Development of a LC-MS/MS Method for the Estimation of Clinofibrate In
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Ezetimibe: a Novel Selective Cholesterol Absorption Inhibitor by Michele Koder, Pharm.D
OREGON DUR BOARD NEWSLETTER A N E VIDENCE B ASED D RUG T HERAPY R ESOURCE COPYRIGHT 2003 OREGON STATE UNIVERSITY. ALL RIGHTS RESERVED Volume 5, Issue 2 Also available on the web and via e-mail list-serve at February 2003 http://pharmacy.orst.edu/drug_policy/newsletter_email.html Ezetimibe: A novel selective cholesterol absorption inhibitor By Michele Koder, Pharm.D. , OSU College of Pharmacy Ezetimibe (Zetia) is a novel selective cholesterol absorption inhibitor that was approved by the FDA in October 2002. Unlike statins (HMG-CoA reductase inhibitors) and bile acid sequestrants, ezetimibe does not inhibit hepatic cholesterol synthesis or increase bile acid secretion. In contrast, ezetimibe selectively inhibits the uptake of dietary cholesterol from enterocytes in the brush border of the intestinal lumen resulting in a decrease in the delivery of dietary cholesterol to the liver and a subsequent decrease in hepatic cholesterol stores and increased cholesterol clearance from the blood.1 Ezetimibe’s unique action has generated interest in its use in combination with other cholesterol-lowering agents. It is indicated for the treatment of primary hypercholesterolemia as monotherapy and in combination with a statin. Ezetimibe is also approved for homozygous familial hypercholesterolemia and homozygous sitosterolemia. TABLE 1: EZETIMIBE CLINICAL TRIAL SUMMARY Study / Design Population Treatment % Change LDL % Change HDL % Change TG Bays et al3 N=432 EZ 5 mg -15.7 +2.9 MC, R, DB, PC LDL 130-250mg/dl EZ 10 mg -18.5 +3.5 NS 12 wk; Phase II TG -
Statins and Lipid Lowering Agents
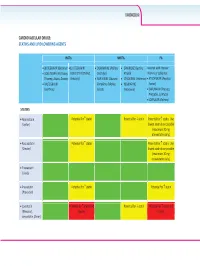
CARDIOVASCULAR Z/Ks^h>ZZh'^͗ ^dd/E^E>/W/>KtZ/E''Ed^ /E^d/Ɛ EEZd/Ɛ W/Ɛ x/d'Zs/Z ;ŝŬƚĂƌǀLJͿ x>s/d'Zs/Zͬ x KZs/Z/E ;WŝĨĞůƚƌŽ͕ x &s/ZE ;^ƵƐƚŝǀĂ͕ ŽŽƐƚĞĚǁŝƚŚƌŝƚŽŶĂǀŝƌ xK>hd'Zs/Z ;dŝǀŝĐĂLJ͕ K//^dd ;^ƚƌŝďŝůĚ͕ ĞůƐƚƌŝŐŽͿ ƚƌŝƉůĂͿ ;EŽƌǀŝƌͿŽƌĐŽďŝĐŝƐƚĂƚ dƌŝƵŵĞƋ͕:ƵůƵĐĂ͕ŽǀĂƚŽͿ 'ĞŶǀŽLJĂͿ x Z/>W/s/Z/E ;ĚƵƌĂŶƚ͕ x dZs/Z/E ;/ŶƚĞůĞŶĐĞͿ xdEs/Z ;ZĞLJĂƚĂnj͕ xZ>d'Zs/Z ŽŵƉůĞƌĂ͕KĚĞĨƐĞLJ͕ x Es/ZW/E ǀŽƚĂnjͿ ;/ƐĞŶƚƌĞƐƐͿ :ƵůƵĐĂͿ ;sŝƌĂŵƵŶĞͿ xZhEs/Z ;WƌĞnjŝƐƚĂ͕ WƌĞnjĐŽďŝdž͕^LJŵƚƵnjĂͿ x>KW/Es/Z ;<ĂůĞƚƌĂͿ ^dd/E^ xƚŽƌǀĂƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ nƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ pƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ nƐƚĂƚŝŶ͘hƐĞ ;>ŝƉŝƚŽƌͿ ůŽǁĞƐƚƐƚĂƚŝŶĚŽƐĞƉŽƐƐŝďůĞ ;ŵĂdžŝŵƵŵϮϬŵŐ ĂƚŽƌǀĂƐƚĂƚŝŶĚĂŝůLJͿ͘ xZŽƐƵǀĂƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ nƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ nƐƚĂƚŝŶ͘hƐĞ ;ƌĞƐƚŽƌͿ ůŽǁĞƐƚƐƚĂƚŝŶĚŽƐĞƉŽƐƐŝďůĞ ;ŵĂdžŝŵƵŵϭϬŵŐ ƌŽƐƵǀĂƐƚĂƚŝŶĚĂŝůLJͿ͘ xWŝƚĂǀĂƐƚĂƚŝŶ ;>ŝǀĂůŽͿ xWƌĂǀĂƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ nƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ nƐƚĂƚŝŶ ;WƌĂǀĂĐŚŽůͿ x>ŽǀĂƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ nƐƚĂƚŝŶĂŶĚ WŽƚĞŶƚŝĂůĨŽƌ pƐƚĂƚŝŶ WŽƚĞŶƚŝĂůĨŽƌ nƐƚĂƚŝŶĂŶĚ ;DĞǀĂĐŽƌͿ͕ ƚŽdžŝĐŝƚLJ ƚŽdžŝĐŝƚLJ ƐŝŵǀĂƐƚĂƚŝŶ ;ŽĐŽƌͿ CARDIOVASCULAR INSTIs NNRTIs PIs xBICTEGRAVIR (Biktarvy) xELVITEGRAVIR/ x DORAVIRINE (Pifeltro, x EFAVIRENZ (Sustiva, Boosted with ritonavir xDOLUTEGRAVIR (Tivicay, COBICISTAT (Stribild, Delstrigo) Atripla) (Norvir) or cobicistat Triumeq, Juluca) Genvoya) x RILPIVIRINE (Edurant, x ETRAVIRINE (Intelence) xATAZANAVIR (Reyataz, xRALTEGRAVIR Complera, Odefsey, x NEVIRAPINE Evotaz) (Isentress) Juluca) (Viramune) xDARUNAVIR (Prezista, Prezcobix, Symtuza) xLOPINAVIR (Kaletra) FIBRATES xFenofibrate, bezafibrate, gemfibrozil CHOLESTEROL ABSORPTION INHIBITOR xEzetimibe -

FIELD Study Revealed Fenofibrate Reduced Need for Laser Treatment for Diabetic Retinopathy by Anthony C
