Intracranial Penetration During Temporal Soft Tissue Filler Injection—Is It Possible?
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-
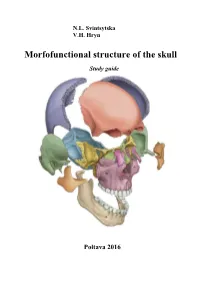
Morfofunctional Structure of the Skull
N.L. Svintsytska V.H. Hryn Morfofunctional structure of the skull Study guide Poltava 2016 Ministry of Public Health of Ukraine Public Institution «Central Methodological Office for Higher Medical Education of MPH of Ukraine» Higher State Educational Establishment of Ukraine «Ukranian Medical Stomatological Academy» N.L. Svintsytska, V.H. Hryn Morfofunctional structure of the skull Study guide Poltava 2016 2 LBC 28.706 UDC 611.714/716 S 24 «Recommended by the Ministry of Health of Ukraine as textbook for English- speaking students of higher educational institutions of the MPH of Ukraine» (minutes of the meeting of the Commission for the organization of training and methodical literature for the persons enrolled in higher medical (pharmaceutical) educational establishments of postgraduate education MPH of Ukraine, from 02.06.2016 №2). Letter of the MPH of Ukraine of 11.07.2016 № 08.01-30/17321 Composed by: N.L. Svintsytska, Associate Professor at the Department of Human Anatomy of Higher State Educational Establishment of Ukraine «Ukrainian Medical Stomatological Academy», PhD in Medicine, Associate Professor V.H. Hryn, Associate Professor at the Department of Human Anatomy of Higher State Educational Establishment of Ukraine «Ukrainian Medical Stomatological Academy», PhD in Medicine, Associate Professor This textbook is intended for undergraduate, postgraduate students and continuing education of health care professionals in a variety of clinical disciplines (medicine, pediatrics, dentistry) as it includes the basic concepts of human anatomy of the skull in adults and newborns. Rewiewed by: O.M. Slobodian, Head of the Department of Anatomy, Topographic Anatomy and Operative Surgery of Higher State Educational Establishment of Ukraine «Bukovinian State Medical University», Doctor of Medical Sciences, Professor M.V. -

Morphology of the Pterion in Serbian Population
Int. J. Morphol., 38(4):820-824, 2020. Morphology of the Pterion in Serbian Population Morfología del Pterion en Población Serbia Knezi Nikola1; Stojsic Dzunja Ljubica1; Adjic Ivan2; Maric Dusica1 & Pupovac Nikolina4 KNEZI, N.; STOJSIC, D. L.; ADJIC, I.; MARIC, D. & PUPOVAC, N. Morphology of the pterion in Serbian population. Int. J. Morphol., 38(4):820-824, 2020. SUMMARY: The pterion is a topographic point on the lateral aspect of the skull where frontal, sphenoid, parietal and temporal bones form the H or K shaped suture. This is an important surgical point for the lesions in anterior and middle cranial fossa. This study was performed on 50 dry skulls from Serbian adult individuals from Department of Anatomy, Faculty of Medicine in Novi Sad. The type of the pterion on both sides of each skull was determined and they are calcified in four types (sphenoparietal, frontotemporal, stellate and epipteric). The distance between the center of the pterion and defined anthropological landmarks were measured using the ImageJ software. Sphenoparietal type is predominant with 86 % in right side and 88 % in left side. In male skulls, the distance from the right pterion to the frontozygomatic suture is 39.89±3.85 mm and 39.67±4.61 mm from the left pterion to the frontozygomatic suture. In female skulls the distance is 37.38±6.38 mm on the right and 35.94±6.46 mm on the left. The shape and the localization of the pterion are important because it is an anatomical landmark and should be used in neurosurgery, traumatology and ophthalmology. -

