Organization and Development of Stock Enhancement in Japan Sugaya, Takuma Date Published: 2006
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Disease List for Aquaculture Health Certificate
Quarantine Standard for Designated Species of Imported/Exported Aquatic Animals [Attached Table] 4. Listed Diseases & Quarantine Standard for Designated Species Listed disease designated species standard Common name Disease Pathogen 1. Epizootic haematopoietic Epizootic Perca fluviatilis Redfin perch necrosis(EHN) haematopoietic Oncorhynchus mykiss Rainbow trout necrosis virus(EHNV) Macquaria australasica Macquarie perch Bidyanus bidyanus Silver perch Gambusia affinis Mosquito fish Galaxias olidus Mountain galaxias Negative Maccullochella peelii Murray cod Salmo salar Atlantic salmon Ameirus melas Black bullhead Esox lucius Pike 2. Spring viraemia of Spring viraemia of Cyprinus carpio Common carp carp, (SVC) carp virus(SVCV) Grass carp, Ctenopharyngodon idella white amur Hypophthalmichthys molitrix Silver carp Hypophthalmichthys nobilis Bighead carp Carassius carassius Crucian carp Carassius auratus Goldfish Tinca tinca Tench Sheatfish, Silurus glanis European catfish, wels Negative Leuciscus idus Orfe Rutilus rutilus Roach Danio rerio Zebrafish Esox lucius Northern pike Poecilia reticulata Guppy Lepomis gibbosus Pumpkinseed Oncorhynchus mykiss Rainbow trout Abramis brama Freshwater bream Notemigonus cysoleucas Golden shiner 3.Viral haemorrhagic Viral haemorrhagic Oncorhynchus spp. Pacific salmon septicaemia(VHS) septicaemia Oncorhynchus mykiss Rainbow trout virus(VHSV) Gadus macrocephalus Pacific cod Aulorhynchus flavidus Tubesnout Cymatogaster aggregata Shiner perch Ammodytes hexapterus Pacific sandlance Merluccius productus Pacific -

Evaluation of Genes Involved in Norway Lobster (Nephrops Norvegicus) Female Sexual Maturation Using Transcriptomic Analysis
Hydrobiologia https://doi.org/10.1007/s10750-018-3521-3 CRUSTACEAN GENOMICS Evaluation of genes involved in Norway lobster (Nephrops norvegicus) female sexual maturation using transcriptomic analysis Guiomar Rotllant . Tuan Viet Nguyen . David Hurwood . Valerio Sbragaglia . Tomer Ventura . Joan B. Company . Silvia Joly . Abigail Elizur . Peter B. Mather Received: 6 October 2017 / Revised: 17 January 2018 / Accepted: 20 January 2018 Ó Springer International Publishing AG, part of Springer Nature 2018 Abstract The Norway lobster Nephrops norvegicus technology applied to multiple tissues. Ovarian mat- is the most important commercial crustacean species uration-related differential expression patterns were in Europe. Recent decline in wild captures and a observed for 4362 transcripts in ovary, hepatopan- reduction in total abundance and size at first matura- creas, eyestalk, brain, and thoracic ganglia in N. tion indicate that the species is overexploited. Increas- norvegicus. Transcripts detected in the study include ing knowledge of its reproduction, specifically at the vitellogenin, crustacean hyperglycaemic hormone, molecular level will be mandatory to improving retinoid X receptor, heat shock protein 90 and proteins fisheries management. The current study investigated encoding lipid and carbohydrate metabolizing differences between immature and mature N. norvegi- enzymes. From the study, data were collected that cus females using Next Generation Sequencing can prove valuable in developing more comprehensive knowledge of the reproductive system in this lobster species during the ovarian maturation process. Addi- Electronic supplementary material The online version of this article (https://doi.org/10.1007/s10750-018-3521-3) con- tional studies will be required, however, to identify tains supplementary material, which is available to authorized potential novel genes and to develop a molecular users. -