Supplement to Supported by an unrestricted educational grant from Abbott Laboratories March/April 2008 FIELD Study Revealed Fenofibrate Reduced Need for Laser Treatment for Diabetic Retinopathy By Anthony C. Keech, MBBS, Msc Epid, FRANZCS, FRACP; and Paul Mitchell, MBBS(Hons), MD, PhD, FRANZCO, FRACS, FRCOphth, FAFPHM This agent’s mechanism of benefit in diabetic retinopathy appears to go beyond its effects on lipid concentration or blood pressure, and this potential mechanism of action operates even when glycemic control and blood pressure levels are within goal. ABSTRACT icant relative reduction was seen of almost one-third in PURPOSE the rate of first laser application for retinopathy after The FIELD (Fenofibrate Intervention and Event an average treatment duration of 5 years with fenofi- Lowering in Diabetes) study sought to investigate brate 200 mg/day. whether long-term lipid-lowering therapy with fenofi- In this report, we detail the effects of fenofibrate brate would reduce macro- and microvascular compli- administration on ophthalmic microvascular compli- cations among patients with type 2 diabetes. We previ- cations and attempt to clarify some of the underlying ously reported that in type 2 diabetes patients with pathologies being addressed among patients undergo- adequate glycemic and blood pressure control, a signif- ing laser treatment. Jointly sponsored by The Dulaney Foundation and Retina Today MARCH/APRIL 2008 I SUPPLEMENT TO RETINA TODAY I 1 FIELD Study Revealed Fenofibrate Reduced Need for Laser Treatment for Diabetic Retinopathy Jointly sponsored by The Dulaney Foundation and Retina Today. Release date: April 2008. Expiration date: April 2009. This continuing medical education activity is supported by an unrestricted educational grant from Abbott Laboratories. -

Clinofibrate Improved Canine Lipid Metabolism in Some but Not All Breeds
NOTE Internal Medicine Clinofibrate improved canine lipid metabolism in some but not all breeds Yohtaro SATO1), Nobuaki ARAI2), Hidemi YASUDA3) and Yasushi MIZOGUCHI4)* 1)Graduate School of Agriculture, Meiji University, 1-1-1 Higashimita, Tama-ku, Kawasaki, Kanagawa 214-8571, Japan 2)Spectrum Lab Japan, 1-5-22-201 Midorigaoka, Meguro-ku, Tokyo 152-0034, Japan 3)Yasuda Veterinary Clinic, 1-5-22 Midorigaoka, Meguro-ku, Tokyo 152-0034, Japan 4)School of Agriculture, Meiji University, 1-1-1 Higashimita, Tama-ku, Kawasaki, Kanagawa 214-8571, Japan ABSTRACT. The objectives of this study were to assess if Clinofibrate (CF) treatment improved J. Vet. Med. Sci. lipid metabolism in dogs, and to clarify whether its efficacy is influenced by canine characteristics. 80(6): 945–949, 2018 We collected medical records of 306 dogs and performed epidemiological analyses. Lipid values of all lipoproteins were significantly decreased by CF medication, especially VLDL triglyceride doi: 10.1292/jvms.17-0703 (TG) concentration (mean reduction rate=54.82%). However, 17.65% of dogs showed drug refractoriness in relation to TG level, and Toy Poodles had a lower CF response than other breeds (OR=5.36, 95% CI=2.07–13.90). Therefore, our study suggests that genetic factors may have an Received: 22 December 2017 effect on CF response, so genetic studies on lipid metabolism-related genes might be conducted Accepted: 9 March 2018 to identify variations in CF efficacy. Published online in J-STAGE: KEY WORDS: clinofibrate, descriptive epidemiology, drug response, dyslipidemia, Toy Poodle 26 March 2018 High serum cholesterol (Cho) and triglyceride (TG) concentrations in dogs are caused by various factors such as lack of exercise, high fat diets, obesity, neutralization, age, diseases and breed [6, 21, 24]. -

Effects of Clofibrate Derivatives on Hyperlipidemia Induced by a Cholesterol-Free, High-Fructose Diet in Rats