CLOSURE of CRANIAL ARTICULATIONS in the SKULI1 of the AUSTRALIAN ABORIGINE by A
CLOSURE OF CRANIAL ARTICULATIONS IN THE SKULI1 OF THE AUSTRALIAN ABORIGINE By A. A. ABBIE, Department of Anatomy, University of Adelaide INTRODUCTION While it is well known that joint closure advances more or less progressively with age, there is still little certainty in matters of detail, mainly for lack of adequate series of documented skulls. In consequence, sundry beliefs have arisen which tend to confuse the issue. One view, now disposed of (see Martin, 1928), is that early suture closure indicates a lower or more primitive type of brain. A corollary, due to Broca (see Topinard, 1890), that the more the brain is exercised the more is suture closure postponed, is equally untenable. A very widespread belief is based on Gratiolet's statement (see Topinard, 1890; Frederic, 1906; Martin, 1928; Fenner, 1939; and others) that in 'lower' skulls the sutures are simple and commence to fuse from in front, while in 'higher' skulls the sutures are more complicated and tend to fuse from behind. This view was disproved by Ribbe (quoted from Frederic, 1906), who substituted the generalization that in dolicocephals synostosis begins in the coronal suture, and in brachycephals in the lambdoid suture. In addition to its purely anthropological interest the subject raises important biological considerations of brain-skull relationship, different foetalization in different ethnological groups (see Bolk, 1926; Weidenreich, 1941; Abbie, 1947), and so on. A survey of the literature reveals very little in the way of data on the age incidence of suture closure. The only substantial contribution accessible here comes from Todd & Lyon (1924) for Europeans, but their work is marred by arbitrary rejection of awkward material. -

MBB: Head & Neck Anatomy
MBB: Head & Neck Anatomy Skull Osteology • This is a comprehensive guide of all the skull features you must know by the practical exam. • Many of these structures will be presented multiple times during upcoming labs. • This PowerPoint Handout is the resource you will use during lab when you have access to skulls. Mind, Brain & Behavior 2021 Osteology of the Skull Slide Title Slide Number Slide Title Slide Number Ethmoid Slide 3 Paranasal Sinuses Slide 19 Vomer, Nasal Bone, and Inferior Turbinate (Concha) Slide4 Paranasal Sinus Imaging Slide 20 Lacrimal and Palatine Bones Slide 5 Paranasal Sinus Imaging (Sagittal Section) Slide 21 Zygomatic Bone Slide 6 Skull Sutures Slide 22 Frontal Bone Slide 7 Foramen RevieW Slide 23 Mandible Slide 8 Skull Subdivisions Slide 24 Maxilla Slide 9 Sphenoid Bone Slide 10 Skull Subdivisions: Viscerocranium Slide 25 Temporal Bone Slide 11 Skull Subdivisions: Neurocranium Slide 26 Temporal Bone (Continued) Slide 12 Cranial Base: Cranial Fossae Slide 27 Temporal Bone (Middle Ear Cavity and Facial Canal) Slide 13 Skull Development: Intramembranous vs Endochondral Slide 28 Occipital Bone Slide 14 Ossification Structures/Spaces Formed by More Than One Bone Slide 15 Intramembranous Ossification: Fontanelles Slide 29 Structures/Apertures Formed by More Than One Bone Slide 16 Intramembranous Ossification: Craniosynostosis Slide 30 Nasal Septum Slide 17 Endochondral Ossification Slide 31 Infratemporal Fossa & Pterygopalatine Fossa Slide 18 Achondroplasia and Skull Growth Slide 32 Ethmoid • Cribriform plate/foramina -

MORPHOMETRIC STUDY of PTERION in DRY ADULT HUMAN SKULLS Pratima Kulkarni 1, Shivaji Sukre 2, Mrunal Muley *3
International Journal of Anatomy and Research, Int J Anat Res 2017, Vol 5(3.3):4365-68. ISSN 2321-4287 Original Research Article DOI: https://dx.doi.org/10.16965/ijar.2017.337 MORPHOMETRIC STUDY OF PTERION IN DRY ADULT HUMAN SKULLS Pratima Kulkarni 1, Shivaji Sukre 2, Mrunal Muley *3. 1 Associate Professor, Department of Anatomy, G.M.C. Aurangabad, Maharashtra, India. 2 Professor and Head of department, Department of Anatomy, G.M.C. Aurangabad, Maharashtra, India. *3 Assistant Professor, Department of Anatomy, G.M.C. Aurangabad, Maharashtra, India. ABSTRACT Introduction: The pterion corresponds to the site of anterolateral fontanelle of the neonatal skull which closes at third month after birth. In the pterional fractures the anterior and middle meningeal arterial ramus ruptures commonly which results in extradural hemorrhage. Pterional approach is most suitable and minimally invasive approach in neurosurgery. Materials and Methods: The present study was carried out on the pterion of 36 dry adult skulls of known sex from department of anatomy GMC Aurangabad Maharashtra. Results: The mean and standard deviation of the distance between the centre of pterion to various anatomical landmarks. The distance between Pterion- frontozygomatic (P-FZ) suture 29.81±4.42mm on right side, 29.81±4.07mm on left side; Pterion-Zygomatic arch (P-Z) 37.16±3.77mm on right side, 37.56±3.71mm on left side, Pterion-asterion (P-A) 89.73±6.16mm on right side, 89.46±6.35mm on left side; Pterion-external acoustic meatus (P- EAM) 53.40±7.28mm on right side, 53.57±6.73mm on left side, Pterion- Mastoid process (P-M) 80.35±3.44mm on right side, 80.96±3.79mm on left side and Pterion- Pterion (P-P) 194.54±16.39mm were measured. -

