Using the Structural Kinome to Systematize Kinase Drug Discovery
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Ripretinib Demonstrated Activity Across All KIT/PDGFRA Mutations In
Ripretinib demonstrated activity across all KIT/PDGFRA mutations in patients with fourth-line advanced gastrointestinal stromal tumor: Analysis from the phase 3 INVICTUS study Patrick Schöffski1, Sebastian Bauer2, Michael Heinrich3, Suzanne George4, John Zalcberg5, Hans Gelderblom6, Cesar Serrano Garcia7, Robin L Jones8, Steven Attia9, Gina D’Amato10, Ping Chi11, Peter Reichardt12, Julie Meade13, Kelvin Shi13, Ying Su13, Rodrigo Ruiz-Soto13, Margaret von Mehren14, Jean-Yves Blay15 1University Hospitals Leuven, Leuven, Belgium; 2West German Cancer Center, Essen, Germany; 3OHSU Knight Cancer Institute, Portland, OR, USA; 4Dana-Farber Cancer Institute, Boston, MA, USA; 5Monash University, Melbourne, VIC, Australia; 6Leiden University Medical Center, Leiden, Netherlands; 7Vall d’Hebron Institute of Oncology, Barcelona, Spain; 8Royal Marsden and Institute of Cancer Research, London, UK; 9Mayo Clinic, Jacksonville, FL, USA; 10Sylvester Comprehensive Cancer Center, University of Miami, Miami, FL, USA; 11Memorial Sloan Kettering Cancer Center, New York, NY, USA; 12Sarcoma Center, Helios Klinikum Berlin-Buch, Berlin, Germany; 13Deciphera Pharmaceuticals, LLC, Waltham, MA, USA; 14Fox Chase Cancer Center, Philadelphia, PA, USA; 15Centre Leon Berard, Lyon, France KIT mutation analysis by combined tumor and liquid biopsy INTRODUCTION RESULTS Figure 7. Hazard ratio of PFS with different mutation groups by combined • Patients were grouped into 4 subsets: any KIT exon 9, any KIT exon 11, any KIT exon 13, and any KIT exon 17 tumor and liquid biopsy • Patients -

Cstone's Partner Blueprint Medicines Presented Updated Part 1 Data from PIONEER Trial of Avapritinib
Hong Kong Exchanges and Clearing Limited and The Stock Exchange of Hong Kong Limited take no responsibility for the contents of this announcement, make no representation as to its accuracy or completeness and expressly disclaim any liability whatsoever for any loss howsoever arising from or in reliance upon the whole or any part of the contents of this announcement. The forward-looking statements made in this announcement relate only to the events or information as of the date on which the statements are made in this announcement. Except as required by law, we undertake no obligation to update or revise publicly any forward-looking statements, whether as a result of new information, future events or otherwise, after the date on which the statements are made or to reflect the occurrence of unanticipated events. You should read this announcement completely and with the understanding that our actual future results or performance may be materially different from what we expect. In this announcement, statements of, or references to, our intentions or those of any of our directors and/or our Company are made as of the date of this announcement. Any of these intentions may alter in light of future development. CStone Pharmaceuticals 基 石 藥 業 (Incorporated in the Cayman Islands with limited liability) (Stock Code: 2616) VOLUNTARY ANNOUNCEMENT CSTONE’S PARTNER BLUEPRINT MEDICINES PRESENTED UPDATED PART 1 DATA FROM PIONEER TRIAL OF AVAPRITINIB SHOWING ROBUST REDUCTIONS IN CUTANEOUS DISEASE SYMPTOMS IN PATIENTS WITH INDOLENT SYSTEMIC MASTOCYTOSIS The partner of CStone Pharmaceuticals (the “Company” or “CStone”), Blueprint Medicines Corporation (NASDAQ: BPMC) (“Blueprint Medicines”), announced on June 6 updated clinical data from part 1 of the PIONEER trial showing robust and consistent clinical activity for avapritinib across multiple qualitative and quantitative measures of cutaneous disease in patients with indolent systemic mastocytosis (“ISM”). -

