African Meningitis Belt
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Myalgic Encephalomyelitis/Chronic Fatigue
2019 Science & Discovery Webinar Series ME/CFS in the Era of the Human Microbiome: Persistent Pathogens Drive Chronic Symptoms by Interfering With Host Metabolism, Gene Expression, and Immunity with Amy Proal, Ph.D. November 14, 2019 | 1:00 PM Eastern www.SolveME.org About Our Webinars • Welcome to the 2019 Webinar Series! • The audience is muted; use the question box to send us questions. Dr. Proal will address as many questions as time permits at the end of the webinar • Webinars are recorded and the recording is made available on our YouTube channel http://youtube.com/SolveCFS • The Solve ME/CFS Initiative does not provide medical advice www.SolveCFS.org 2019 Science & Discovery Webinar Series ME/CFS in the Era of the Human Microbiome: Persistent Pathogens Drive Chronic Symptoms by Interfering With Host Metabolism, Gene Expression, and Immunity with Amy Proal, Ph.D. November 14, 2019 | 1:00 PM Eastern www.SolveME.org Myalgic Encephalomyelitis/Chronic Fatigue Syndrome in the Era of the Human Microbiome: Persistent Pathogens Drive Chronic Symptoms by Interfering With Host Metabolism, Gene Expression, and Immunity Amy Proal, Autoimmunity Research Foundation/PolyBio Millions of patients across the globe are suffering with myalgic encephalomyelitis (ME/CFS) Currently there is no one disease-specific biomarker and severely ill patients are often wheelchair dependent, bedridden and unable to perform basic tasks of work or daily living. #millionsmissing Myalgic Encephalomeylitis (ME) = swelling of the brain • Unrelenting fatigue that does -

Global Burden of Norovirus and Prospects for Vaccine Development
Global Burden of Norovirus and Prospects for Vaccine Development Primary author Ben Lopman Centers for Disease Control and Prevention Contributors and Reviewers Robert Atmar, Baylor College of Medicine Ralph Baric, University of North Carolina Mary Estes, Baylor College of Medicine Kim Green, NIH; National Institute of Allergy and Infectious Diseases Roger Glass, NIH; Fogarty International Center Aron Hall, Centers for Disease Control and Prevention Miren Iturriza-Gómara, University of Liverpool Cherry Kang, Christian Medical College Bruce Lee, Johns Hopkins University Umesh Parashar, Centers for Disease Control and Prevention Mark Riddle, Naval Medical Research Center Jan Vinjé, Centers for Disease Control and Prevention The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention, or the US Department of Health and Human Services. This work was funded in part by a grant from the Bill & Melinda Gates Foundation to the CDC Foundation. GLOBAL BURDEN OF NOROVIRUS AND PROSPECTS FOR VACCINE DEVELOPMENT | 1 Table of Contents 1. Executive summary ....................................................................3 2. Burden of disease and epidemiology 7 a. Burden 7 i. Global burden and trends of diarrheal disease in children and adults 7 ii. The role of norovirus 8 b. Epidemiology 9 i. Early childhood infections 9 ii. Risk factors, modes and settings of transmission 10 iii. Chronic health consequences associated with norovirus infection? 11 c. Challenges in attributing disease to norovirus 12 3. Norovirus biology, diagnostics and their interpretation for field studies and clinical trials..15 a. Norovirus virology 15 i. Genetic diversity, evolution and related challenges for diagnosis 15 ii. -

