WO 2012/069150 A2 31 May 20 12 (31.05.2012) W P O P C T
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Nitrate Prodrugs Able to Release Nitric Oxide in a Controlled and Selective
Europäisches Patentamt *EP001336602A1* (19) European Patent Office Office européen des brevets (11) EP 1 336 602 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: (51) Int Cl.7: C07C 205/00, A61K 31/00 20.08.2003 Bulletin 2003/34 (21) Application number: 02425075.5 (22) Date of filing: 13.02.2002 (84) Designated Contracting States: (71) Applicant: Scaramuzzino, Giovanni AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU 20052 Monza (Milano) (IT) MC NL PT SE TR Designated Extension States: (72) Inventor: Scaramuzzino, Giovanni AL LT LV MK RO SI 20052 Monza (Milano) (IT) (54) Nitrate prodrugs able to release nitric oxide in a controlled and selective way and their use for prevention and treatment of inflammatory, ischemic and proliferative diseases (57) New pharmaceutical compounds of general effects and for this reason they are useful for the prep- formula (I): F-(X)q where q is an integer from 1 to 5, pref- aration of medicines for prevention and treatment of in- erably 1; -F is chosen among drugs described in the text, flammatory, ischemic, degenerative and proliferative -X is chosen among 4 groups -M, -T, -V and -Y as de- diseases of musculoskeletal, tegumental, respiratory, scribed in the text. gastrointestinal, genito-urinary and central nervous sys- The compounds of general formula (I) are nitrate tems. prodrugs which can release nitric oxide in vivo in a con- trolled and selective way and without hypotensive side EP 1 336 602 A1 Printed by Jouve, 75001 PARIS (FR) EP 1 336 602 A1 Description [0001] The present invention relates to new nitrate prodrugs which can release nitric oxide in vivo in a controlled and selective way and without the side effects typical of nitrate vasodilators drugs. -

Protocol Supplementary
Optimal Pharmacological Management and Prevention of Glucocorticoid-Induced Osteoporosis (GIOP) Protocol for a Systematic Review and Network Meta-Analysis Supplementary Materials: Sample Search Strategy Supplementary 1: MEDLINE Search Strategy Database: OVID Medline Epub Ahead of Print, In-Process & Other Non-Indexed Citations, Ovid MEDLINE(R) Daily and Ovid MEDLINE(R) 1946 to Present Line 1 exp Osteoporosis/ 2 osteoporos?s.ti,ab,kf. 3 Bone Diseases, Metabolic/ 4 osteop?eni*.ti,ab,kf. 5 Bone Diseases/ 6 exp Bone Resorption/ 7 malabsorption.ti,ab,kf. 8 Bone Density/ 9 BMD.ti,ab,kf. 10 exp Fractures, Bone/ 11 fracture*.ti,ab,kf. 12 (bone* adj2 (loss* or disease* or resorption* or densit* or content* or fragil* or mass* or demineral* or decalcif* or calcif* or strength*)).ti,ab,kf. 13 osteomalacia.ti,ab,kf. 14 or/1-13 15 exp Glucocorticoids/ 16 exp Steroids/ 17 (glucocorticoid* or steroid* or prednisone or prednisolone or hydrocortisone or cortisone or triamcinolone or dexamethasone or betamethasone or methylprednisolone).ti,ab,kf. 18 or/15-17 19 14 and 18 20 ((glucocorticoid-induced or glucosteroid-induced or corticosteroid-induced or glucocorticosteroid-induced) adj1 osteoporos?s).ti,ab,kf. 21 19 or 20 22 exp Diphosphonates/ 23 (bisphosphon* or diphosphon*).ti,ab,kf. 24 exp organophosphates/ or organophosphonates/ 25 (organophosphate* or organophosphonate*).ti,ab,kf. 26 (alendronate or alendronic acid or Fosamax or Binosto or Denfos or Fosagen or Lendrate).ti,ab,kf. 27 (Densidron or Adrovance or Alenotop or Alned or Dronat or Durost or Fixopan or Forosa or Fosval or Huesobone or Ostemax or Oseolen or Arendal or Beenos or Berlex or Fosalen or Fosmin or Fostolin or Fosavance).ti,ab,kf. -