DRUGS REQUIRING PRIOR AUTHORIZATION in the MEDICAL BENEFIT Page 1
Effective Date: 08/01/2021 DRUGS REQUIRING PRIOR AUTHORIZATION IN THE MEDICAL BENEFIT Page 1 Therapeutic Category Drug Class Trade Name Generic Name HCPCS Procedure Code HCPCS Procedure Code Description Anti-infectives Antiretrovirals, HIV CABENUVA cabotegravir-rilpivirine C9077 Injection, cabotegravir and rilpivirine, 2mg/3mg Antithrombotic Agents von Willebrand Factor-Directed Antibody CABLIVI caplacizumab-yhdp C9047 Injection, caplacizumab-yhdp, 1 mg Cardiology Antilipemic EVKEEZA evinacumab-dgnb C9079 Injection, evinacumab-dgnb, 5 mg Cardiology Hemostatic Agent BERINERT c1 esterase J0597 Injection, C1 esterase inhibitor (human), Berinert, 10 units Cardiology Hemostatic Agent CINRYZE c1 esterase J0598 Injection, C1 esterase inhibitor (human), Cinryze, 10 units Cardiology Hemostatic Agent FIRAZYR icatibant J1744 Injection, icatibant, 1 mg Cardiology Hemostatic Agent HAEGARDA c1 esterase J0599 Injection, C1 esterase inhibitor (human), (Haegarda), 10 units Cardiology Hemostatic Agent ICATIBANT (generic) icatibant J1744 Injection, icatibant, 1 mg Cardiology Hemostatic Agent KALBITOR ecallantide J1290 Injection, ecallantide, 1 mg Cardiology Hemostatic Agent RUCONEST c1 esterase J0596 Injection, C1 esterase inhibitor (recombinant), Ruconest, 10 units Injection, lanadelumab-flyo, 1 mg (code may be used for Medicare when drug administered under Cardiology Hemostatic Agent TAKHZYRO lanadelumab-flyo J0593 direct supervision of a physician, not for use when drug is self-administered) Cardiology Pulmonary Arterial Hypertension EPOPROSTENOL (generic) -

Clinical Policy: Sorafenib (Nexavar)
Clinical Policy: Sorafenib (Nexavar) Reference Number: CP.PHAR.69 Effective Date: 07.01.11 Last Review Date: 05.21 Line of Business: Commercial, HIM, Medicaid Revision Log See Important Reminder at the end of this policy for important regulatory and legal information. Description Sorafenib (Nexavar®) is a kinase inhibitor. FDA Approved Indication(s) Nexavar (sorafenib) is indicated for the treatment of: Unresectable hepatocellular carcinoma (HCC); Advanced renal cell carcinoma (RCC); Locally recurrent or metastatic, progressive, differentiated thyroid carcinoma (DTC) that is refractory to radioactive iodine treatment. Policy/Criteria Provider must submit documentation (such as office chart notes, lab results or other clinical information) supporting that member has met all approval criteria. It is the policy of health plans affiliated with Centene Corporation® that Nexavar is medically necessary when the following criteria are met: I. Initial Approval Criteria A. Hepatocellular Carcinoma (must meet all): 1. Diagnosis of HCC; 2. Prescribed by or in consultation with an oncologist; 3. Age ≥ 18 years; 4. Confirmation of Child-Pugh class A or B7 status; 5. Request meets one of the following (a or b):* a. Dose does not exceed 800 mg per day; b. Dose is supported by practice guidelines or peer-reviewed literature for the relevant off-label use (prescriber must submit supporting evidence). *Prescribed regimen must be FDA-approved or recommended by NCCN Approval duration: Medicaid/HIM – 6 months Commercial – Length of Benefit B. Renal Cell Carcinoma (must meet all): 1. Diagnosis of advanced RCC; 2. Prescribed by or in consultation with an oncologist; 3. Age ≥ 18 years; 4. Request meets one of the following (a or b):* Page 1 of 8 CLINICAL POLICY Sorafenib a. -

Kinome Profiling of Clinical Cancer Specimens
Published OnlineFirst March 23, 2010; DOI: 10.1158/0008-5472.CAN-09-3989 Review Cancer Research Kinome Profiling of Clinical Cancer Specimens Kaushal Parikh and Maikel P. Peppelenbosch Abstract Over the past years novel technologies have emerged to enable the determination of the transcriptome and proteome of clinical samples. These data sets will prove to be of significant value to our elucidation of the mechanisms that govern pathophysiology and may provide biological markers for future guidance in person- alized medicine. However, an equally important goal is to define those proteins that participate in signaling pathways during the disease manifestation itself or those pathways that are made active during successful clinical treatment of the disease: the main challenge now is the generation of large-scale data sets that will allow us to define kinome profiles with predictive properties on the outcome-of-disease and to obtain insight into tissue-specific analysis of kinase activity. This review describes the current techniques available to gen- erate kinome profiles of clinical tissue samples and discusses the future strategies necessary to achieve new insights into disease mechanisms and treatment targets. Cancer Res; 70(7); 2575–8. ©2010 AACR. Background the current state of the cell or tissue as characterized by pro- teomic and metabolic measurements (3). The past five years have seen an exponential increase in In this review we describe and evaluate the various tech- technological development to explore the genome and the niques that have recently become available to study the ki- proteome to understand the molecular basis of disease. How- nomes of clinical tissue samples. -