Showa Univ. J. Med. Sci. 7(2), 173•`182, December 1995 Original Effects of Clofibrate Derivatives on Hyperlipidemia Induced by a Cholesterol-Free, High-Fructose Diet in Rats Hideyukl KURISHIMA,Sadao NAKAYAMA,Minoru FURUYA and Katsuji OGUCHI Abstract: The effects of the clofibrate derivatives fenofibrate (FF), bezafibrate (BF), and clinofibrate (CF), on hyperlipidemia induced by a cholesterol-free, high-fructose diet (HFD) in rats were investigated. Feeding of HFD for 2 weeks increased the high-density lipoprotein subfraction (HDL1) and decreased the low-density lipoprotein (LDL) fraction. The levels of total cholesterol (TC), free cholesterol, triglyceride (TG), and phospholipid in serum were increased by HFD feeding. Administration of CF inhibited the increase in HDL1 content. All three agents inhibited the decrease in LDL level. Both BF and CF decreased VLDL level. Administration of FF, BF, or CF inhibited the increases of serum lipids, especially that of TC and TG. The inhibitory effects of CF on HFD- induced increases in HDL1, TC, and TG were greater than those of FF and BF. These results demonstrate that FF, BF, and CF improve the intrinsic hyper- lipidemia induced by HFD feeding in rats. Key words: fenofibrate, bezafibrate, clinofibrate, fructose-induced hyperlipide- mia, lipoprotein. Introduction Clofibrate is one of the most effective antihypertriglycedemic agents currently available. However, because of its adverse effects, such as hepatomegaly1, several derivatives, such as clinofibrate (CF) and bezafibrate (BF) have been developed which are more effective and have fewer adverse effects. For example, it has been shown that the hypolipidemic effect of CF is greater than that of clofibrate while its tendency to produce hepatomegaly is less1. -
Bezalip, 200 Mg Film Coated Tablets Bezalip Retard, 400 Mg Sustained Release, Film Coated Tablets
NEW ZEALAND DATA SHEET 1. PRODUCT NAME Bezalip, 200 mg film coated tablets Bezalip Retard, 400 mg sustained release, film coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film coated tablet contains 200 mg of bezafibrate. Each sustained release, film coated tablets contains 400 mg of bezafibrate. Excipient with known effect: lactose monohydrate (Bezalip Retard) For the full list of excipients, see section 6.1. 3. PHARMACEUTICAL FORM Bezalip is a white, round film-coated tablet imprinted with ‘G6’ at reverse. Bezalip Retard is a white, round film-coated tablet engraved D9 on one face. 4. CLINICAL PARTICULARS 4.1 Therapeutic indications Bezalip and Bezalip is indicated for: • primary hyperlipidaemia types IIa, IIb, III, IV and V (Fredrickson classification) corresponding to groups I, II and III of the European Atherosclerosis Society guidelines – when diet alone or improvements in lifestyle such as increased exercise or weight reduction do not lead to an adequate response. • secondary hyperlipidaemias, e.g. severe hypertriglyceridemias, when sufficient improvement does not occur after correction of the underlying disorder (e.g. diabetes mellitus). 4.2 Dose and method of administration Adults The standard dosage for Bezalip 200 mg tablets is 1 tablet (200 mg) 3 times daily. In cases of good therapeutic response, especially in hypertriglyceridaemia, the dosage can be reduced to 1 tablet twice daily. For patients with a history of gastric sensitivity, the dosage may be gradually increased to the maintenance level. The standard dosage for Bezalip Retard 400 mg tablets is 1 tablet once daily. Special populations Patient with renal Impairment The dosage in patients with impaired renal function must be adjusted according to serum creatinine levels or creatinine clearance. -

(CPG) for the Management of Dyslipidemia for Cardiovascular
