DLC1 Is a Direct Target of Activated YAP/TAZ That Drives Collective Migration and Sprouting Angiogenesis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
Supplemental Tables4.Pdf
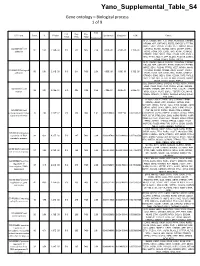
Yano_Supplemental_Table_S4 Gene ontology – Biological process 1 of 9 Fold List Pop Pop GO Term Count % PValue Bonferroni Benjamini FDR Genes Total Hits Total Enrichment DLC1, CADM1, NELL2, CLSTN1, PCDHGA8, CTNNB1, NRCAM, APP, CNTNAP2, FERT2, RAPGEF1, PTPRM, MPDZ, SDK1, PCDH9, PTPRS, VEZT, NRXN1, MYH9, GO:0007155~cell CTNNA2, NCAM1, NCAM2, DDR1, LSAMP, CNTN1, 50 5.61 2.14E-08 510 311 7436 2.34 4.50E-05 4.50E-05 3.70E-05 adhesion ROR2, VCAN, DST, LIMS1, TNC, ASTN1, CTNND2, CTNND1, CDH2, NEO1, CDH4, CD24A, FAT3, PVRL3, TRO, TTYH1, MLLT4, LPP, NLGN1, PCDH19, LAMA1, ITGA9, CDH13, CDON, PSPC1 DLC1, CADM1, NELL2, CLSTN1, PCDHGA8, CTNNB1, NRCAM, APP, CNTNAP2, FERT2, RAPGEF1, PTPRM, MPDZ, SDK1, PCDH9, PTPRS, VEZT, NRXN1, MYH9, GO:0022610~biological CTNNA2, NCAM1, NCAM2, DDR1, LSAMP, CNTN1, 50 5.61 2.14E-08 510 311 7436 2.34 4.50E-05 4.50E-05 3.70E-05 adhesion ROR2, VCAN, DST, LIMS1, TNC, ASTN1, CTNND2, CTNND1, CDH2, NEO1, CDH4, CD24A, FAT3, PVRL3, TRO, TTYH1, MLLT4, LPP, NLGN1, PCDH19, LAMA1, ITGA9, CDH13, CDON, PSPC1 DCC, ENAH, PLXNA2, CAPZA2, ATP5B, ASTN1, PAX6, ZEB2, CDH2, CDH4, GLI3, CD24A, EPHB1, NRCAM, GO:0006928~cell CTTNBP2, EDNRB, APP, PTK2, ETV1, CLASP2, STRBP, 36 4.04 3.46E-07 510 205 7436 2.56 7.28E-04 3.64E-04 5.98E-04 motion NRG1, DCLK1, PLAT, SGPL1, TGFBR1, EVL, MYH9, YWHAE, NCKAP1, CTNNA2, SEMA6A, EPHA4, NDEL1, FYN, LRP6 PLXNA2, ADCY5, PAX6, GLI3, CTNNB1, LPHN2, EDNRB, LPHN3, APP, CSNK2A1, GPR45, NRG1, RAPGEF1, WWOX, SGPL1, TLE4, SPEN, NCAM1, DDR1, GRB10, GRM3, GNAQ, HIPK1, GNB1, HIPK2, PYGO1, GO:0007166~cell RNF138, ROR2, CNTN1, -
DLC1 Expression Is Reduced in Human Cutaneous Melanoma And
Modern Pathology (2014) 27, 1203–1211 & 2014 USCAP, Inc. All rights reserved 0893-3952/14 $32.00 1203 DLC1 expression is reduced in human cutaneous melanoma and correlates with patient survival Cecilia Sjoestroem1, Shahram Khosravi1, Yabin Cheng1, Gholamreza Safaee Ardekani1, Magdalena Martinka2 and Gang Li1 1Department of Dermatology and Skin Science, Vancouver Coastal Health Research Institute, University of British Columbia, Vancouver, BC, Canada and 2Department of Pathology, Vancouver General Hospital, Vancouver, BC, Canada Deleted in Liver Cancer-1 (DLC1) is a Rho-GTPase-activating protein known to be downregulated and function as a tumor suppressor in numerous solid and hematological cancers. Its expression status in melanoma is currently unknown however, prompting us to examine this. Using immunohistochemistry and tissue microarrays containing a large set of melanocytic lesions (n ¼ 539), we examined the expression profile of DLC1 in melanoma progression, as well as the association between DLC1 and patient survival. We detected both cytoplasmic and nuclear DLC1 expression, and found that whereas cytoplasmic DLC1 was significantly downregulated in metastatic melanoma compared with nevi and primary melanoma, nuclear DLC1 expression was significantly down in primary melanoma compared with nevi, and then further down in metastatic melanoma. Loss of cytoplasmic DLC1 was significantly associated with poorer overall and disease-specific 5-year survival rates of all melanoma (Po0.001 and P ¼ 0.001, respectively) and metastatic melanoma patients (P ¼ 0.020 and 0.008, respectively), and similar results were seen for nuclear DLC1 (Po0.001 for both overall and disease-specific survival for all melanoma patients, and P ¼ 0.004 for metastatic melanoma patients). -

