The Structural Basis of Myomesin Stretching
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

A Case of Prenatal Diagnosis of 18P Deletion Syndrome Following
Zhao et al. Molecular Cytogenetics (2019) 12:53 https://doi.org/10.1186/s13039-019-0464-y CASE REPORT Open Access A case of prenatal diagnosis of 18p deletion syndrome following noninvasive prenatal testing Ganye Zhao, Peng Dai, Shanshan Gao, Xuechao Zhao, Conghui Wang, Lina Liu and Xiangdong Kong* Abstract Background: Chromosome 18p deletion syndrome is a disease caused by the complete or partial deletion of the short arm of chromosome 18, there were few cases reported about the prenatal diagnosis of 18p deletion syndrome. Noninvasive prenatal testing (NIPT) is widely used in the screening of common fetal chromosome aneuploidy. However, the segmental deletions and duplications should also be concerned. Except that some cases had increased nuchal translucency or holoprosencephaly, most of the fetal phenotype of 18p deletion syndrome may not be evident during the pregnancy, 18p deletion syndrome was always accidentally discovered during the prenatal examination. Case presentations: In our case, we found a pure partial monosomy 18p deletion during the confirmation of the result of NIPT by copy number variation sequencing (CNV-Seq). The result of NIPT suggested that there was a partial or complete deletion of X chromosome. The amniotic fluid karyotype was normal, but result of CNV-Seq indicated a 7.56 Mb deletion on the short arm of chromosome 18 but not in the couple, which means the deletion was de novo deletion. Finally, the parents chose to terminate the pregnancy. Conclusions: To our knowledge, this is the first case of prenatal diagnosis of 18p deletion syndrome following NIPT.NIPT combined with ultrasound may be a relatively efficient method to screen chromosome microdeletions especially for the 18p deletion syndrome. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

Pathogenic Variant Identified
PERFORMED AT PHOSPHORUS DIAGNOSTICS, 400 PLAZA DRIVE, SUITE 401, SECAUCUS, NJ 07094 ORDERING PRACTICE JANE DOE TEST INFORMATION Practice Code: 100 DOB: 1973-02-19 Panel: Pan Arrhythmia and Cardiomyopathy Panel Sample Cardiology Clinic Gender: Female Indication: Diagnostic for Patient History 374 Broadway Ethnicity: European New York, NY 10001 Procedure ID: 87000 Physician: Dr. Sample Kit Barcode: 201612092248585 Specimen: Blood, #10000 Specimen Collected: 2016-01-12 Specimen Received: 2016-01-13 Specimen Analyzed: 2016-01-21 Report Generated: 2016-02-03 SUMMARY OF RESULTS Positive: Pathogenic Variant Identified GENE LOCATION VARIANT INHERITANCE ZYGOSITY CLASSIFICATION KCNQ1 Exon 6; Chrom 11 c.914G>A Autosomal Dominant Heterozygous PATHOGENIC (p.Trp205Ter) MYH7 Exon 38; Chrom 14 c.26647G>A Unknown Heterozygous UNCERTAIN (p.Glu1883Lys) SIGNIFICANCE INTERPRETATION This patient is heterozygous for the c.914G>A (p.Trp205Ter) pathogenic variant in KCNQ1. This result indicates that this patient may be affected with, or predisposed to developing, long QT syndrome. This patient is also heterozygous for the c.185G>A (p.Lys65Ala) variant of unknown clinical significance in MYH7. The contribution of this variant to the patient’s phenotype is unknown. RECOMMENDATIONS Clinical follow-up with a cardiologist or cardiogeneticist is recommended to discuss medical management. Genetic counseling is recommended for this patient. Genetic testing may be useful in identifying family members at risk for disease. Genetic counseling is recommended for all individuals undergoing genetic testing. VARIANT INFORMATION PATHOGENIC KCNQ1 c.914G>A (p.Trp305Ter) This variant leads to a premature termination at codon 305, which is expected to result in an absent or significantly disrupted protein product. -