Tiludronate 1105 Osteoporosis
Strontium Ranelate/Tiludronate 1105 Osteoporosis. Strontium ranelate, given orally with calcium temic hypersensitivity reactions should be borne in mind. Hyper- Data on combination therapy are limited, but some studies sug- and vitamin D supplements, has been found to reduce the risk of calcaemia may develop with teriparatide or the acetate and it is gest that teriparatide with HRT is more effective than HRT vertebral1 and non-vertebral2 fractures in postmenopausal wom- therefore contra-indicated in patients with pre-existing hypercal- alone.9 The effect of teriparatide with the antiresorptive bisphos- en with osteoporosis. A pooled analysis of data from these 2 caemia. phonates has yet to be determined.2,6,9,10 Although there is some studies concluded that strontium ranelate reduced both vertebral suggestion that teriparatide still increases bone formation after 3 Teriparatide is contra-indicated in patients with severe renal im- 9 and non-vertebral fractures in patients aged 80 years or older. pairment and should be used with caution with those with mod- treatment with alendronate, a study in men found that, when Protection against fractures was detected within 12 months, and erate impairment. given together, alendronate impaired the anabolic effects of teri- sustained throughout 3 years of treatment. Hip fractures were paratide.11 For this reason, some consider that teriparatide be also reduced over 3 years, but this did not reach statistical signif- There have been reports of osteosarcoma in rats given teripara- started immediately after stopping bisphosphonates.10 It has been icance; the authors concluded that the analysis may not have tide and patients who may be at increased risk, including those suggested that the degree of suppression of bone turnover before been sufficiently powered in this respect. -

Date Database Search Strategy Filters Results Results After Duplicates
Date Database Search Strategy Filters Results Results after Duplicates Removed 12/12/2018 PubMed (("Patient Participation"[Mesh] OR "Patient Participation" OR “Patient Filters: 120 108 Involvement” OR “Patient Empowerment” OR “Patient Participation Rates” English OR “Patient Participation Rate” OR “Patient Activation” OR “Patient Engagement” OR "Refusal to Participate"[Mesh] OR "Refusal to Participate" OR "Self Care"[Mesh] OR "Self Care" OR "Self-Care" OR “Well-being” OR Wellbeing OR “well being” OR "Walking"[Mesh] OR Walking OR Walk OR Walked OR Ambulation OR "Gait"[Mesh] OR Gait OR Gaits OR "Mobility Limitation"[Mesh] OR "Mobility Limitation" OR Mobility OR “Mobility Limitations” OR “Ambulation Difficulty” OR “Ambulation Difficulties” OR “Difficulty Ambulation” OR “Ambulatory Difficulty” OR “Ambulatory Difficulties” OR “Difficulty Walking” OR "Dependent Ambulation"[Mesh] OR "Dependent Ambulation" OR “functional status” OR “functional state” OR "Community Participation"[Mesh] OR "Community Participation" OR “Community Involvement” OR “Community Involvements” OR “Consumer Participation” OR “Consumer Involvement” OR “Public Participation” OR “Community Action” OR “Community Actions” OR "Social Participation"[Mesh] OR "Social Participation" OR "Activities of Daily Living"[Mesh] OR "Activities of Daily Living" OR ADL OR “Daily Living Activities” OR “Daily Living Activity” OR “Chronic Limitation of Activity” OR "Quality of Life"[Mesh] OR "Quality of Life" OR “Life Quality” OR “Health- Related Quality Of Life” OR “Health Related Quality Of -