Cooperative Antiproliferative Effect of Coordinated Ectopic Expression Of
ONCOLOGY LETTERS 12: 1591-1596, 2016 Cooperative antiproliferative effect of coordinated ectopic expression of DLC1 tumor suppressor protein and silencing of MYC oncogene expression in liver cancer cells: Therapeutic implications XUYU YANG1,2, XIAOLING ZHOU1,3, PAUL TONE4, MARIAN E. DURKIN1,5 and NICHOLAS C. POPESCU1,5 1Laboratory of Experimental Carcinogenesis, Center for Cancer Research, National Cancer Institute, National Institutes of Health; 2Section on Cellular Neurobiology, Eunice Kennedy Shriver National Institute of Child Health and Development; 3Laboratory of Cancer Biology and Genetics, National Cancer Institute, National Institutes of Health, Bethesda, MD 2089-4262; 4Department of Medicine, Richmond University Medical Center, Staten Island, NY 10310; 5Laboratory of Cellular Oncology, National Cancer Institute, National Institutes of Health, Bethesda, MD 20892-4262, USA Received November 11, 2015; Accepted June 3, 2016 DOI: 10.3892/ol.2016.4781 Abstract. Human hepatocellular carcinoma (HCC) is one of the injection to mice bearing subcutaneous xenografts of Huh7 cells most common types of cancer and has a very poor prognosis; transfected with shMyc or control shRNA. A cooperative thus, the development of effective therapies for the treatment of inhibitory effect of DLC1 expression and c-Myc knockdown on advanced HCC is of high clinical priority. In the present study, the growth of Huh7-derived tumors was observed, suggesting the anti-oncogenic effect of combined knockdown of c-Myc that targeted liver cell delivery of DLC1 and c-Myc shRNA expression and ectopic restoration of deleted in liver cancer 1 may serve as a possible gene therapy modality for the treatment (DLC1) expression was investigated in human liver cancer of human HCC. -

Transcriptome Analysis of Human Diabetic Kidney Disease
ORIGINAL ARTICLE Transcriptome Analysis of Human Diabetic Kidney Disease Karolina I. Woroniecka,1 Ae Seo Deok Park,1 Davoud Mohtat,2 David B. Thomas,3 James M. Pullman,4 and Katalin Susztak1,5 OBJECTIVE—Diabetic kidney disease (DKD) is the single cases, mild and then moderate mesangial expansion can be leading cause of kidney failure in the U.S., for which a cure has observed. In general, diabetic kidney disease (DKD) is not yet been found. The aim of our study was to provide an considered a nonimmune-mediated degenerative disease unbiased catalog of gene-expression changes in human diabetic of the glomerulus; however, it has long been noted that kidney biopsy samples. complement and immunoglobulins sometimes can be de- — tected in diseased glomeruli, although their role and sig- RESEARCH DESIGN AND METHODS Affymetrix expression fi arrays were used to identify differentially regulated transcripts in ni cance is not clear (4). 44 microdissected human kidney samples. The DKD samples were The understanding of DKD has been challenged by multi- significant for their racial diversity and decreased glomerular ple issues. First, the diagnosis of DKD usually is made using filtration rate (~20–30 mL/min). Stringent statistical analysis, using clinical criteria, and kidney biopsy often is not performed. the Benjamini-Hochberg corrected two-tailed t test, was used to According to current clinical practice, the development of identify differentially expressed transcripts in control and diseased albuminuria in patients with diabetes is sufficient to make the glomeruli and tubuli. Two different Web-based algorithms were fi diagnosis of DKD (5). We do not understand the correlation used to de ne differentially regulated pathways. -