Meningococcal a Conjugate Vaccine Into the Routine Immunization Programme
GUIDE TO INTRODUCING MENINGOCOCCAL A CONJUGATE VACCINE INTO THE ROUTINE IMMUNIZATION PROGRAMME GUIDE TO INTRODUCING MENINGOCOCCAL A CONJUGATE VACCINE INTO THE ROUTINE IMMUNIZATION PROGRAMME This publication was jointly developed by the WHO Regional Office for Africa and WHO headquarters. Guide to introducing meningococcal A conjugate vaccine into the routine immunization programme ISBN 978-92-4-151686-0 © World Health Organization 2019 Some rights reserved. This work is available under the Creative Commons Attribution-NonCommercial- ShareAlike 3.0 IGO licence (CC BY-NC-SA 3.0 IGO; https://creativecommons.org/licenses/by-nc- sa/3.0/igo). Under the terms of this licence, you may copy, redistribute and adapt the work for non-commercial purposes, provided the work is appropriately cited, as indicated below. In any use of this work, there should be no suggestion that WHO endorses any specific organization, products or services. The use of the WHO logo is not permitted. If you adapt the work, then you must license your work under the same or equivalent Creative Commons licence. If you create a translation of this work, you should add the following disclaimer along with the suggested citation: “This translation was not created by the World Health Organization (WHO). WHO is not responsible for the content or accuracy of this translation. The original English edition shall be the binding and authentic edition”. Any mediation relating to disputes arising under the licence shall be conducted in accordance with the mediation rules of the World Intellectual Property Organization. Suggested citation. Guide to introducing meningococcal A conjugate vaccine into the routine immunization programme. -
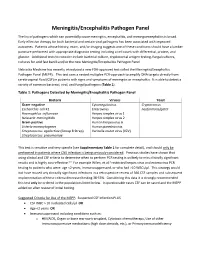
Meningitis/Encephalitis Pathogen Panel
Meningitis/Encephalitis Pathogen Panel The list of pathogens which can potentially cause meningitis, encephalitis, and meningoencephalitis is broad. Early effective therapy for both bacterial and certain viral pathogens has been associated with improved outcomes. Patients whose history, exam, and/or imaging suggests one of these conditions should have a lumber puncture performed with appropriate diagnostic testing including a cell count with differential, protein, and glucose. Additional tests to consider include bacterial culture, cryptococcal antigen testing, fungal cultures, cultures for acid fast bacilli and/or the new Meningitis/Encephalitis Pathogen Panel. Nebraska Medicine has recently introduced a new FDA-approved test called the Meningitis/Encephalitis Pathogen Panel (MEPP). This test uses a nested multiplex PCR-approach to amplify DNA targets directly from cerebrospinal fluid (CSF) in patients with signs and symptoms of meningitis or encephalitis. It is able to detect a variety of common bacterial, viral, and fungal pathogens (Table 1). Table 1: Pathogens Detected by Meningitis/Encephalitis Pathogen Panel Bacteria Viruses Yeast Gram-negative Cytomegalovirus Cryptococcus Escherichia coli K1 Enterovirus neoformans/gattii Haemophilus influenzae Herpes simplex virus 1 Neisseria meningitidis Herpes simplex virus 2 Gram-positive Human herpesvirus 6 Listeria monocytogenes Human parechovirus Streptococcus agalactiae (Group B Strep) Varicella zoster virus (VZV) Streptococcus pneumoniae This test is sensitive and very specific (see Supplementary Table 1 for complete detail), and should only be performed in patients where CNS infection is being seriously considered. Previous studies have shown that using clinical and CSF criteria to determine when to perform PCR testing is unlikely to miss clinically significant results and is highly cost-effective.1-3 For example Wilen, et al.3 restricted herpes virus and enterovirus PCR testing to patients who were: age <2 years, immunosuppressed, or who had >10 WBCs/µl. -