VA/DoD CLINICAL PRACTICE GUIDELINE FOR THE MANAGEMENT OF DYSLIPIDEMIA FOR CARDIOVASCULAR RISK REDUCTION Department of Veterans Affairs Department of Defense QUALIFYING STATEMENTS The Department of Veterans Affairs and the Department of Defense guidelines are based upon the best information available at the time of publication. They are designed to provide information and assist decision making. They are not intended to define a standard of care and should not be construed as one. Neither should they be interpreted as prescribing an exclusive course of management. This Clinical Practice Guideline is based on a systematic review of both clinical trial and epidemiological evidence. Developed by a panel of multidisciplinary experts, it provides a clear explanation of the logical relationships between various care options and health outcomes while rating both the quality of the evidence and the strength of the recommendation. Variations in practice will inevitably and appropriately occur when clinicians take into account the needs of individual patients, available resources, and limitations unique to an institution or type of practice. Every healthcare professional making use of these guidelines is responsible for evaluating the appropriateness of applying them in the setting of any particular clinical situation. These guidelines are not intended to represent Department of Veterans Affairs nor TRICARE policy. Further, inclusion of recommendations for specific testing and/or therapeutic interventions within these guidelines does not guarantee coverage. Additional information on current TRICARE benefits may be found at www.tricare.mil or by contacting your regional TRICARE Managed Care Support Contractor. Version 4.0 – 2020 VA/DoD Clinical Practice Guideline for the Management of Dyslipidemia for Cardiovascular Risk Reduction Prepared by: The Management of Dyslipidemia for Cardiovascular Risk Reduction Work Group With support from: The Office of Quality and Patient Safety, VA, Washington, DC & Office of Evidence Based Practice, U.S. -

Bempedoic Acid and Inclisiran for Patients with Heterozygous Familial Hypercholesterolemia and for Secondary Prevention of ASCVD: Effectiveness and Value
Bempedoic Acid and Inclisiran for Patients with Heterozygous Familial Hypercholesterolemia and for Secondary Prevention of ASCVD: Effectiveness and Value Draft Evidence Report November 12, 2020 Prepared for ©Institute for Clinical and Economic Review, 2020 ICER Staff and Consultants Modeling Team Grace A. Lin, MD, MAS Dhruv S. Kazi, MD, MSc, MS Associate Professor of Medicine and Health Policy Associate Director, Smith Center for Outcomes University of California, San Francisco Research in Cardiology Director, Cardiac Critical Care Unit Jane Jih, MD, MPH Beth Israel Deaconess Medical Center Assistant Professor, Division of General Internal Associate Professor, Harvard Medical School Medicine University of California, San Francisco Foluso Agboola, MBBS, MPH Director, Evidence Synthesis Dr. Kazi was responsible for the development of the ICER cost-effectiveness model, interpretation of results, and drafting of the economic sections of this report; the Rick Chapman, PhD, MS resulting ICER reports do not necessarily represent the Director of Health Economics views of Beth Israel Deaconess Medical Center or ICER Harvard Medical School. Steven D. Pearson, MD, MSc President ICER DATE OF PUBLICATION: November 12, 2020 How to cite this document: Lin GA, Kazi DS, Jih J, Agboola F, Chapman R, Pearson SD. Inclisiran and Bempedoic Acid for Patients with Heterozygous Familial Hypercholesterolemia and for Secondary Prevention of ASCVD: Effectiveness and Value; Draft Evidence Report. Institute for Clinical and Economic Review, November 12, 2020. https://icer-review.org/material/high-cholesterol-update- draft-evidence-report/ Grace Lin served as the lead author for the report and wrote the background, other benefits, and contextual considerations sections of the report, with Jane Jih serving as a co-author. -