Ectocranial Suture Closure in Pan Troglodytes and Gorilla Gorilla: Pattern and Phylogeny James Cray Jr.,1* Richard S
AMERICAN JOURNAL OF PHYSICAL ANTHROPOLOGY 136:394–399 (2008) Ectocranial Suture Closure in Pan troglodytes and Gorilla gorilla: Pattern and Phylogeny James Cray Jr.,1* Richard S. Meindl,2 Chet C. Sherwood,3 and C. Owen Lovejoy2 1Department of Anthropology, University of Pittsburgh, Pittsburgh, PA 15260 2Department of Anthropology and Division of Biomedical Sciences, Kent State University, Kent, OH 44242 3Department of Anthropology, The George Washington University, Washington, DC 20052 KEY WORDS cranial suture; synostosis; variation; phylogeny; Guttman analysis ABSTRACT The order in which ectocranial sutures than either does with G. gorilla, we hypothesized that this undergo fusion displays species-specific variation among phylogenetic relationship would be reflected in the suture primates. However, the precise relationship between suture closure patterns of these three taxa. Results indicated that closure and phylogenetic affinities is poorly understood. In while all three species do share a similar lateral-anterior this study, we used Guttman Scaling to determine if the closure pattern, G. gorilla exhibits a unique vault pattern, modal progression of suture closure differs among Homo which, unlike humans and P. troglodyte s, follows a strong sapiens, Pan troglodytes,andGorilla gorilla.BecauseDNA posterior-to-anterior gradient. P. troglodytes is therefore sequence homologies strongly suggest that P. tr og lodytes more like Homo sapiens in suture synostosis. Am J Phys and Homo sapiens share a more recent common ancestor Anthropol 136:394–399, 2008. VC 2008 Wiley-Liss, Inc. The biological basis of suture synostosis is currently Morriss-Kay et al. (2001) found that maintenance of pro- poorly understood, but appears to be influenced by a liferating osteogenic stem cells at the margins of mem- combination of vascular, hormonal, genetic, mechanical, brane bones forming the coronal suture requires FGF and local factors (see review in Cohen, 1993). -

Foramina, Fossa and Vacuities in the Skull and Lower Jaw of Mud Turtle, Trionyx Gangeticus (Cuv.) by D
FORAMINA, FOSSA AND VACUITIES IN THE SKULL AND LOWER JAW OF MUD TURTLE, TRIONYX GANGETICUS (CUV.) BY D. K. MANSHARAMANI (Department of Zoology, ttolkar Science College, lndore) Received March 3, 1965 (Communicated by Dr. Benicharan Mahendra, F.A.SC.) As far as the author is aware, no work has been done on the skull of mud turtles specially the foramina, fossa and vacuities. I have therefore studied the cranial peculiarities of Trionyx gangeticus, with special reference to foramina, fossa and vacuities of the skull and lower jaw. Trionyx gangeticus, a monotypic, trionychid testudine, exhibits many foramina, fossa and vacuities in its skull, which is typically akinetic moni- mostylic and anapsidian. The latter condition undergoes partial modifi- cation in the shape of temporal region which reveals emargination. The temporal arch is formed by jugal and quadratojugal. The orbits are close to the anterior half separated by prefrontal on the dorsal side. The anterior nares are near the tip of the snout, bounded by prcunaxilla below and prefrontal above. The skull is oblong-swollen, nose convex-arched, forehead convex, upper jaw'with broad flat rugose alveolar plate, which is narrow in front and wide behind. It has three long posterior processes formed by supra- occipital in the middle and squamosals on either side. The premaxilla is extremely small, unpaired and does not reach the nasal cavity or the vomer. The maxillaries are correspondingly enlarged surrounding the choanae, which are separated by narrow vomer. The palatines form a broad deep concavity which is joined behind by long basi-sphenoid, which separates the long pterygoids from each other. -