Human Induced Pluripotent Stem Cell–Derived Podocytes Mature Into Vascularized Glomeruli Upon Experimental Transplantation
BASIC RESEARCH www.jasn.org Human Induced Pluripotent Stem Cell–Derived Podocytes Mature into Vascularized Glomeruli upon Experimental Transplantation † Sazia Sharmin,* Atsuhiro Taguchi,* Yusuke Kaku,* Yasuhiro Yoshimura,* Tomoko Ohmori,* ‡ † ‡ Tetsushi Sakuma, Masashi Mukoyama, Takashi Yamamoto, Hidetake Kurihara,§ and | Ryuichi Nishinakamura* *Department of Kidney Development, Institute of Molecular Embryology and Genetics, and †Department of Nephrology, Faculty of Life Sciences, Kumamoto University, Kumamoto, Japan; ‡Department of Mathematical and Life Sciences, Graduate School of Science, Hiroshima University, Hiroshima, Japan; §Division of Anatomy, Juntendo University School of Medicine, Tokyo, Japan; and |Japan Science and Technology Agency, CREST, Kumamoto, Japan ABSTRACT Glomerular podocytes express proteins, such as nephrin, that constitute the slit diaphragm, thereby contributing to the filtration process in the kidney. Glomerular development has been analyzed mainly in mice, whereas analysis of human kidney development has been minimal because of limited access to embryonic kidneys. We previously reported the induction of three-dimensional primordial glomeruli from human induced pluripotent stem (iPS) cells. Here, using transcription activator–like effector nuclease-mediated homologous recombination, we generated human iPS cell lines that express green fluorescent protein (GFP) in the NPHS1 locus, which encodes nephrin, and we show that GFP expression facilitated accurate visualization of nephrin-positive podocyte formation in -

Murine Obscurin and Obsl1 Have Functionally Redundant Roles in Sarcolemmal Integrity, Sarcoplasmic Reticulum Organization, and Muscle Metabolism
UC San Diego UC San Diego Previously Published Works Title Murine obscurin and Obsl1 have functionally redundant roles in sarcolemmal integrity, sarcoplasmic reticulum organization, and muscle metabolism. Permalink https://escholarship.org/uc/item/46t7g5hw Journal Communications biology, 2(1) ISSN 2399-3642 Authors Blondelle, Jordan Marrocco, Valeria Clark, Madison et al. Publication Date 2019 DOI 10.1038/s42003-019-0405-7 Peer reviewed eScholarship.org Powered by the California Digital Library University of California ARTICLE https://doi.org/10.1038/s42003-019-0405-7 OPEN Murine obscurin and Obsl1 have functionally redundant roles in sarcolemmal integrity, sarcoplasmic reticulum organization, and muscle metabolism 1234567890():,; Jordan Blondelle1,7, Valeria Marrocco1,7, Madison Clark1, Patrick Desmond1, Stephanie Myers1, Jim Nguyen1, Matthew Wright1, Shannon Bremner2, Enrico Pierantozzi3, Samuel Ward2, Eric Estève1,4, Vincenzo Sorrentino 3, Majid Ghassemian5 & Stephan Lange 1,6 Biological roles of obscurin and its close homolog Obsl1 (obscurin-like 1) have been enig- matic. While obscurin is highly