Identification of a Region of Homozygous Deletion on 8P22–23.1 in Medulloblastoma
Oncogene (2002) 21, 1461 ± 1468 ã 2002 Nature Publishing Group All rights reserved 0950 ± 9232/02 $25.00 www.nature.com/onc Identi®cation of a region of homozygous deletion on 8p22 ± 23.1 in medulloblastoma Xiao-lu Yin1, Jesse Chung-sean Pang1 and Ho-keung Ng*,1 1Department of Anatomical and Cellular Pathology, Prince of Wales Hospital, The Chinese University of Hong Kong, Hong Kong, China To identify critical tumor suppressor loci that are radiotherapy and chemotherapy, have greatly improved associated with the development of medulloblastoma, the outcomes of patients. In the past 30 years, the 5- we performed a comprehensive genome-wide allelotype year survival rate has increased from 10 to about 50%. analysis in a series of 12 medulloblastomas. Non-random However, long-term survival in children with advanced allelic imbalances were identi®ed on chromosomes 7q disease is only about 30% (Giangaspero et al., 2000; (58.3%), 8p (66.7%), 16q (58.3%), 17p (58.3%) and Heideman et al., 1997). Further enhancement of 17q (66.7%). Comparative genomic hybridization analy- survival will rely on a better understanding of the sis con®rmed that allelic imbalances on 8p, 16q and 17p biology of this malignant disease to improve current were due to loss of genetic materials. Finer deletion treatments or to develop novel therapy. mapping in an expanded series of 23 medulloblastomas Non-random loss of genetic material from chromo- localized the common deletion region on 8p to an interval somal loci is a common feature in the development of of 18.14 cM on 8p22 ± 23.2. -

Looking for Missing Proteins in the Proteome Of
Looking for Missing Proteins in the Proteome of Human Spermatozoa: An Update Yves Vandenbrouck, Lydie Lane, Christine Carapito, Paula Duek, Karine Rondel, Christophe Bruley, Charlotte Macron, Anne Gonzalez de Peredo, Yohann Coute, Karima Chaoui, et al. To cite this version: Yves Vandenbrouck, Lydie Lane, Christine Carapito, Paula Duek, Karine Rondel, et al.. Looking for Missing Proteins in the Proteome of Human Spermatozoa: An Update. Journal of Proteome Research, American Chemical Society, 2016, 15 (11), pp.3998-4019. 10.1021/acs.jproteome.6b00400. hal-02191502 HAL Id: hal-02191502 https://hal.archives-ouvertes.fr/hal-02191502 Submitted on 19 Mar 2021 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Journal of Proteome Research 1 2 3 Looking for missing proteins in the proteome of human spermatozoa: an 4 update 5 6 Yves Vandenbrouck1,2,3,#,§, Lydie Lane4,5,#, Christine Carapito6, Paula Duek5, Karine Rondel7, 7 Christophe Bruley1,2,3, Charlotte Macron6, Anne Gonzalez de Peredo8, Yohann Couté1,2,3, 8 Karima Chaoui8, Emmanuelle Com7, Alain Gateau5, AnneMarie Hesse1,2,3, Marlene 9 Marcellin8, Loren Méar7, Emmanuelle MoutonBarbosa8, Thibault Robin9, Odile Burlet- 10 Schiltz8, Sarah Cianferani6, Myriam Ferro1,2,3, Thomas Fréour10,11, Cecilia Lindskog12,Jérôme 11 1,2,3 7,§ 12 Garin , Charles Pineau . -

A Novel JAK1 Mutant Breast Implant-Associated Anaplastic Large Cell Lymphoma Patient-Derived Xenograft Fostering Pre- Clinical Discoveries