Cogent Biosciences Announces Creation of Cogent Research Team
Cogent Biosciences Announces Creation of Cogent Research Team April 6, 2021 Names industry veteran John Robinson, PhD as Chief Scientific Officer New Boulder-based team with exceptional track record of drug discovery and development focused on creating novel small molecule therapies for rare, genetically driven diseases Strong year-end cash position of $242.2 million supports company goals into 2024, including three CGT9486 clinical trials on-track to start this year, beginning with ASM in 1H21 BOULDER, Colo. and CAMBRIDGE, Mass., April 6, 2021 /PRNewswire/ -- Cogent Biosciences, Inc. (Nasdaq: COGT), a biotechnology company focused on developing precision therapies for genetically defined diseases, today announced the formation of the Cogent Research Team led by newly appointed Chief Scientific Officer, John Robinson, PhD. "Today marks an important step forward for Cogent Biosciences as we announce the formation of the Cogent Research Team with a focus on discovering and developing new small molecule therapies for patients fighting rare, genetically driven diseases," said Andrew Robbins, President and Chief Executive Officer of Cogent Biosciences. "I am thrilled to welcome John onboard as Cogent Biosciences' Chief Scientific Officer. John's expertise and seasoned leadership make him ideally suited to lead this new team of world class scientists. Given the team's impressive experience and accomplishments, we are excited for Cogent Biosciences' future and the opportunity to expand our pipeline and deliver novel precision therapies for patients." With an exceptional track record of innovation, the Cogent Research Team will focus on pioneering best-in-class, small molecule therapeutics to both improve upon existing drugs with clear limitations, as well as create new breakthroughs for diseases where others have been unable to find solutions. -

Prior Authorization Oncology – Tukysa™ (Tucatinib Tablets)
Cigna National Formulary Coverage Policy Prior Authorization Oncology – Tukysa™ (tucatinib tablets) Table of Contents Product Identifier(s) National Formulary Medical Necessity ................ 1 64724 Conditions Not Covered....................................... 2 Background .......................................................... 2 References .......................................................... 2 Revision History ................................................... 2 INSTRUCTIONS FOR USE The following Coverage Policy applies to health benefit plans administered by Cigna Companies. Certain Cigna Companies and/or lines of business only provide utilization review services to clients and do not make coverage determinations. References to standard benefit plan language and coverage determinations do not apply to those clients. Coverage Policies are intended to provide guidance in interpreting certain standard benefit plans administered by Cigna Companies. Please note, the terms of a customer’s particular benefit plan document [Group Service Agreement, Evidence of Coverage, Certificate of Coverage, Summary Plan Description (SPD) or similar plan document] may differ significantly from the standard benefit plans upon which these Coverage Policies are based. For example, a customer’s benefit plan document may contain a specific exclusion related to a topic addressed in a Coverage Policy. In the event of a conflict, a customer’s benefit plan document always supersedes the information in the Coverage Policies. In the absence of a controlling federal or state coverage mandate, benefits are ultimately determined by the terms of the applicable benefit plan document. Coverage determinations in each specific instance require consideration of 1) the terms of the applicable benefit plan document in effect on the date of service; 2) any applicable laws/regulations; 3) any relevant collateral source materials including Coverage Policies and; 4) the specific facts of the particular situation. -