(2006.01) (84) Designated States (Unless Otherwise Indicated, For
) ( (51) International Patent Classification: ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, A61K 47/68 (2017.01) A61P 35/00 (2006.01) NZ, OM, PA, PE, PG, PH, PL, PT, QA, RO, RS, RU, RW, SA, SC, SD, SE, SG, SK, SL, ST, SV, SY, TH, TJ, TM, TN, (21) International Application Number: TR, TT, TZ, UA, UG, US, UZ, VC, VN, WS, ZA, ZM, ZW. PCT/EP2020/070149 (84) Designated States (unless otherwise indicated, for every (22) International Filing Date: kind of regional protection available) . ARIPO (BW, GH, 16 July 2020 (16.07.2020) GM, KE, LR, LS, MW, MZ, NA, RW, SD, SL, ST, SZ, TZ, (25) Filing Language: English UG, ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, RU, TJ, TM), European (AL, AT, BE, BG, CH, CY, CZ, DE, DK, (26) Publication Language: English EE, ES, FI, FR, GB, GR, HR, HU, IE, IS, IT, LT, LU, LV, (30) Priority Data: MC, MK, MT, NL, NO, PL, PT, RO, RS, SE, SI, SK, SM, 19187692.9 23 July 2019 (23.07.2019) EP TR), OAPI (BF, BJ, CF, CG, Cl, CM, GA, GN, GQ, GW, KM, ML, MR, NE, SN, TD, TG). (71) Applicants: BAYER PHARMA AKTIENGESEL- LSCHAFT [DE/DE]; Mullerstr. 178, 13353 Berlin (DE). Declarations under Rule 4.17: BAYER AKTIENGESELLSCHAFT [DE/DE]; Kaiser- — as to applicant's entitlement to apply for and be granted a Wilhelm-Allee 1, 51373 Leverkusen (DE). patent (Rule 4.17(H)) (72) Inventors: BOHNKE, Niels; Sachsische Str. 41, 10713 Published: Berlin (DE). GRIEBENOW, Nils; Kurfurstenstr. -

Phvwp Class Review Bisphosphonates and Osteonecrosis of the Jaw (Alendronic Acid, Clodronic Acid, Etidronic Acid, Ibandronic
PhVWP Class Review Bisphosphonates and osteonecrosis of the jaw (alendronic acid, clodronic acid, etidronic acid, ibandronic acid, neridronic acid, pamidronic acid, risedronic acid, tiludronic acid, zoledronic acid), SPC wording agreed by the PhVWP in February 2006 Section 4.4 Pamidronic acid and zoledronic acid: “Osteonecrosis of the jaw has been reported in patients with cancer receiving treatment regimens including bisphosphonates. Many of these patients were also receiving chemotherapy and corticosteroids. The majority of reported cases have been associated with dental procedures such as tooth extraction. Many had signs of local infection including osteomyelitis. A dental examination with appropriate preventive dentistry should be considered prior to treatment with bisphosphonates in patients with concomitant risk factors (e.g. cancer, chemotherapy, radiotherapy, corticosteroids, poor oral hygiene). While on treatment, these patients should avoid invasive dental procedures if possible. For patients who develop osteonecrosis of the jaw while on bisphosphonate therapy, dental surgery may exacerbate the condition. For patients requiring dental procedures, there are no data available to suggest whether discontinuation of bisphosphonate treatment reduces the risk of osteonecrosis of the jaw. Clinical judgement of the treating physician should guide the management plan of each patient based on individual benefit/risk assessment.” Remaining bisphosphonates: “Osteonecrosis of the jaw, generally associated with tooth extraction and/or local infection (including osteomyelits) has been reported in patients with cancer receiving treatment regimens including primarily intravenously administered bisphophonates. Many of these patients were also receiving chemotherapy and corticosteroids. Osteonecrosis of the jaw has also been reported in patients with osteoporosis receiving oral bisphophonates. A dental examination with appropriate preventive dentistry should be considered prior to treatment with bisphosphonates in patients with concomitant risk factors (e.g. -