(12) United States Patent (10) Patent No.: US 7,795,310 B2 Lee Et Al
US00779531 OB2 (12) United States Patent (10) Patent No.: US 7,795,310 B2 Lee et al. (45) Date of Patent: Sep. 14, 2010 (54) METHODS AND REAGENTS FOR THE WO WO 2005/025673 3, 2005 TREATMENT OF METABOLIC DISORDERS OTHER PUBLICATIONS (75) Inventors: Margaret S. Lee, Middleton, MA (US); Tenenbaum et al., “Peroxisome Proliferator-Activated Receptor Grant R. Zimmermann, Somerville, Ligand Bezafibrate for Prevention of Type 2 Diabetes Mellitus in MA (US); Alyce L. Finelli, Patients With Coronary Artery Disease'. Circulation, 2004, pp. 2197 Framingham, MA (US); Daniel Grau, 22O2.* Shen et al., “Effect of gemfibrozil treatment in sulfonylurea-treated Cambridge, MA (US); Curtis Keith, patients with noninsulin-dependent diabetes mellitus'. The Journal Boston, MA (US); M. James Nichols, of Clinical Endocrinology & Metabolism, vol. 73, pp. 503-510, Boston, MA (US) 1991 (see enclosed abstract).* International Search Report from PCT/US2005/023030, mailed Dec. (73) Assignee: CombinatoRx, Inc., Cambridge, MA 1, 2005. (US) Lin et al., “Effect of Experimental Diabetes on Elimination Kinetics of Diflunisal in Rats.” Drug Metab. Dispos. 17:147-152 (1989). (*) Notice: Subject to any disclaimer, the term of this Abstract only. patent is extended or adjusted under 35 Neogi et al., “Synthesis and Structure-Activity Relationship Studies U.S.C. 154(b) by 0 days. of Cinnamic Acid-Based Novel Thiazolidinedione Antihyperglycemic Agents.” Bioorg. Med. Chem. 11:4059-4067 (21) Appl. No.: 11/171,566 (2003). Vessby et al., “Effects of Bezafibrate on the Serum Lipoprotein Lipid and Apollipoprotein Composition, Lipoprotein Triglyceride Removal (22) Filed: Jun. 30, 2005 Capacity and the Fatty Acid Composition of the Plasma Lipid Esters.” Atherosclerosis 37:257-269 (1980). -

Low Doses of Fenofibrate and Bezafibrate Stimulate Renal 2-Oxoglutarate Dehydrogenase (2-Ogdh) in Protein-Restricted Rats
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Jagiellonian Univeristy Repository Acta Poloniae Pharmaceutica ñ Drug Research, Vol. 77 No. 2 pp. 281ñ288, 2020 ISSN 2353-5288 DOI: 10.32383/appdr/118535 Polish Pharmaceutical Society Received 12 December 2019, Received in revised form 21 February 2020, Accepted 28 February 2020. DRUG BIOCHEMISTRY LOW DOSES OF FENOFIBRATE AND BEZAFIBRATE STIMULATE RENAL 2-OXOGLUTARATE DEHYDROGENASE (2-OGDH) IN PROTEIN-RESTRICTED RATS MALGORZATA E. KNAPIK-CZAJKA*, JAGODA M. DRAG and ANNA GAWEDZKA Department of Biochemical Analytics, Faculty of Pharmacy, Jagiellonian University Medical College, 9 Medyczna St., 30-688 KrakÛw, Poland Abstract: 2-oxoglutarate dehydrogenase (2-OGDH) is the key regulatory enzyme of cell metabolism. It has been previously demonstrated that in rats subjected to protein restriction low, clinically relevant doses of fibrates up-regulate liver 2-OGDH and promote 2-oxoglutarate catabolism. The aim of the present study was to evaluate the effect of low doses of fenofibrate and bezafibrate on renal 2-OGDH complex in rats fed low-pro- tein chow. Fibrates were administered for 14 days to Wistar male rats at one daily dose of 5, 10 or 20 mg/kg b.wt./day. The 2-OGDH activity was assayed spectrophotometrically. The mRNA levels for 2-OGDH catalyt- ic subunits (E1 and E2) and PPARα were quantified by means of semi-quantitative reverse-transcription-PCR. 2-OGDH activity increased in response to administration of fenofibrate and bezafibrate (by 11, 24, 32% and 9, 12, 21%, respectively). The difference was statistically significant for the doses of 10 and 20 mg/kg b.wt of fenofibrate (p < 0.001) and the highest dose of bezafibrate (p < 0.05). -

Product Monograph