NCCN Guidelines for Myeloid/Lymphoid Neoplasms with Eosinophilia and Tyrosine Kinase Fusion Genes V.2.2021 –Interim on 07/21/20
NCCN Guidelines for Myeloid/Lymphoid Neoplasms with Eosinophilia and Tyrosine Kinase Fusion Genes V.2.2021 –Interim on 07/21/20 Guideline Page Institution Vote Panel Discussion/References and Request YES NO ABSTAIN ABSENT MLNE-5 The panel consensus was to include the use of avapritinib in a 18 1 1 6 Internal request: clinical trial with the following footnote: "Avapritinib is approved for adult patients with unresectable or metastatic Consider the addition of avapritinib for gastrointestinal stromal tumors (GISTs) harboring a PDGFRA Myeloid/lymphoid Neoplasms with FIP1L1- exon 18 mutation, including D842V mutations. This suggests a PDGFRA rearrangement in patients with possible role for avapritinib in patients with FIP1L1-PDGFRA– PDGFRA D842V mutation. positive myeloid/lymphoid neoplasms with eosinophilia harboring PDGFRA D842V mutation resistant to imatinib. If this mutation is identified, a clinical trial of avapritinib is preferred (if available), rather than off-label use." MLNE-7 The panel consensus was to include pemigatinib as a TKI with 18 1 1 6 Internal request: activity against FGFR1 under other recommended regimens with the following footnote corresponding to clinical trial and Consider the addition of the pemigatinib for pemigatinib: "Pemigatinib (FGFR1, 2, and 3 inhibitor) is Myeloid/lymphoid Neoplasms with FGFR1 approved for the treatment of adult patients with previously rearrangement. treated, unresectable, locally advanced or metastatic cholangiocarcinoma and a FGFR2 fusion or other rearrangement, as detected by an FDA-approved test. Pemigatinib has received orphan drug designation for the treatment of patients with myeloid/lymphoid neoplasms with eosinophilia and FGFR1 rearrangement and is currently being evaluated in a clinical trial for this indication. -
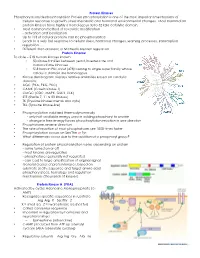
Protein Kinases Phosphorylation/Dephosphorylation Protein Phosphorylation Is One of the Most Important Mechanisms of Cellular Re
Protein Kinases Phosphorylation/dephosphorylation Protein phosphorylation is one of the most important mechanisms of cellular responses to growth, stress metabolic and hormonal environmental changes. Most mammalian protein kinases have highly a homologous 30 to 32 kDa catalytic domain. • Most common method of reversible modification - activation and localization • Up to 1/3 of cellular proteins can be phosphorylated • Leads to a very fast response to cellular stress, hormonal changes, learning processes, transcription regulation .... • Different than allosteric or Michealis Menten regulation Protein Kinome To date – 518 human kinases known • 50 kinase families between yeast, invertebrate and mammaliane kinomes • 518 human PKs, most (478) belong to single super family whose catalytic domain are homologous. • Kinase dendrogram displays relative similarities based on catalytic domains. • AGC (PKA, PKG, PKC) • CAMK (Casein kinase 1) • CMGC (CDC, MAPK, GSK3, CLK) • STE (Sterile 7, 11 & 20 kinases) • TK (Tryosine kinases memb and cyto) • TKL (Tyrosine kinase-like) • Phosphorylation stabilized thermodynamically - only half available energy used in adding phosphoryl to protein - change in free energy forces phosphorylation reaction in one direction • Phosphatases reverse direction • The rate of reaction of most phosphatases are 1000 times faster • Phosphorylation occurs on Ser/The or Tyr • What differences occur due to the addition of a phosphoryl group? • Regulation of protein phosphorylation varies depending on protein - some turned on or off -

Leukemia Insights June 2021
JUNE 2021 In this month’s Leukemia Insights newsletter, written by Prithviraj Bose, M.D. and Srdan Verstovsek, M.D., Ph.D., and sponsored in part by the Charif Souki Cancer Research ABOUT MyMDAnderson Fund, we discuss our novel therapeutic approaches for the rare hematologic malignancies, systemic mastocytosis and myeloid/lymphoid neoplasms with myMDAnderson is a secure, eosinophilia. Learn more about our Leukemia program. personalized web site helping community physicians expedite patient referrals, as well as improve continuity Spotlight on rare, atypical, myeloid of care through information access and streamlined communications. neoplasms: systemic mastocytosis and Physicians who have referred patients myeloid/lymphoid neoplasms with to MD Anderson or plan to do so, can utilize the HIPAA compliant features eosinophilia and FGFR1 rearrangements of myMDAnderson to: • Refer a patient • View your patient's appointments Systemic Mastocytosis Access patient reports • Send and receive secure Systemic mastocytosis (SM) is a rare myeloid neoplasm messages driven in approximately 95% of cases by an activating mutation in c-KIT, usually D816V. SM is characterized as indolent, smoldering or advanced, based on the presence JOIN THE COVERSATION and number of so-called B- and C-findings. The latter Connect with us. signify organ damage and are a hallmark of advanced SM (AdvSM). AdvSM, in turn, is typically sub-classified as aggressive SM (ASM), SM with an associated hematologic neoplasm (SM-AHN, the most common subtype) and mast cell leukemia (MCL). Patients with indolent SM (ISM) and smoldering SM (SSM) enjoy much better survival than JOIN OUR MAILING LIST those with AdvSM, although symptoms in all three subtypes can be severe and debilitating. -