June 2011 Circular No
7 th June 2011 Circular No. P06/2011 Dear Healthcare Professional, Re: European Medicines Agency finalises review of bisphosphonates and atypical stress fractures Bisphosphonates have been authorised in the EU for hypercalcaemia and the prevention of bone problems in patients with cancer since the early 1990s. They have also been available since the mid 1990s for the treatment of osteoporosis and Paget’s disease of the bone. Bisphosphonates include alendronic acid, clodronic acid, etidronic acid, ibandronic acid, neridronic acid, pamidronic acid, risedronic acid, tiludronic acid and zoledronic acid. They are available in the EU as tablets and as solutions for infusion under various trade names and as generic medicines2. In 2008, the CHMP’s Pharmacovigilance Working Party (PhVWP) noted that alendronic acid was associated with an increased risk of atypical fracture of the femur (thigh bone) that developed with low or no trauma. As a result, a warning was added to the product information of alendronic acid-containing medicines across Europe. The PhVWP also concluded at the time that it was not possible to rule out the possibility that the effect could be a class effect (an effect common to all bisphosphonates), and decided to keep the issue under close review. In April 2010, the PhVWP noted that further data from both the published literature and post- marketing reports were now available that suggested that atypical stress fractures of the femur may be a class effect. The working party concluded that there was a need to conduct a further review to determine if any regulatory action was necessary. Page 1 of 3 Medicines Authority 203 Level 3, Rue D'Argens, Gzira, GZR 1368 – Malta. -

A New IC-MS/MS Approach for the Analysis of Bisphosphonates in Horse Plasma
CUSTOMER APPLICATION NOTE 73058 A new IC-MS/MS approach for the analysis of bisphosphonates in horse plasma Authors Application benefits Patrice Garcia1, Sylvain Morel2, • Significant simplification and method performance improvements over Justine Pinetre1, Xavier Deruy2, current reference methods Murielle Jaubert1, Claudia P.B. • Derivatization-free protocol with fewer sample preparation steps Martins3, Isabelle Perot1, Marie- Agnès Popot1, Ludovic Bailly- • Quantification of all bisphosphonates including N-containing Chouriberry1 bisphosphonates 1GIE L.C.H. (Laboratoire des Courses Hippiques), Introduction Verrières le Buisson, France Bisphosphonates are prohibited drugs according to Article 6 of the International Agreement on Breeding, Racing and Wagering of the 2Thermo Fisher Scientific, International Federation of Horseracing Authorities (IFHA).1 These Villebon / Yvette, France compounds are used for the treatment of navicular syndrome and related 3 Thermo Fisher Scientific, diseases in horses and are divided in two groups: non-nitrogen-containing San Jose, CA, USA bisphosphonate drugs (e.g., tiludronic acid) and nitrogen-containing bisphosphonate drugs (e.g., zoledronic acid). Keywords IC-MS/MS, HPIC, TSQ Altis, Due to their very polar and strong chelating characteristics, determination ICS-6000, Dionex IonPac AS18 of bisphosphonates represents a real analytical challenge for routine 4µm Fast Column, horse doping, screening. The scientific literature reports different approaches to perform bisphosphonates bisphosphonates analysis: -