Cancers 2019 S1 of S18 Supplementary Materials: A Novel JAK1 Mutant Breast Implant-Associated Anaplastic Large Cell Lymphoma Patient-Derived Xenograft Fostering Pre- Clinical Discoveries Danilo Fiore, Luca Vincenzo Cappelli, Paul Zumbo, Jude M. Phillip, Zhaoqi Liu, Shuhua Cheng, Liron Yoffe, Paola Ghione, Federica Di Maggio, Ahmet Dogan, Inna Khodos, Elisa de Stanchina, Joseph Casano, Clarisse Kayembe, Wayne Tam, Doron Betel, Robin Foa’, Leandro Cerchietti, Raul Rabadan, Steven Horwitz, David M. Weinstock and Giorgio Inghirami A B C Figure S1. (A) Histology micrografts on IL89 PDTX show overall similarity between T1 T3 and T7 passages (upper panels). Immunohistochemical stains with the indicated antibodies (anti-CD3, anti- CD25 and anti-CD8 [x20]) (lower panels). (B) Flow cytometry panel comprehensive of the most represented surface T-cell lymphoma markers, including: CD2, CD3, CD4, CD5, CD8, CD16, CD25, CD30, CD56, TCRab, TCRgd. IL89 PDTX passage T3 is here depicted for illustration purposes. (C) Analysis of the TCR gamma specific rearrangement clonality in IL89 diagnostic sample and correspondent PDTX after 1 and 5 passages (T1 and T5). A WT Primary p.G1097D IL89 T1 p.G1097D IL89 T5 p.G1097D IL89 cell line B Figure S2. (A) Sanger sequencing confirms the presence of the JAK1 p.G1097D mutation in IL89 PDTX samples and in the cell line, but the mutation is undetectable in the primary due to the low sensitivity of the technique. (B) Manual backtracking of mutations in the primary tumor using deep sequencing data allowed for the identification of several hits at a very low VAF compared to the PDTX-T5. A B IL89 CTRL 30 CTRL Ruxoli?nib S 20 M Ruxoli?nib A R G 10 0 1 2 3 4 5 6 7 8 9 0 1 2 3 4 1 1 1 1 1 WEEKS AFTER ENGRAFTMENT Figure S3. -

Purification of Full-Length Bcl-2 Family Proteins and Molecular Analysis of Bim-DLC1 Interaction
Purification of full-length Bcl-2 family proteins and molecular analysis of Bim-DLC1 interaction Inaugural-Dissertation zur Erlangung des akademischen Grades des Doktors der Naturwissenschaften (Dr. rer. nat.) an der Fakultät für Biologie der Albert-Ludwigs-Universität Freiburg im Breisgau vorgelegt von Aristomenis Roukounakis geboren in Heraklion, Griechenland Freiburg im Breisgau Februar 2018 Die Untersuchungen zur vorliegenden Arbeit wurden von Oktober 2013 bis Februar 2018 am Institut für Medizinische Mikrobiologie und Hygiene des Universitätsklinikums der Albert- Ludwigs-Universität Freiburg unter der Leitung von Prof. Dr. Georg Häcker durchgeführt. Dekan der Fakultät für Biologie: Prof. Dr. Bettina Warscheid Promotionsvorsitzender: Prof. Dr. Andreas Hiltbrunner Betreuer der Arbeit: Prof. Dr. Georg Häcker Referent: Prof. Dr. Georg Häcker Koreferent: Prof. Dr. Tilman Brummer Drittprüfer: Prof. Dr. Christof Meisinger Datum der mündlichen Prüfung: 07.05.2018 Publication Major part of this thesis has been published in Genes & Development journal (doi: 10.1101/gad.302497.117). Dynein light chain 1 induces assembly of large Bim complexes on mitochondria that stabilize Mcl-1 and regulate apoptosis Prafull Kumar Singh* Aristomenis Roukounakis* Daniel O. Frank, Susanne Kirschnek, Kushal Kumar Das, Simon Neumann, Josef Madl, Winfried Römer, Carina Zorzin, Christoph Borner, Ana Garcia-Saez, Arnim Weber and Georg Häcker* *equal contribution Table of contents I. Summary ............................................................................................................................. -

Intragenic Integration in DLC1 Sustains Factor VIII Expression in Primary Human Cells Without Insertional Oncogenicity