Insights from Drug Discovery Life Cycle Management in Pharmaceutical Industry: a Case-Study B
et International Journal on Emerging Technologies 10 (4): 212-217(2019) ISSN No. (Print): 0975-8364 ISSN No. (Online): 2249-3255 Insights from Drug Discovery Life Cycle Management in Pharmaceutical Industry: A Case-study B. Thomas 1 and P. Chugan 2 1Research Scholar, Institute of Management, Nirma University, Ahmedabad (Gujarat), India. 2Professor (Retd.), Institute of Management, Nirma University, Ahmedabad (Gujarat), India. (Corresponding author: B. Thomas) (Received 20 September 2019, Revised 16 November 2019, Accepted 22 November 2019) (Published by Research Trend, Website: www.researchtrend.net) ABSTRACT: The Biotechnology and Pharmaceutical Industry has always stood up to the challenge of discovering innovative products fulfilling high unmet medical needs for mankind. The drug discovery and development pathway is very challenging and time consuming from efficacy, safety, regulatory and investments perspective. Hence, there is a significant need for collaboration among the biotechnology and large pharmaceutical companies to develop and commercialize their product innovations across the globe. This paper reviews the discovery and development of Selumetinib by Array Biopharma from year 2000 to 2019 when the USFDA accepted the first NDA filing for neurofibromatosis type one (NF1) disease. Array biopharma successfully developed several other drugs to the marketplace and several drugs to late stage registration trials either itself or in collaboration with strategic partners. These strategic partnerships helped mitigate risks in drug development and registration. This study of the clinical, regulatory and partnership strategy untaken for Selumetinib has created important contribution in the field of product life cycle management in the pharmaceutical and biotechnology industry. Keywords: Product development, drug development, life-cycle management, strategic partnership. -

New Century Health Policy Changes April 2021
Policy # Drug(s) Type of Change Brief Description of Policy Change new Pepaxto (melphalan flufenamide) n/a n/a new Fotivda (tivozanib) n/a n/a new Cosela (trilaciclib) n/a n/a Add inclusion criteria: NSCLC UM ONC_1089 Libtayo (cemiplimab‐rwlc) Negative change 2.Libtayo (cemiplimab) may be used as montherapy in members with locally advanced, recurrent/metastatic NSCLC, with PD‐L1 ≥ 50%, negative for actionable molecular markers (ALK, EGFR, or ROS‐1) Add inclusion criteria: a.As a part of primary/de�ni�ve/cura�ve‐intent concurrent chemo radia�on (Erbitux + Radia�on) as a single agent for members with a UM ONC_1133 Erbitux (Cetuximab) Positive change contraindication and/or intolerance to cisplatin use OR B.Head and Neck Cancers ‐ For recurrent/metasta�c disease as a single agent, or in combination with chemotherapy. Add inclusion criteria: UM ONC_1133 Erbitux (Cetuximab) Negative change NOTE: Erbitux (cetuximab) + Braftovi (encorafenib) is NCH preferred L1 pathway for second‐line or subsequent therapy in the metastatic setting, for BRAFV600E positive colorectal cancer.. Add inclusion criteria: B.HER‐2 Posi�ve Breast Cancer i.Note #1: For adjuvant (post‐opera�ve) use in members who did not receive neoadjuvant therapy/received neoadjuvant therapy and did not have any residual disease in the breast and/or axillary lymph nodes, Perjeta (pertuzumab) use is restricted to node positive stage II and III disease only. ii.Note #2: Perjeta (pertuzumab) use in the neoadjuvant (pre‐opera�ve) se�ng requires radiographic (e.g., breast MRI, CT) and/or pathologic confirmation of ipsilateral (same side) axillary nodal involvement.