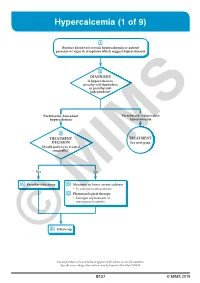
Hypercalcemia (1 of 9)
Hypercalcemia (1 of 9) 1 Routine blood test reveals hypercalcemia or patient presents w/ signs & symptoms which suggest hypercalcemia 2 DIAGNOSIS Is hypercalcemia parathyroid-dependent or parathyroid- independent? Parathyroid-dependent Parathyroid-independent hypercalcemia hypercalcemia 3 TREATMENT TREATMENT DECISION See next page Should patient be treated surgically? Yes No A Parathyroidectomy B Measures to lower serum calcium • In asymptomatic patients C Pharmacological therapy • Estrogen replacement in menopausalMIMS patients D © Follow-up Not all products are available or approved for above use in all countries. Specifi c prescribing information may be found in the latest MIMS. B127 © MIMS 2019 Hypercalcemia (2 of 9) PARATHYROID INDEPENDENT HYPERCALCEMIA 2 HYPERCALCEMIA DIAGNOSIS Determine etiology HYPERCALCEMIA VITAMIN D GRANULOMATOUS OF MALIGNANCY INTOXICATION DISEASES • Usually presents as • eg Sarcoidosis severe hypercalcemia B Measures to lower serum calcium B Measures to lower serum calcium C Pharmacological therapy C Pharmacological therapy • Loop diuretics • Corticosteroid hormones • Bisphosphonates • Calcitonin • Corticosteroid hormones 1 HYPERCALCEMIA • Normal serum Ca level: 8-10 mg/dL (2-2.5 mmol/L) • Hypercalcemia: Serum Ca >10.5 mg/dL (>2.5 mmol/L) - Use total serum Ca level corrected for albumin concentration, by adding 0.8 mg/dL to the total serum Ca level for every 1 g/dL drop in serum albumin <4 g/dL Signs & Symptoms Mild Hypercalcemia • Usually asymptomatic More Severe Hypercalcemia • Symptoms usually become more -

NETSCC, HTA 1 March 2012
NETSCC, HTA 1 March 2012 The Health Technology Assessment programme is managed by NETSCC, HTA as part of the NIHR Evaluation, Trials and Studies Coordinating Centre at the University of Southampton. Alpha House, University of Southampton Science Park tel: +44(0)23 8059 5586 Southampton SO16 7NS fax: +44(0)23 8059 5639 Suggest a topic for research via our online form at www.hta.ac.uk/suggest email: [email protected] www.hta.ac.uk Bone turnover markers Technology Assessment Report commissioned by the NIHR HTA – Protocol 1. Title of the project: Bone turnover markers for monitoring the response to osteoporosis treatment: the secondary prevention of fractures, and primary prevention of fractures in high risk groups. 2. Name of TAR team and ‘lead’ Centre for Reviews and Dissemination/Centre for Health Economics Technology Assessment Group, University of York. Dawn Craig, Research Fellow, Centre for Reviews and Dissemination, University of York, Heslington, York YO10 5DD Tel: (01904) 321052 Fax: (01904) 321041 Email: [email protected] 3. Plain English Summary Bone turnover is the process of bone breakdown and renewal; under normal circumstances, these two parts of the process are balanced to ensure a constant bone density. If this balance is not maintained, bone structure, mass and strength may be altered. Osteoporosis is a disease in which bone mineral density is reduced as a result of increased bone breakdown and/or decreased bone renewal. Osteoporosis is thought to be responsible for 200,000 fractures every year, with broken wrists, hips and spinal bones the most common. The measurement of products in the blood or urine as a result of either bone breakdown or formation can be used to monitor bone turnover. -