A C-Type Lectin Highly Expressed in Portunus Trituberculatus Intestine Functions in AMP Regulation and Prophenoloxidase Activation
antibiotics Article A C-Type Lectin Highly Expressed in Portunus trituberculatus Intestine Functions in AMP Regulation and Prophenoloxidase Activation Yuan Liu 1,2,3,4,*, Yue Su 1,4, Ao Zhang 1 and Zhaoxia Cui 5 1 CAS and Shandong Province Key Laboratory of Experimental Marine Biology, Institute of Oceanology, Chinese Academy of Sciences, Qingdao 266071, China; [email protected] (Y.S.); [email protected] (A.Z.) 2 Laboratory for Marine Biology and Biotechnology, Qingdao National Laboratory for Marine Science and Technology, Qingdao 266071, China 3 Center for Ocean Mega-Science, Chinese Academy of Sciences, Qingdao 266071, China 4 University of Chinese Academy of Sciences, Beijing 100049, China 5 School of Marine Science, Ningbo University, Ningbo 315211, China; [email protected] * Correspondence: [email protected]; Tel.: +86-532-8289-8637 Abstract: A C-type lectin (PtCLec2) from Portunus trituberculatus was identified for characterization of its role in defense and innate immunity. PtCLec2 contains a single carbohydrate-recognition domain (CRD) with a conserved QPD motif, which was predicted to have galactose specificity. The mRNA expression of PtCLec2 was predominantly detected in intestine and increased rapidly and significantly upon pathogen challenge. The recombinant PtCLec2 (rPtCLec2) could bind various microorganisms and PAMPs with weak binding ability to yeast and PGN. It agglutinated the tested Gram-negative bacteria (Vibrio alginolyticus and Pseudomonas aeruginosa), Gram-positive bacteria Citation: Liu, Y.; Su, Y.; Zhang, A.; (Staphylococcus aureus and Micrococcus luteus), and rabbit erythrocytes in the presence of exogenous Cui, Z. A C-Type Lectin Highly Ca2+, and these agglutination activities were suppressed by LPS, D-galactose, and D-mannose. -

(Jasus Edwardsii Hutton, 1875) Larvae
Environmental Physiology of Cultured Early-Stage Southern Rock Lobster (Jasus edwardsii Hutton, 1875) Larvae Michel Francois Marie Bermudes Submitted in fulfilment of the requirements for the degree of Doctor Of Philosophy University of Tasmania November 2002 Declarations This thesis contains no material which has been accepted for a degree or diploma by the University or any other institution, except by way of background information in duly acknowledged in the thesis, and to the best of the candidate's knowledge and belief, no material previously published or written by another person except where due acknowledgment is made in the text of the thesis. Michel Francois Marie Bermudes This thesis may be available for loan and limited copying in accordance with the Copyright Act 1968. Michel Francois Marie Bermudes Abstract The aim of this project was to define more clearly the culture conditions for the propagation of the southern rock lobster (Jasus echvardsii) in relation to environmental bioenergetic constraints. The effects of temperature and photoperiod on the first three stages of development were first studied in small-scale culture experiments. Larvae reared at 18°C developed faster and reached a larger size at stage IV than larvae cultured at 14°C. Development through stage II was shorter under continuous light. However, the pattern of response to photoperiod shifted at stage III when growth was highest in all the light/dark phase treatments than under continuous light. The influence of temperature and light intensity in early-stage larvae was further investigated through behavioural and physiological studies. Results obtained in stages I, II and III larvae indicated an energetic imbalance at high temperature (-22°C). -