Endoscopic Access to the Infratemporal Fossa and Skull Base a Cadaveric Study
ORIGINAL ARTICLE Endoscopic Access to the Infratemporal Fossa and Skull Base A Cadaveric Study Christopher J. Hartnick, MD; John S. Myseros, MD; Charles M. Myer III, MD Objectives: To demonstrate that the regions of the in- Endoscopic visualization and instrumentation was then fratemporal fossa and skull base at the level of the fora- performed. The infratemporal fossa was readily identi- men ovale can be visualized endoscopically and that struc- fied. The skull base at the level of the foramen ovale and tures can be manipulated within these regions using the branches of the third division of the trigeminal nerve endoscopic instruments. were seen distinctly. A probe was placed with ease within the foramen ovale itself. Methods: Cadaveric dissection of 3 human cadavers us- ing an endoscopic optical dissector. In all, 6 endoscopic Conclusions: Endoscopic access to the infratemporal infratemporal fossa and skull base approaches were per- fossa is readily accomplished, with excellent visualiza- formed. tion and instrumentation ability. This novel technique provides access to this remote region for evaluation, pos- Setting: Human temporal bone laboratory. sible biopsy, and potential treatment of infratemporal fossa lesions. Results: A Gillies incision was coupled with a lateral brow incision, and then subperiosteal planes were developed. Arch Otolaryngol Head Neck Surg. 2001;127:1325-1327 HE INFRATEMPORAL fossa is lymphoma, and juvenile nasopharyngeal a relatively remote region angiofibroma. Many of these tumors can beneath the skull base. Ac- undergo biopsy at some other, more readily cess to this region requires accessible area, or the diagnosis is se- thorough knowledge of the cured using imaging studies (computed Tanatomy of the region itself and of the sur- tomography or magnetic resonance rounding structures. -

A Study on Sutural Morphology and Anatomical Position of Pterion
A STUDY ON SUTURAL MORPHOLOGY AND ANATOMICAL POSITION OF PTERION Varsha Shenoy, P. Saraswathi, T. Siva, D. Jagadeesh ijcrr Department of Anatomy, Saveetha Medical College and Hospital, Saveetha Vol 04 issue 09 University, Saveetha Nagar, Thandalam, Chennai, Tamil Nadu Category: Research Received on:21/03/12 Revised on:29/03/12 E-mail of Corresponding Author: [email protected] Accepted on:06/04/12 ABSTRACT Objective: Pterion is an interesting bony landmark on the lateral aspect of the skull. It is ‗H‘ shaped sutural confluence formed by the articulation of four bones: frontal, parietal, greater wing of sphenoid and squamous part of temporal bone. Pterional approach is most widely used in neurosurgery for a variety of lesions in the anterior and middle cranial fossae. Since previous studies have reported the racial difference in the pterional morphology, in the present study it was aimed to study the sutural morphology of pteria and also to locate it with respect to the frontozygomatic suture. Methods: Seventy five (50 male and 25 female) skulls were studied on both the sides to evaluate the sutural pattern of pteria. The distance between center of the pterion to frontozygomatic suture was measured on both the sides. Results and Conclusion: According to the present study Sphenoparietal pterion was the commonest (77.33%) finding and was followed in frequency by Epipteric type (21.33%) and Stellate type (1.33%). Frontotemporal pterion was not observed. Average distance between the center of pterion and frontozygomatic suture was found to be 2.95 cm. ____________________________________________________________________________________ Keywords: Pterion, Frontozygomatic suture, neuro-vascular disorders located in the anterior Burr-hole surgery, sutural bone, fontanelle, and middle cranial fossae. -