expressed in striated muscles, Obsl1 is found ubiquitously. Accordingly, obscurin mutations have been linked to myopathies, whereas mutations in Obsl1 result in 3M-growth syndrome. To further study unique and redundant functions of these closely related proteins, we generated and characterized Obsl1 knockouts. Global Obsl1 knockouts are embryonically lethal. In contrast, skeletal muscle-specific Obsl1 knockouts show a benign phenotype similar to obscurin knockouts. Only deletion of both proteins and removal of their functional redundancy revealed their roles for sarcolemmal stability and sarcoplasmic reticulum organization. To gain unbiased insights into changes to the muscle proteome, we analyzed tibialis anterior and soleus muscles by mass spectrometry, unco- vering additional changes to the muscle metabolism. -

Cardiovascular Diseases Genetic Testing Program Information
Cardiovascular Diseases Genetic Testing Program Description: Congenital Heart Disease Panels We offer comprehensive gene panels designed to • Congenital Heart Disease Panel (187 genes) diagnose the most common genetic causes of hereditary • Heterotaxy Panel (114 genes) cardiovascular diseases. Testing is available for congenital • RASopathy/Noonan Spectrum Disorders Panel heart malformation, cardiomyopathy, arrythmia, thoracic (31 genes) aortic aneurysm, pulmonary arterial hypertension, Marfan Other Panels syndrome, and RASopathy/Noonan spectrum disorders. • Pulmonary Arterial Hypertension (PAH) Panel Hereditary cardiovascular disease is caused by variants in (20 genes) many different genes, and may be inherited in an autosomal dominant, autosomal recessive, or X-linked manner. Other Indications: than condition-specific panels, we also offer single gene Panels: sequencing for any gene on the panels, targeted variant • Confirmation of genetic diagnosis in a patient with analysis, and targeted deletion/duplication analysis. a clinical diagnosis of cardiovascular disease Tests Offered: • Carrier or pre-symptomatic diagnosis identification Arrythmia Panels in individuals with a family history of cardiovascular • Comprehensive Arrhythmia Panel (81 genes) disease of unknown genetic basis • Atrial Fibrillation (A Fib) Panel (28 genes) Gene Specific Sequencing: • Atrioventricular Block (AV Block) Panel (7 genes) • Confirmation of genetic diagnosis in a patient with • Brugada Syndrome Panel (21 genes) cardiovascular disease and in whom a specific -
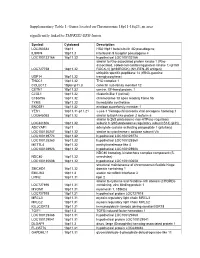
Supplementary Table 1: Genes Located on Chromosome 18P11-18Q23, an Area Significantly Linked to TMPRSS2-ERG Fusion
Supplementary Table 1: Genes located on Chromosome 18p11-18q23, an area significantly linked to TMPRSS2-ERG fusion Symbol Cytoband Description LOC260334 18p11 HSA18p11 beta-tubulin 4Q pseudogene IL9RP4 18p11.3 interleukin 9 receptor pseudogene 4 LOC100132166 18p11.32 hypothetical LOC100132166 similar to Rho-associated protein kinase 1 (Rho- associated, coiled-coil-containing protein kinase 1) (p160 LOC727758 18p11.32 ROCK-1) (p160ROCK) (NY-REN-35 antigen) ubiquitin specific peptidase 14 (tRNA-guanine USP14 18p11.32 transglycosylase) THOC1 18p11.32 THO complex 1 COLEC12 18pter-p11.3 collectin sub-family member 12 CETN1 18p11.32 centrin, EF-hand protein, 1 CLUL1 18p11.32 clusterin-like 1 (retinal) C18orf56 18p11.32 chromosome 18 open reading frame 56 TYMS 18p11.32 