Adhesive Preparation
(19) & (11) EP 2 062 584 A1 (12) EUROPEAN PATENT APPLICATION published in accordance with Art. 153(4) EPC (43) Date of publication: (51) Int Cl.: 27.05.2009 Bulletin 2009/22 A61K 31/663 (2006.01) A61K 9/70 (2006.01) A61K 47/06 (2006.01) A61K 47/08 (2006.01) (2006.01) (2006.01) (21) Application number: 07807007.5 A61K 47/10 A61K 47/14 A61K 47/32 (2006.01) A61P 1/02 (2006.01) (2006.01) (2006.01) (22) Date of filing: 10.09.2007 A61P 3/14 A61P 19/00 A61P 19/02 (2006.01) A61P 19/10 (2006.01) A61P 29/00 (2006.01) A61P 35/00 (2006.01) A61P 35/02 (2006.01) A61P 35/04 (2006.01) (86) International application number: PCT/JP2007/067597 (87) International publication number: WO 2008/032678 (20.03.2008 Gazette 2008/12) (84) Designated Contracting States: • HAYASHI, Noriyuki AT BE BG CH CY CZ DE DK EE ES FI FR GB GR Imizu-shi HU IE IS IT LI LT LU LV MC MT NL PL PT RO SE Toyama 939-0351 (JP) SI SK TR • SAKAI, Yoshiki Designated Extension States: Mishima-gun AL BA HR MK RS Osaka 618-8585 (JP) (30) Priority: 11.09.2006 JP 2006245965 (74) Representative: Keller, Günter et al Lederer & Keller (71) Applicant: Kyukyu Pharmaceutical Co., Ltd. Patentanwälte Tokyo 103-0023 (JP) Prinzregentenstrasse 16 80538 München (DE) (72) Inventors: • YAMAZAKI, Yuuhiro Imizu-shi Toyama 939-0351 (JP) (54) ADHESIVE PREPARATION (57) The present invention provides an adhesive of either of the bisphosphonic acid derivative or the salt, preparation having a plaster layer disposed on a support, a solubilizer for the active ingredient, propylene glycol, a the adhesive preparation comprising at least one active hydrogenated terpene resin, an adhesive base, and a ingredient selected from the group consisting of a bi- softening agent in the plaster layer. -

Patent Application Publication ( 10 ) Pub . No . : US 2019 / 0192440 A1
US 20190192440A1 (19 ) United States (12 ) Patent Application Publication ( 10) Pub . No. : US 2019 /0192440 A1 LI (43 ) Pub . Date : Jun . 27 , 2019 ( 54 ) ORAL DRUG DOSAGE FORM COMPRISING Publication Classification DRUG IN THE FORM OF NANOPARTICLES (51 ) Int . CI. A61K 9 / 20 (2006 .01 ) ( 71 ) Applicant: Triastek , Inc. , Nanjing ( CN ) A61K 9 /00 ( 2006 . 01) A61K 31/ 192 ( 2006 .01 ) (72 ) Inventor : Xiaoling LI , Dublin , CA (US ) A61K 9 / 24 ( 2006 .01 ) ( 52 ) U . S . CI. ( 21 ) Appl. No. : 16 /289 ,499 CPC . .. .. A61K 9 /2031 (2013 . 01 ) ; A61K 9 /0065 ( 22 ) Filed : Feb . 28 , 2019 (2013 .01 ) ; A61K 9 / 209 ( 2013 .01 ) ; A61K 9 /2027 ( 2013 .01 ) ; A61K 31/ 192 ( 2013. 01 ) ; Related U . S . Application Data A61K 9 /2072 ( 2013 .01 ) (63 ) Continuation of application No. 16 /028 ,305 , filed on Jul. 5 , 2018 , now Pat . No . 10 , 258 ,575 , which is a (57 ) ABSTRACT continuation of application No . 15 / 173 ,596 , filed on The present disclosure provides a stable solid pharmaceuti Jun . 3 , 2016 . cal dosage form for oral administration . The dosage form (60 ) Provisional application No . 62 /313 ,092 , filed on Mar. includes a substrate that forms at least one compartment and 24 , 2016 , provisional application No . 62 / 296 , 087 , a drug content loaded into the compartment. The dosage filed on Feb . 17 , 2016 , provisional application No . form is so designed that the active pharmaceutical ingredient 62 / 170, 645 , filed on Jun . 3 , 2015 . of the drug content is released in a controlled manner. Patent Application Publication Jun . 27 , 2019 Sheet 1 of 20 US 2019 /0192440 A1 FIG .