Anthropometric Evaluation of Pterion in Dry Human Skulls Found in Southern India
Jemds.com Original Research Article Anthropometric Evaluation of Pterion in Dry Human Skulls Found in Southern India Pretty Rathnakar1, Remya Vinod2, Swathi3, Anuja Sinha4 1Associate Professor, Department of Anatomy, K. S. Hegde Medical Academy, Mangalore, Karnataka, India. 2Lecturer, Department of Anatomy, K. S. Hegde Medical Academy, Mangalore, Karnataka, India. 3Associate Professor, Department of Anatomy, K. S. Hegde Medical Academy, Mangalore, Karnataka, India. 4Assistant Professor, Department of Anatomy, K. S. Hegde Medical Academy, Mangalore, Karnataka, India. ABSTRACT BACKGROUND Pterion is a H- shaped sutural convergence seen in the Norma Lateralis of skull. After Corresponding Author: 2-3 months of birth, the anterolateral fontanelle in the neonatal skulls close to form Remya Vinod, Department of Anatomy, the pterion. It is the meeting point of four bones sphenoid, parietal, temporal and K. S. Hegde Medical Academy, frontal. Four types have been noted- spheno-parietal, fronto-temporal, epipteric and Deralakatte, Mangalore-575018, stellate. Pterional approach is commonly undertaken in surgical management of Karnataka, India. tumours involving inferior aspects of frontal lobe, like olfactory meningiomas, orbital, E-mail: [email protected] retro-orbital, sellar, chiasmatic, subfrontal, prepontine areas, anterior circulation and basilar artery aneurysm. The knowledge regarding the various shapes and distances DOI: 10.14260/jemds/2019/541 from different points to pterion (distance of centre of pterion was calculated from mid-point of superior margin of zygomatic arch (PZA), Frontozygomatic suture (PFZ), Financial or Other Competing Interests: None. tip of the mastoid process (PMP), and anterosuperior margin of external acoustic meatus (PEAM)) is useful for treating number of pathologies in brain. So, this is also How to Cite This Article: useful for neurosurgeons, anatomists, anthropologists and forensic medicine Rathnakar P, Vinod R, Swathi, et al. -
Morphometric Study of Different Types of Pterion and It's Relation With
Anatomy Praba Antony Mary A and Venkatramaniah / JPBMS, 2012, 21 (04) Available online at www.jpbms.info ISSN NO- 2230 – 7885 CODEN JPBSCT ResearchJPBMS article NLM Title: J Pharm Biomed Sci. JOURNAL OF PHARMACEUTICAL AND BIOMEDICAL SCIENCES Morphometric Study of different types of Pterion and It’s relation with middle meningeal artery in dry skulls of Tamil Nadu * A.Mary Antony Praba¹, C.Venkatramaniah². ¹Assistant Professor, Department of Anatomy, Tagore Medical College, Chennai, India. ²Assistant Professor, Department of Anatomy, Sri Lakshmi Narayana Institute of Medical Sciences, Pondy, India. Abstract: Pterion is a region in the anterior part of the floor of the temporal fossa where the greater wing of the sphenoid, the parietal, frontal and the squamous temporal bones meet and form a H shaped suture. Alternatively it is the meeting region of these 4 bones(1,2). It is an commonly used landmark to find the place of anterior division of middle meningeal artery inside. There are four different types of pterions they are the spenoparietal, frontotemporal, stellate and the epipteric varieties(3,2,4). Because the anatomical variation is been so much cared by the forensic anthropologists, neurosurgeons and the forensic pathologists, we find it necessary to study the occurrence of different types of pterion in the skulls of Tamil Nadu regions. So as to full fill the criteria the different types of pterion and it’s occurrence in relation with the middle meningeal artery is been studied. The most occurring type of pterion among tamil nadu skulls are found to be the spenoparietal variety and the frontotemporal the least. -

1 TERMINOLOGIA ANTHROPOLOGICA Names of The
TERMINOLOGIA ANTHROPOLOGICA Names of the parts of the human body, terms of aspects and relationships, and osteological terminology are as in Terminologia Anatomica. GENERAL TERMS EXPLANANTION ADAPTATION Adjustment and change of an organism to a specific environment, due primarily to natural selection. ADAPTIVE RADIATION Divergence of an ancestral population through adaption and speciation into a number of ecological niches. ADULT Fully developed and mature individual ANAGENESIS The progressive adaption of a single evolutionary line, where the population becomes increasingly specialized to a niche that has remained fairly constant through time. ANCESTRY One’s family or ethnic descent, the evolutionary or genetic line of descent of an animal or plant / Ancestral descent or lineage ANTEMORTEM Biological processes that can result in skeletal modifications before death ANTHROPOCENTRICISM The belief that humans are the most important elements in the universe. ANTHROPOLOGY The study of human biology and behavior in the present and in the past ANTHROPOLOGIST BIOLOGICAL A specialist in the subfield of anthropology that studies humans as a biological species FORENSIC A specialist in the use of anatomical structures and physical characteristics to identify a subject for legal purposes PHYSICAL A specialist in the subfield of anthropology dealing with evolutionary changes in the human bodily structure and the classification of modern races 1 SOCIAL A specialist in the subfield of anthropology that deals with cultural and social phenomena such as kingship systems or beliefs ANTHROPOMETRY The study of human body measurement for use in anthropological classification and comparison ARCHETYPE That which is taken as the blueprint for a species or higher taxonomic category ARTIFACT remains of past human activity.