thymidylate synthetase ENOSF1 18p11.32 enolase superfamily member 1 YES1 18p11.31-p11.21 v-yes-1 Yamaguchi sarcoma viral oncogene homolog 1 LOC645053 18p11.32 similar to BolA-like protein 2 isoform a similar to 26S proteasome non-ATPase regulatory LOC441806 18p11.32 subunit 8 (26S proteasome regulatory subunit S14) (p31) ADCYAP1 18p11 adenylate cyclase activating polypeptide 1 (pituitary) LOC100130247 18p11.32 similar to cytochrome c oxidase subunit VIc LOC100129774 18p11.32 hypothetical LOC100129774 LOC100128360 18p11.32 hypothetical LOC100128360 METTL4 18p11.32 methyltransferase like 4 LOC100128926 18p11.32 hypothetical LOC100128926 NDC80 homolog, kinetochore complex component (S. NDC80 18p11.32 cerevisiae) LOC100130608 18p11.32 hypothetical LOC100130608 structural maintenance -

Figure S1. Gene Ontology Classification of Abeliophyllum Distichum Leaves Extract-Induced Degs
Figure S1. Gene ontology classification of Abeliophyllum distichum leaves extract-induced DEGs. The results are summarized in three main categories: Biological process, Cellular component and Molecular function. Figure S2. KEGG pathway enrichment analysis using Abeliophyllum distichum leaves extract-DEGs (A). Venn diagram analysis of DEGs involved in PI3K/Akt signaling pathway and Rap1 signaling pathway (B). Figure S3. The expression (A) and protein levels (B) of Akt3 in AL-treated SK-MEL2 cells. Values with different superscripted letters are significantly different (p < 0.05). Table S1. Abeliophyllum distichum leaves extract-induced DEGs. log2 Fold Gene name Gene description Change A2ML1 alpha-2-macroglobulin-like protein 1 isoform 2 [Homo sapiens] 3.45 A4GALT lactosylceramide 4-alpha-galactosyltransferase [Homo sapiens] −1.64 ABCB4 phosphatidylcholine translocator ABCB4 isoform A [Homo sapiens] −1.43 ABCB5 ATP-binding cassette sub-family B member 5 isoform 1 [Homo sapiens] −2.99 ABHD17C alpha/beta hydrolase domain-containing protein 17C [Homo sapiens] −1.62 ABLIM2 actin-binding LIM protein 2 isoform 1 [Homo sapiens] −2.53 ABTB2 ankyrin repeat and BTB/POZ domain-containing protein 2 [Homo sapiens] −1.48 ACACA acetyl-CoA carboxylase 1 isoform 1 [Homo sapiens] −1.76 ACACB acetyl-CoA carboxylase 2 precursor [Homo sapiens] −2.03 ACSM1 acyl-coenzyme A synthetase ACSM1, mitochondrial [Homo sapiens] −3.05 disintegrin and metalloproteinase domain-containing protein 19 preproprotein [Homo ADAM19 −1.65 sapiens] disintegrin and metalloproteinase -

Interactions with Titin and Myomesin Target Obscurin and Obscurin-Like 1 to the M-Band – Implications for Hereditary Myopathies
Research Article 1841 Interactions with titin and myomesin target obscurin and obscurin-like 1 to the M-band – implications for hereditary myopathies Atsushi Fukuzawa1,*, Stephan Lange1,*,‡, Mark Holt1, Anna Vihola2, Virginie Carmignac3,4,§, Ana Ferreiro3,4, Bjarne Udd2,5 and Mathias Gautel1,¶ 1Kingʼs College London, The Randall Division for Cell and Molecular Biophysics, and Cardiovascular Division, New Huntʼs House, London SE1 1UL, UK 2The Folkhälsan Institute of Genetics and Department of Medical Genetics, University of Helsinki, Biomedicum, Helsinki, Finland 3INSERM, U582, Institut de Myologie, Paris, France 4Université Pierre et Marie Curie, Paris, France 5Department of Neurology, Vasa Central Hospital, Vasa, Finland *These authors contributed equally to this work ‡Current address: Department of Medicine, University of California San Diego, USA §Current address: Department of Experimental Medical Science, Lund University, Sweden ¶Author for correspondence (e-mail: [email protected]) Accepted 11 March 2008 Journal of Cell Science 121, 1841-1851 Published by The Company of Biologists 2008 doi:10.1242/jcs.028019 Summary Obscurin, a giant modular muscle protein implicated in G- does overexpression of the binding sites on either myomesin, protein and protein-kinase signalling, can localize to both obscurin or Obsl1. Furthermore, all titin mutations that have sarcomeric Z-disks and M-bands. Interaction of obscurin with been linked to limb-girdle muscular dystrophy 2J (LGMD2J) the Z-disk is mediated by Z-disk titin. Here, we unravel the or Salih myopathy weaken or abrogate titin-obscurin and titin- molecular basis for the unusual localization of obscurin, a Z- Obsl1 binding, and lead to obscurin mislocalization, suggesting disk-associated protein, to the M-band, where its invertebrate that interference with the interaction of these proteins might analogue UNC-89 is also localized. -

Insulin and Glucose Alter Death-Associated Protein Kinase 3 (DAPK3) DNA Methylation
Page 1 of 89 Diabetes Insulin and glucose alter death-associated protein kinase 3 (DAPK3) DNA methylation in human skeletal muscle Jonathan M. Mudry1, David G. Lassiter1, Carolina Nylén1, Sonia García-Calzón2, Erik Näslund3, Anna Krook2, Juleen R. Zierath1,2,4. Running title: DAPK3 DNA methylation in human skeletal muscle 1Department of Molecular Medicine and Surgery, Integrative Physiology, Karolinska Institutet, Stockholm, Sweden. 2Department of Physiology and Pharmacology, Integrative Physiology, Karolinska Institutet, Stockholm, Sweden. 3Division of Surgery, Department of Clinical Sciences, Danderyd Hospital, Karolinska Institutet, Stockholm, Sweden. 4Section of Integrative Physiology, The Novo Nordisk Foundation Center for Basic Metabolic Research, Faculty of Health and Medical Science, University of Copenhagen, Copenhagen, Denmark. Juleen R. Zierath Department of Molecular Medicine and Surgery, Section of Integrative Physiology, Karolinska Institutet, von Eulers väg 4a, SE 171 77 Stockholm, Sweden Phone: +46 (0)8 524 87581 Fax: +46 (0) 8 33 54 36 E-mail: [email protected] Diabetes Publish Ahead of Print, published online December 23, 2016 Diabetes Page 2 of 89 Abstract DNA methylation is altered by environmental factors. We hypothesized DNA methylation is altered in skeletal muscle in response to either insulin or glucose exposure. We performed a genome-wide DNA methylation analysis in muscle from healthy men before and after insulin exposure. DNA methylation of selected genes was determined in muscle from healthy and type 2 diabetic men before and after a glucose tolerance test. Insulin altered DNA methylation in the 3’UTR of the calcium pump ATP2A3 gene. Insulin increased DNA methylation in the gene body of DAPK3, a gene involved in cell proliferation, apoptosis and autophagy. -

MYOM1 (NM 003803) Human Tagged ORF Clone Product Data
