addressed yet. The vagina is a well- established and practical route for the delivery of various pharmacological molecules, including contraceptives. This review aims to present an overview of different contraceptive methods focusing on the vaginal route of delivery for contraceptives, including current developments, discussing the potentials and limitations of the modern methods, designs, and how well each method performs for delivering the contraceptives and preventing pregnancy. -

Protocol for Look-Alike and Sound-Alike Drugs
Community Mental Health for Central Michigan PROTOCOL FOR LOOK-ALIKE AND SOUND-ALIKE DRUGS This protocol should be posted in all licensed residential group homes who contract with Community Mental Health for Central Michigan ADMINISTRATIVE GUIDELINE In an effort to improve medication safety and to meet The Joint Commission’s National Patient Safety Goal Number 3, Community Mental Health for Central Michigan (CMHCM) has identified a process to address sound-alike and look-alike drugs. The Performance Improvement Team that developed this guideline will continue to monitor new drugs on the market as they are released, review the list included with this guideline on an annual basis, and provide appropriate updates. Appropriate action to prevent errors involving the interchange of these drugs will be taken. Each CMHCM site (including direct and provider network sites) where medication is distributed or administered will post the attached lists and implement a plan for preventing drug mix-ups. This plan may consist of but not be limited to: Listing both the brand and generic names on medication records. Storing products with look-alike or sound-alike names in different locations. Employing double checks in the distribution process. Affixing “name alert” stickers to areas where look-alike or sound-alike products are stored. Changing the appearance of look-alike product names on pharmacy labels, computer screens, shelf labels and bins, and medication records by highlighting, through bold face, color, and/or tall man letters, the parts of the names that are different (e.g. hydrOXYzine, hydrALAzine). Having physicians write prescriptions using both the brand and generic names. -

Managing Opioid-Induced Constipation
Visual summary MODIFY PAIN RELIEF Managing opioid-induced constipation Is the current dose of opioid needed to control pain? Constipation affects many patients using opioids Could pain be relieved in other ways? to relieve pain associated with advanced cancer Non-opioid analgesics Interventional pain management or terminal disease. Constipation is often multifactorial in such patients, Could an opioid with less constipating effect be used? and opioids may be one of several causes. As well Buprenorphine Transdermal fentanyl as direct treatment of the constipation, adjusting medication regimens and treating exacerbating factors may help to provide relief from bowel transit Is a referral to other specialists needed? symptoms. Palliative care Neuro-gastroenterology Pain clinic ADDRESS EXACERBATING FACTORS DIRECTLY TREAT CONSTIPATION Drugs Non-pharmacological options 5HT3 antagonists Anticholinergics Opioids Increase fluid consumption Increase dietary fibres Antipsychotics Diuretics Iron Increase physical activity (within patients’ capabilities) Calcium supplements Calcium channel blockers Privacy and comfort during defecation Chemotherapies Complementary therapy Thalidomide Vinca alkaloids Positioning on toilet (to relax the puborectalis muscle) knees higher than hips, leaning forward Busulfan Carboplatin with elbows on knees, straightened spine Manual removal Nutritional and if the constipation is severe and refractory to other therapies metabolic issues Pharmacological management Dehydration Reduced food and fluids intake Oral laxatives Poor -

2021 Formulary List of Covered Prescription Drugs
2021 Formulary List of covered prescription drugs This drug list applies to all Individual HMO products and the following Small Group HMO products: Sharp Platinum 90 Performance HMO, Sharp Platinum 90 Performance HMO AI-AN, Sharp Platinum 90 Premier HMO, Sharp Platinum 90 Premier HMO AI-AN, Sharp Gold 80 Performance HMO, Sharp Gold 80 Performance HMO AI-AN, Sharp Gold 80 Premier HMO, Sharp Gold 80 Premier HMO AI-AN, Sharp Silver 70 Performance HMO, Sharp Silver 70 Performance HMO AI-AN, Sharp Silver 70 Premier HMO, Sharp Silver 70 Premier HMO AI-AN, Sharp Silver 73 Performance HMO, Sharp Silver 73 Premier HMO, Sharp Silver 87 Performance HMO, Sharp Silver 87 Premier HMO, Sharp Silver 94 Performance HMO, Sharp Silver 94 Premier HMO, Sharp Bronze 60 Performance HMO, Sharp Bronze 60 Performance HMO AI-AN, Sharp Bronze 60 Premier HDHP HMO, Sharp Bronze 60 Premier HDHP HMO AI-AN, Sharp Minimum Coverage Performance HMO, Sharp $0 Cost Share Performance HMO AI-AN, Sharp $0 Cost Share Premier HMO AI-AN, Sharp Silver 70 Off Exchange Performance HMO, Sharp Silver 70 Off Exchange Premier HMO, Sharp Performance Platinum 90 HMO 0/15 + Child Dental, Sharp Premier Platinum 90 HMO 0/20 + Child Dental, Sharp Performance Gold 80 HMO 350 /25 + Child Dental, Sharp Premier Gold 80 HMO 250/35 + Child Dental, Sharp Performance Silver 70 HMO 2250/50 + Child Dental, Sharp Premier Silver 70 HMO 2250/55 + Child Dental, Sharp Premier Silver 70 HDHP HMO 2500/20% + Child Dental, Sharp Performance Bronze 60 HMO 6300/65 + Child Dental, Sharp Premier Bronze 60 HDHP HMO