Novel Risk Factors for Coronavirus Disease-Associated Mucormycosis (CAM): a Case Control Study During the Outbreak in India
medRxiv preprint doi: https://doi.org/10.1101/2021.07.24.21261040; this version posted July 26, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. It is made available under a CC-BY-NC-ND 4.0 International license . Novel risk factors for Coronavirus disease-associated mucormycosis (CAM): a case control study during the outbreak in India Umang Arora1, Megha Priyadarshi1, Varidh Katiyar2, Manish Soneja1, Prerna Garg1, Ishan Gupta1,- Vishwesh Bharadiya1, Parul Berry1, Tamoghna Ghosh1, Lajjaben Patel1 , Radhika Sarda1, Shreya Garg3, Shubham Agarwal1, Veronica Arora4, Aishwarya Ramprasad1, Amit Kumar1, Rohit Kumar Garg1, Parul Kodan1, Neeraj Nischal1, Gagandeep Singh5, Pankaj Jorwal1, Arvind Kumar1, Upendra Baitha1, Ved Prakash Meena1, Animesh Ray1, Prayas Sethi1, , Immaculata Xess5, Naval Vikram1, Sanjeev Sinha1, Ashutosh Biswas1,Alok Thakar3, Sushma Bhatnagar6, Anjan Trikha7, Naveet Wig1 1Department of Medicine, AIIMS, Delhi, India 2Department of Neurosurgery, AIIMS, Delhi, India 3Department of Otolaryngology & Head-Neck Surgery, AIIMS, Delhi, India 4Department of Medical Genetics, Sir Ganga Ram Hospital, Delhi, India 5Department of Microbiology, AIIMS, Delhi, India 6Department of Onco-anaesthesia and Palliative Medicine, AIIMS, Delhi, India 7Department of Anaesthesiology, Pain Medicine and Critical Care, AIIMS, Delhi, India 1. Umang Arora, MD (Medicine), Junior Resident, Department of Medicine, AIIMS, Delhi, India. E-mail ID: [email protected] 2. Megha Priyadarshi, DM (Infectious disease), Junior Resident, Department of Medicine, AIIMS, Delhi, India. E-mail ID: [email protected] 3. Varidh Katiyar, MCh (Neurosurgery), Chief Resident, Department of Neurosurgery AIIMS, Delhi, India. E-mail ID: [email protected] 4. -

The Meningitis Vaccine Project: Frequently Asked Questions
The Meningitis Vaccine Project Frequently asked questions June 2011 The disease What is meningococcal disease? Meningococcal disease is an infection of the meninges, the thin lining that surrounds the brain and the spinal cord. It is usually caused by a virus or bacterium (meningococcus). It is transmitted through droplets of respiratory or throat secretions. Bacterial meningitis, such as meningococcal disease, can be very serious because it evolves rapidly and can kill in a few hours. Even with appropriate treatment, around 10 percent of patients die, and up to 20 percent of survivors have serious permanent health problems as a result of the disease (deafness, epilepsy, cerebral palsy, or mental retardation). What is the extent of meningococcal meningitis in Africa? Sub-Saharan Africa has been experiencing explosive and repeated meningococcal epidemics for more than a hundred years. Group A meningococcus is the main cause of meningitis epidemics and accounts for an estimated 80 to 85 percent of all cases. These deadly epidemics occur at intervals of 8–10 years in the 25 countries of the "meningitis belt," a strip of land that extends from Senegal in the west to Ethiopia in the east. Around 450 million people in this area are at risk of disease. More than one million cases of meningitis have been reported in Africa since 1988. In 1996–1997, one of the largest epidemic waves ever recorded in history swept across Africa, causing more than 250,000 cases and 25,000 deaths. The vaccine What is the expected public health impact of this new vaccine? If introduced in all 25 countries of the African meningitis belt, this vaccine is expected to eliminate the primary cause of epidemic meningitis, group A meningococcus, from the entire region, with an estimated 1 million cases of disease prevented and 150 000 young lives saved by 2020. -

Meningococcal Meningitis (Neisseria Meningitidis)
Division of Disease Control What Do I Need To Know? Meningococcal Meningitis (Neisseria meningitidis) What is meningococcal meningitis ? Meningitis is a severe infection of the bloodstream and meninges (a thin lining covering the brain and spinal cord) caused by a bacteria or virus. Bacterial meningitis is usually more severe than viral meningitis but is less common. Bacterial meningitis is most commonly caused by Haemophilus influenzae type B, Streptococcus pneumoniae or Neisseria meningitidis. The most severe form of bacterial meningitis is called Neisseria meningitidis. It is a relatively rare disease and usually occurs as a single isolated event. Clusters of cases or outbreaks are rare in the United States. Who is at risk for meningococcal meningitis? Anyone can get meningococcal meningitis, but it is more common in infants and children. Other people at increased risk for meningitis are college freshmen living in dormitories, microbiologists who are routinely exposed, military recruits, and travelers to areas where meningitis occurs frequently, such as sub-Saharan Africa. What are the symptoms of meningococcal meningitis? Although most people exposed to the meningococcal bacteria do not become seriously ill, some may develop fever, headache, vomiting, stiff neck and a rash. Meningitis can cause sensitivity to light, confusion, drowsiness, seizures and sometimes coma. The disease is sometimes fatal. How soon do symptoms appear? The symptoms may appear one to 10 days after exposure, but usually less than four days. How is meningococcal meningitis spread? Meningococcal meningitis is spread by direct, close contact with nasal or throat discharges of an infected person. Many people carry meningococcal bacteria in their nose and throat without any signs of illness, while others may develop serious symptoms. -