OriGene Technologies, Inc. 9620 Medical Center Drive, Ste 200 Rockville, MD 20850, US Phone: +1-888-267-4436 [email protected] EU: [email protected] CN: [email protected] Product datasheet for RC213604 MYOM1 (NM_003803) Human Tagged ORF Clone Product data: Product Type: Expression Plasmids Product Name: MYOM1 (NM_003803) Human Tagged ORF Clone Tag: Myc-DDK Symbol: MYOM1 Synonyms: SKELEMIN Vector: pCMV6-Entry (PS100001) E. coli Selection: Kanamycin (25 ug/mL) Cell Selection: Neomycin ORF Nucleotide >RC213604 representing NM_003803 Sequence: Red=Cloning site Blue=ORF Green=Tags(s) TTTTGTAATACGACTCACTATAGGGCGGCCGGGAATTCGTCGACTGGATCCGGTACCGAGGAGATCTGCC GCCGCGATCGCC ATGTCTTTGCCTTTTTATCAGAGGTGCCACCAGCACTATGATCTCAGCTACCGCAACAAGGACGTGCGCA GCACCGTGAGTCACTACCAGCGGGAGAAGAAACGCTCCGCCGTCTACACCCAGGGCTCCACGGCCTACAG CAGCCGCTCCTCCGCCGCGCACCGCCGGGAGTCCGAGGCCTTCCGTCGGGCGTCCGCCTCCTCCTCCCAG CAGCAGGCCTCGCAGCACGCCCTGAGCTCTGAAGTCAGTCGGAAGGCAGCCTCAGCCTACGATTATGGCT CCTCCCATGGACTTACAGATTCCAGTCTGCTGTTAGATGATTATTCATCCAAGTTGAGCCCCAAACCAAA GAGAGCCAAGCACAGCCTACTGTCTGGAGAAGAGAAAGAAAATTTGCCCAGTGACTACATGGTACCCATT TTCTCAGGACGTCAAAAGCATGTCAGTGGAATTACTGATACGGAAGAAGAAAGAATTAAAGAAGCTGCTG CTTATATAGCCCAGAGGAATCTTCTTGCTAGTGAGGAAGGAATCACAACACCTAAACAGTCCACGGCATC CAAGCAGACCACGGCATCTAAGCAGTCCACGGCATCCAAGCAGTCCACAGCATCCAAGCAGTCCACGGCA TCCAGGCAGTCCACGGCATCCAGGCAGTCTGTGGTTTCCAAACAGGCCACATCCGCTCTTCAACAGGAAG AAACTTCTGAAAAGAAGTCAAGGAAAGTTGTGATTCGAGAAAAGGCAGAACGCCTGTCCCTGAGGAAAAC ATTAGAAGAAACCGAGACATATCATGCCAAGCTGAATGAAGACCATCTTCTCCATGCTCCTGAGTTTATC ATTAAACCTCGCTCCCACACGGTTTGGGAGAAGGAGAATGTAAAATTGCATTGCTCCATAGCAGGCTGGC -

SUPPLEMENTARY MATERIALS and METHODS PBMC Transcriptomics
BMJ Publishing Group Limited (BMJ) disclaims all liability and responsibility arising from any reliance Supplemental material placed on this supplemental material which has been supplied by the author(s) Gut SUPPLEMENTARY MATERIALS AND METHODS PBMC transcriptomics identifies immune-metabolism disorder during the development of HBV-ACLF Contents l Supplementary methods l Supplementary Figure 1 l Supplementary Figure 2 l Supplementary Figure 3 l Supplementary Figure 4 l Supplementary Figure 5 l Supplementary Table 1 l Supplementary Table 2 l Supplementary Table 3 l Supplementary Table 4 l Supplementary Tables 5-14 l Supplementary Table 15 l Supplementary Table 16 l Supplementary Table 17 Li J, et al. Gut 2021;0:1–13. doi: 10.1136/gutjnl-2020-323395 BMJ Publishing Group Limited (BMJ) disclaims all liability and responsibility arising from any reliance Supplemental material placed on this supplemental material which has been supplied by the author(s) Gut SUPPLEMENTARY METHODS Test for HBV DNA The levels of HBV DNA were detected using real-time PCR with a COBAS® AmpliPrep/COBAS® TaqMan 48 System (Roche, Basel, Switzerland) and HBV Test v2.0. Criteria for diagnosing cirrhosis Pathology The gold standard for the diagnosis of cirrhosis is a liver biopsy obtained through a percutaneous or transjugular approach.1 Ultrasonography was performed 2-4 hours before biopsy. Liver biopsy specimens were obtained by experienced physicians. Percutaneous transthoracic puncture of the liver was performed according to the standard criteria. After biopsy, patients were monitored in the hospital with periodic analyses of haematocrit and other vital signs for 24 hours. Cirrhosis was diagnosed according to the globally agreed upon criteria.2 Cirrhosis is defined based on its pathological features under a microscope: (a) the presence of parenchymal nodules, (b) differences in liver cell size and appearance, (c) fragmentation of the biopsy specimen, (d) fibrous septa, and (d) an altered architecture and vascular relationships.