Lawrence Berkeley National Laboratory Recent Work
Lawrence Berkeley National Laboratory Recent Work Title Genetic markers in blue crabs (Callinectes sapidus) II: Complete mitochondrial genome sequence and characterization of genetic variation Permalink https://escholarship.org/uc/item/2cv090m0 Journal Journal of Experimental Marine Biology and Ecology, 319(1/2/2008) Authors Place, Allen R. Feng, Xiaojun Steven, Colin R. et al. Publication Date 2004-08-17 eScholarship.org Powered by the California Digital Library University of California Genetic markers in blue crabs (Callinectes sapidus) II: Complete Mitochondrial Genome Sequence and Characterization of Genetic Variation Allen R. Place1, Xiaojun Feng1, Colin R. Steven1, H. Matthew Fourcade2, and Jeffrey L. Boore2 1Center of Marine Biotechnology, University of Maryland Biotechnology Institute, 701 East Pratt Street, Baltimore, Maryland 21202 2DOE Joint Genome and Lawrence Berkeley National Lab, 2800 Mitchell Drive, Walnut Creek, CA 94598 Corresponding author: Allen Place, Tel: (410) 234-8828 Fax: (410) 234-8896 E-mail address: [email protected] Keywords: mitochondrial genome, blue crab, nucleotide variation, long PCR, shotgun sequencing, gene rearrangement Abbreviations: atp6, atp8, ATPase subunits 6 and 8; cox1, cox2, cox3, cytochrome oxidase subunits 1-3; CR, control region; cob, cytochrome b; mtDNA, mitochondrial DNA; nad1-6, 4L, NADH dehydrogenase subunits, 1-6 and 4L; PCR, polymerase chain reaction; RCA, rolling circle amplification; rrnL, rrnS, large and small subunit ribosomal RNA; trnX, tRNA genes, where X stands for the one letter amino acid code, with tRNAs for L and S differentiated by anticodon in parentheses. Abstract Given the commercial and ecological importance of the dwindling Chesapeake Bay blue crab (Callinectes sapidus) fishery there is a surprising scarcity of information concerning the molecular ecology of this species. -

Blue Crab-Japanese Less
A Maryland Sea Grant Report Japanese Hatchery-based Stock Enhancement: Lessons for the Chesapeake Bay Blue Crab David H. Secor, Anson H. Hines and Allen R. Place Japanese Hatchery-based Stock Enhancement: Lessons for the Chesapeake Bay Blue Crab David H. Secor Associate Professor, Fisheries Scientist Chesapeake Biological Laboratory University of Maryland Center for Environmental Science Solomons, Maryland Anson H. Hines Assistant Director, Marine Ecologist Smithsonian Environmental Research Center Edgewater, Maryland Allen R. Place Professor, Biochemist Center of Marine Biotechnology University of Maryland Biotechnology Institute Baltimore, Maryland Maryland Sea Grant College Park, Maryland This publication was prepared by Maryland Sea Grant. The Maryland Sea Grant College, a university-based partnership with the National Oceanic and Atmospheric Administration, is a service organization in the state of Maryland administered by the University System of Maryland; its mission is to conduct a program of research, education and outreach to use and conserve coastal and marine resources for a sustainable economy and environment in Maryland, in the Mid-Atlantic region and in the nation. September 2002 Maryland Sea Grant Publication Number UM-SG-TS-2002-02 Cover photo from Japan Sea Farming Association For more information, write: Maryland Sea Grant College 4321 Hartwick Road, Suite 300 University of Maryland College Park, Maryland 20740 or visit the web: www.mdsg.umd.edu/ This publication is made possible by grant NA86RG0037 awarded by the National Oceanic and Atmospheric Administration to the University of Maryland Sea Grant College Program. The University of Maryland is an equal opportunity employer. Contents Acknowledgments . .3 Preface . .5 Report Highlights . .7 Executive Summary . -