PRODUCT MONOGRAPH Pr AA-FENO-MICRO Fenofibrate Capsules 67 mg and 200 mg fenofibrate, micronized formulation House Standard Pr FENOFIBRATE Fenofibrate Capsules 100 mg fenofibrate, non-micronized formulation House Standard Lipid Metabolism Regulator AA PHARMA INC. Date of Preparation: 1165 Creditstone Road Unit #1 October 08, 2019 Vaughan, Ontario L4K 4N7 Control No.: 230394 PRODUCT MONOGRAPH Pr AA-FENO-MICRO Fenofibrate Capsules 67 mg and 200 mg fenofibrate, micronized formulation House Standard Pr FENOFIBRATE Fenofibrate Capsules 100 mg fenofibrate, non-micronized formulation House Standard THERAPEUTIC CLASSIFICATION Lipid Metabolism Regulator ACTIONS AND CLINICAL PHARMACOLOGY Fenofibrate lowers elevated serum lipids by decreasing the low-density lipoprotein (LDL) fraction rich in cholesterol and the very low density lipoprotein (VLDL) fraction rich in triglycerides. In addition, fenofibrate increases the high density lipoprotein (HDL) cholesterol fraction. Fenofibrate appears to have a greater depressant effect on the VLDL than on the low density lipoproteins (LDL). Therapeutic doses of fenofibrate produce elevations of HDL cholesterol, a reduction in the content of the low density lipoproteins cholesterol, and a substantial reduction in the triglyceride content of VLDL. The mechanism of action of fenofibrate has not been definitively established. Work carried out to date suggests that fenofibrate: · enhances the liver elimination of cholesterol as bile salts; · inhibits the biosynthesis of triglycerides and enhances the catabolism of VLDL by increasing the activity of lipoprotein lipase; · has an inhibitory effect on the biosynthesis of cholesterol by modulating the activity of HMG- CoA reductase. Metabolism and Excretion After oral administration with food, fenofibrate is rapidly hydrolysed to fenofibric acid, the active metabolite. -

Fenofibrate Capsules Apotex Standard 67 Mg and 200 Mg
PRODUCT MONOGRAPH PrAPO-FENO-MICRO Fenofibrate Capsules Apotex Standard 67 mg and 200 mg PrAPO-FENOFIBRATE Fenofibrate Capsules Apotex Standard 100 mg Lipid Metabolism Regulator APOTEX INC. 150 Signet Drive Toronto, Ontario DATE OF REVISION: M9L 1T9 October 7, 2014 Control No.: 169773 - 1 - PRODUCT MONOGRAPH PrAPO-FENO-MICRO Fenofibrate Capsules Apotex Standard 67 mg and 200 mg PrAPO-FENOFIBRATE Fenofibrate Capsules Apotex Standard 100 mg THERAPEUTIC CLASSIFICATION Lipid Metabolism Regulator ACTIONS AND CLINICAL PHARMACOLOGY Fenofibrate lowers elevated serum lipids by decreasing the low-density lipoprotein (LDL) fraction rich in cholesterol and the very low density lipoprotein (VLDL) fraction rich in triglycerides. In addition, fenofibrate increases the high density lipoprotein (HDL) cholesterol fraction. Fenofibrate appears to have a greater depressant effect on the VLDL than on the low density lipoproteins (LDL). Therapeutic doses of fenofibrate produce elevations of HDL cholesterol, a reduction in the content of the low density lipoproteins cholesterol, and a substantial reduction in the triglyceride content of VLDL. The mechanism of action of fenofibrate has not been definitively established. Work carried out to date suggests that fenofibrate: · enhances the liver elimination of cholesterol as bile salts; · inhibits the biosynthesis of triglycerides and enhances the catabolism of VLDL by increasing the activity of lipoprotein lipase; · has an inhibitory effect on the biosynthesis of cholesterol by modulating the activity of HMG- CoA reductase. Metabolism and Excretion After oral administration with food, fenofibrate is rapidly hydrolyzed to fenofibric acid, the active metabolite. In man it is mainly excreted through the kidney. Half-life is about 20 hours. In patients with severe renal failure, significant accumulation was observed with a large increase in half-life.