Gene Therapy (2014) 21, 402–412 OPEN & 2014 Macmillan Publishers Limited All rights reserved 0969-7128/14 www.nature.com/gt ORIGINAL ARTICLE Intragenic integration in DLC1 sustains factor VIII expression in primary human cells without insertional oncogenicity J Sivalingam1,2, TT Phan3,4 and OL Kon1,2 Techniques enabling precise genome modifications enhance the safety of gene-based therapy. DLC1 is a hot spot for phiC31 integrase-mediated transgene integration in vitro and in vivo. Here we show that integration of a coagulation factor VIII transgene into intron 7 of DLC1 supports durable expression of factor VIII in primary human umbilical cord-lining epithelial cells. Oligoclonal cells with factor VIII transgene integrated in DLC1 did not have altered expression of DLC1 or neighbouring genes within a 1-Mb interval. Only 1.9% of all expressed genes were transcriptionally altered; most were downregulated and mapped to cell cycle and DNA repair pathways. DLC1-integrated cells were not tumourigenic in vivo and were normal by high-resolution genomic DNA copy number analysis. Our data identify DLC1 as a locus for durable transgene expression that does not incur features of insertional oncogenesis, thus expanding options for developing ex vivo cell therapy mediated by site-specific integration methods. Gene Therapy (2014) 21, 402–412; doi:10.1038/gt.2014.11; published online 20 February 2014 INTRODUCTION human coagulation factor FVIII (FVIII) transgene and corrected the 17 One of the fundamental requirements for developing gene-based disease phenotype when implanted in FVIII-deficient mice. Up to cell therapy for disorders caused by deficient production of 40% of FVIII transgene integrations in a mixed population of CLECs specific proteins, such as the haemophilias, is durable expression occurred in 8p22, and clonal cultures of genome-modified CLECs of the corrective transgene product. -

SRC and ERK Cooperatively Phosphorylate DLC1 and Attenuate Its Rho-GAP and Tumor Suppressor Functions
ARTICLE SRC and ERK cooperatively phosphorylate DLC1 and attenuate its Rho-GAP and tumor suppressor functions Brajendra K. Tripathi1,MeghanF.Anderman1, Xiaolan Qian1,MingZhou2, Dunrui Wang1, Alex G. Papageorge1, and Douglas R. Lowy1 SRC and ERK kinases control many cell biological processes that promote tumorigenesis by altering the activity of oncogenic and tumor suppressor proteins. We identify here a physiological interaction between DLC1, a focal adhesion protein and tumor suppressor, with SRC and ERK. The tumor suppressor function of DLC1 is attenuated by phosphorylation of tyrosines Y451 and Y701 by SRC, which down-regulates DLC1’s tensin-binding and Rho-GAP activities. ERK1/2 phosphorylate DLC1 on serine S129, which increases both the binding of SRC to DLC1 and SRC-dependent phosphorylation of DLC1. SRC inhibitors exhibit potent antitumor activity in a DLC1-positive transgenic cancer model and a DLC1-positive tumor xenograft model, due to reactivation of the tumor suppressor activities of DLC1. Combined treatment of DLC1-positive tumors with SRC plus AKT inhibitors has even greater antitumor activity. Together, these findings indicate cooperation between the SRC, ERK1/2, and AKT kinases to reduce DLC1 Rho-GAP and tumor suppressor activities in cancer cells, which can be reactivated by the kinase inhibitors. Introduction The SRC gene is the prototypic member of the SRC family kinases 2014; Takahashi et al., 2017). In addition, SRC phosphorylation of (SFKs), whose nine members encode nonreceptor tyrosine- other targets, such as PP2A and caveolin-1, can attenuate their protein kinases that share a similar structure and have key tumor suppressor activities. roles in normal physiology and cancer (Sen and Johnson, 2011). -