Meningococcal a Conjugate Vaccine Roll-Out in the African Meningitis Belt
Meningococcal A conjugate vaccine roll-out in the African meningitis belt Summary update prepared by the Meningitis Vaccine Project & partners SAGE, October 2014 Background Over the last century sub-Saharan Africa has been plagued by repeated epidemics of meningococcal meningitis. Almost all of the major outbreaks have been caused by group A Neisseria meningitidis . Reactive immunizations with polysaccharide vaccines have been used for the last 30 years but have not succeeded in controlling the problem. After the disastrous 1996–1997 epidemic with more than 250,000 cases and 25,000 deaths, there arose renewed interest in developing a preventive strategy based on new meningococcal conjugate vaccines. In June 2001, the Bill & Melinda Gates Foundation provided core funding for the establishment of the Meningitis Vaccine Project (MVP), a partnership between PATH and the World Health Organization (WHO), with the goal of eliminating epidemic meningitis as a public health problem in sub-Saharan Africa through the development, testing, licensure, and widespread introduction of meningococcal conjugate vaccines. A monovalent group A meningococcal (MenA) conjugate vaccine, MenAfriVac, a registered trademark of the Serum Institute of India, was developed through the MVP. The vaccine was licensed, for use in individuals aged 1 to 29 years, in 2009 and prequalified by WHO in 2010. Comprehensive mass immunization campaigns of 1- to 29-year olds with a single dose of MenAfriVac have been a cornerstone of the MenA conjugate vaccine introduction plan. This strategy aims to strongly and immediately protect individuals directly and reduce bacterial carriage and transmission, and thereby rapidly reduce overall disease-related morbidity and mortality rates within the community. -

Seasonal Meningococcal Meningitis Outbreaks in Nigeria: a Need for An
Available online at www.ijmrhs.com al R edic ese M a of rc l h a & n r H u e o a J l l t h International Journal of Medical Research & a n S ISSN No: 2319-5886 o c i t i Health Sciences, 2020, 9(6): 31-37 e a n n c r e e t s n I • • I J M R H S Seasonal Meningococcal Meningitis Outbreaks in Nigeria: A Need for an Accelerated Introduction of a Conjugated Meningococcal Vaccine into the National Routine Immunization Schedule Semeeh Akinwale Omoleke1 and Kehinde Kazeem Kanmodi2,3* 1 World Health Organization, Kebbi State Field Office, Birnin Kebbi, Nigeria 2 Cephas Health Research Initiative Inc, Ibadan, Nigeria 3 Mental and Oral Health Development Organization Inc, Birnin Kebbi, Nigeria *Corresponding e-mail: [email protected] ABSTRACT Epidemic meningococcal meningitis affects huge populations annually in sub-Saharan Africa with differentially higher death rates among children. Nigeria is one of the twenty-six countries that lie in ‘African meningitis belt’. This paper briefly describes the epidemiology of seasonal recurrent meningococcal meningitis, current efforts to address the epidemics, and then argues for an accelerated introduction of conjugated meningococcal vaccine into routine immunization in Nigeria. This paper also highlights the nature of the epidemics with its attendant impacts on the population; the weaknesses of the current strategies; the emergence of mixed pathogens; the challenges and potential opportunities associated with an introduction of routine vaccination against meningococcal meningitis. The quick introduction of the conjugated meningococcal vaccine into expanded program on immunization (EPI) schedule will mitigate the risk of future massive outbreaks and its attendant morbidity, mortality and larger societal cost. -