Transcriptomic Analysis Reveals Sex-Specific Differences in The
López-Nieva et al. BMC Genomics (2016) 17:698 DOI 10.1186/s12864-016-3036-0 RESEARCH ARTICLE Open Access Transcriptomic analysis reveals sex-specific differences in the expression of Dcl1 and Fis1 genes in the radio-adaptive response of thymocytes to TRP53-mediated apoptosis Pilar López-Nieva1,2, Manuel Malavé1,2, Laura González-Sánchez1,2,3, José Fernández-Piqueras1,2,3,4*, Pablo Fernández-Navarro5,6* and Javier Santos1,2,3,4* Abstract Background: Radio-Adaptive Response (RAR) is a biological defense mechanism whereby exposure to low dose ionizing radiation (IR) mitigates the detrimental effects of high dose irradiation. RAR has been widely observed in vivo using as endpoint less induction of apoptosis. However, sex differences associated with RAR and variations between males and females on global gene expression influenced by RAR have not been still investigated. In addition, the response to radiation-induced apoptosis is associated with phosphorylation of TRP53 at both the serine 15 (ser-18 in the mouse) and serine 392 (ser-389 in mice) residues, but the role of these two phosphorylated forms in male and female RAR remains to be elucidated. Results: We analyzed the effect of administering priming low dose radiation (0.075 Gy of X-rays) prior to high dose radiation (1.75 Gy of γ-rays) on the level of caspase-3-mediated apoptosis and on global transcriptional expression in thymocytes of male and female mice. Here, we provide the first evidence of a differential sex effect of RAR on the reduction of thymocyte apoptosis with males showing lesser levels of caspase-3-mediated apoptosis than females. -

Mutations in Six Nephrosis Genes Delineate a Pathogenic Pathway Amenable to Treatment
Mutations in six nephrosis genes delineate a pathogenic pathway amenable to treatment The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation Ashraf, S., H. Kudo, J. Rao, A. Kikuchi, E. Widmeier, J. A. Lawson, W. Tan, et al. 2018. “Mutations in six nephrosis genes delineate a pathogenic pathway amenable to treatment.” Nature Communications 9 (1): 1960. doi:10.1038/s41467-018-04193-w. http://dx.doi.org/10.1038/s41467-018-04193-w. Published Version doi:10.1038/s41467-018-04193-w Citable link http://nrs.harvard.edu/urn-3:HUL.InstRepos:37160222 Terms of Use This article was downloaded from Harvard University’s DASH repository, and is made available under the terms and conditions applicable to Other Posted Material, as set forth at http:// nrs.harvard.edu/urn-3:HUL.InstRepos:dash.current.terms-of- use#LAA ARTICLE DOI: 10.1038/s41467-018-04193-w OPEN Mutations in six nephrosis genes delineate a pathogenic pathway amenable to treatment Shazia Ashraf et al.# No efficient treatment exists for nephrotic syndrome (NS), a frequent cause of chronic kidney disease. Here we show mutations in six different genes (MAGI2, TNS2, DLC1, CDK20, ITSN1, ITSN2) as causing NS in 17 families with partially treatment-sensitive NS (pTSNS). These 1234567890():,; proteins interact and we delineate their roles in Rho-like small GTPase (RLSG) activity, and demonstrate deficiency for mutants of pTSNS patients. We find that CDK20 regulates DLC1. Knockdown of MAGI2, DLC1,orCDK20 in cultured podocytes reduces migration rate. Treatment with dexamethasone abolishes RhoA activation by knockdown of DLC1 or CDK20 indicating that steroid treatment in patients with pTSNS and mutations in these genes is mediated by this RLSG module.