Functional Genomics of Neisseria Meningitidis Pathogenesis
© 2000 Nature America Inc. • http://medicine.nature.com ARTICLES Functional genomics of Neisseria meningitidis pathogenesis YAO-HUI SUN1, SHARMILA BAKSHI1, RONALD CHALMERS2 & CHRISTOPH M. TANG1 1University Department of Paediatrics, University of Oxford, John Radcliffe Hospital, Oxford OX3 9DU, United Kingdom 2Department of Biochemistry, University of Oxford, South Parks Road, Oxford OX1 3QU, United Kingdom Correspondence to be addressed to C.M.T.; Email: [email protected] and R.C.; Email: [email protected] The pathogenic bacterium Neisseria meningitidis is an important containing unique signature tags (fig, 1b), in 96 separate trans- cause of septicemia and meningitis, especially in childhood1. The position reactions. The modified genomic DNA was repaired, establishment and maintenance of bacteremic infection is a pre- and returned to the host by transformation. We selected 30 mu- requisite for all the pathological sequelae of meningococcal in- tants from each reaction, resulting in a library of 2,880 mutants fection. To further understand the genetic basis of this essential (30 pools × 96). To determine whether Tn10 insertion occurs at step in pathogenesis, we analyzed a library of 2,850 insertional diverse sites, we assessed 40 transformants from a single trans- .com mutants of N. meningitidis for their capacity to cause systemic in- position reaction by Southern blot analysis. Each had a single, fection in an infant rat model. The library was constructed by in distinct Tn10 insertion (Fig. 1c). To establish whether Tn10 in- .nature vitro modification of Neisseria genomic DNA with the purified tegration was stable during systemic infection of infant rats, we components of Tn10 transposition2. -

The Interaction Between Human Antimicrobial Use and the Risk of Foodborne Zoonotic Bacteria
Downloaded from orbit.dtu.dk on: Dec 20, 2017 The interaction between human antimicrobial use and the risk of foodborne zoonotic bacteria Koningstein, Maike; Hald, Tine; Mølbak, Kåre Publication date: 2014 Document Version Peer reviewed version Link back to DTU Orbit Citation (APA): Koningstein, M., Hald, T., & Mølbak, K. (2014). The interaction between human antimicrobial use and the risk of foodborne zoonotic bacteria. National Food Institute, Technical University of Denmark. General rights Copyright and moral rights for the publications made accessible in the public portal are retained by the authors and/or other copyright owners and it is a condition of accessing publications that users recognise and abide by the legal requirements associated with these rights. • Users may download and print one copy of any publication from the public portal for the purpose of private study or research. • You may not further distribute the material or use it for any profit-making activity or commercial gain • You may freely distribute the URL identifying the publication in the public portal If you believe that this document breaches copyright please contact us providing details, and we will remove access to the work immediately and investigate your claim. The interaction between human antimicrobial use and the risk of foodborne zoonotic bacteria Maike Koningstein, M.Sc. PhD Thesis 2013 Statens Serum Institute Department of Infectious Disease Epidemiology Artillerivej 5, 2300 Copenhagen, Denmark and National Food Institute, DTU Department for Microbiology -

Yaoundé Declaration
YAOUNDE DECLARATION ON ELIMINATION OF MENINGOCOCCAL MENINGITIS TYPE A EPIDEMICS AS A PUBLIC HEALTH PROBLEM IN AFRICA _________________________________________________________________________ Sub-Saharan Africa is plagued by repeated meningitis epidemics concentrated in the “meningitis belt”, a vast region stretching from Senegal, on the west coast, to Somalia, on the east coast. The epidemics occur every year, with recurrent peaks every 8 to 12 years. The disease takes a heavy socioeconomic and human toll. More than a decade after a major meningitis epidemic in 1996 which resulted in more than 250 000 cases and 25 000 deaths, there is another threat to the countries of the African meningitis belt. An epidemiological survey of previous epidemics shows that Neisseria meningitis A is the main cause of meningococcal meningitis epidemics in Africa. About 500 million people living in countries of the meningitis belt are at risk of contracting meningococcus type A. Despite an appropriate management of cases, at least 10% of patients still die from the disease and nearly 20% recover with serious irreversible sequelae. For more than 20 years now, the control of meningitis epidemics is principally based on epidemiological surveillance and response mass immunization campaigns with meningococcal polysaccaharide vaccines which have shown their limits. The implementation of a new strategy to combat meningococcal meningitis epidemics in the African belt is necessary. It consists of organizing preventive mass campaigns and seeks to introduce a new meningococcal type A conjugate vaccine. It will help immunize, during the 2009 to 2015 period, about 250 million people aged 1 to 29 years, including 23 million children living in 25 African countries.