Molecular Phylogeny of the Western Atlantic Species of the Genus Portunus (Crustacea, Brachyura, Portunidae)
Blackwell Publishing LtdOxford, UKZOJZoological Journal of the Linnean Society0024-4082The Lin- nean Society of London, 2007? 2007 1501 211220 Original Article PHYLOGENY OF PORTUNUS FROM ATLANTICF. L. MANTELATTO ET AL. Zoological Journal of the Linnean Society, 2007, 150, 211–220. With 3 figures Molecular phylogeny of the western Atlantic species of the genus Portunus (Crustacea, Brachyura, Portunidae) FERNANDO L. MANTELATTO1*, RAFAEL ROBLES2 and DARRYL L. FELDER2 1Laboratory of Bioecology and Crustacean Systematics, Department of Biology, FFCLRP, University of São Paulo (USP), Ave. Bandeirantes, 3900, CEP 14040-901, Ribeirão Preto, SP (Brazil) 2Department of Biology, Laboratory for Crustacean Research, University of Louisiana at Lafayette, Lafayette, LA 70504-2451, USA Received March 2004; accepted for publication November 2006 The genus Portunus encompasses a comparatively large number of species distributed worldwide in temperate to tropical waters. Although much has been reported about the biology of selected species, taxonomic identification of several species is problematic on the basis of strictly adult morphology. Relationships among species of the genus are also poorly understood, and systematic review of the group is long overdue. Prior to the present study, there had been no comprehensive attempt to resolve taxonomic questions or determine evolutionary relationships within this genus on the basis of molecular genetics. Phylogenetic relationships among 14 putative species of Portunus from the Gulf of Mexico and other waters of the western Atlantic were examined using 16S sequences of the rRNA gene. The result- ant molecularly based phylogeny disagrees in several respects with current morphologically based classification of Portunus from this geographical region. Of the 14 species generally recognized, only 12 appear to be valid. -

Proceedings of the 40Th U.S.-Japan Aquaculture Panel Symposium
Hatchery Technology for High Quality Juvenile Production Proceedings of the 40th U.S.-Japan Aquaculture Panel Symposium University of Hawaii East West Center Honolulu, Hawaii October 22-23 2012 U.S. DEPARTMENT OF COMMERCE National Oceanic and Atmospheric Administration National Marine Fisheries Service NOAA Technical Memorandum NMFS-F/SPO-136 Hatchery Technology for High Quality Juvenile Production Proceedings of the 40th U.S.-Japan Aquaculture Panel Symposium University of Hawaii East West Center Honolulu, Hawaii October 22-23 2012 Mike Rust1, Paul Olin2, April Bagwill3, and Marie Fujitani3, editors 1Northwest Fisheries Science Center 2725 Montlake Boulevard East Seattle, Washington 98112 2California Sea Grant UCSD / Scripps Institution of Oceanography 133 Aviation Blvd., Suite 109 Santa Rosa CA 95403 3NOAA National Marine Fisheries Service 1315 East-West Highway Silver Spring, MD 20910 NOAA Technical Memorandum NMFS-F/SPO-136 December 2013 U.S. Department of Commerce Penny Pritzker, Secretary of Commerce National Oceanic and Atmospheric Administration Dr. Kathryn Sullivan, (Acting) NOAA Administrator National Marine Fisheries Service Samuel D. Rauch III, (Acting) Assistant Administrator for Fisheries SUGGESTED CITATION: Rust, M., P. Olin, A. Bagwill and M. Fujitani (editors). 2013. Hatchery Technology for High Quality Juvenile Production: Proceedings of the 40th U.S.-Japan Aquaculture Panel Symposium, Honolulu, Hawaii, October 22-23, 2012. U.S. Dept. Commerce, NOAA Tech. Memo. NMFS-F/SPO-136. A COPY OF THIS REPORT MAY BE OBTAINED FROM: Northwest Fisheries Science Center 2725 Montlake Boulevard East Seattle, Washington 98112 OR ONLINE AT: http://spo.nmfs.noaa.gov/tm/ Reference throughout this document to trade names does not imply endorsement by the National Marine Fisheries Service, NOAA. -
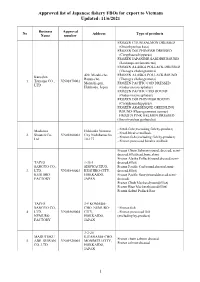
Approved List of Japanese Fishery Fbos for Export to Vietnam Updated: 11/6/2021
Approved list of Japanese fishery FBOs for export to Vietnam Updated: 11/6/2021 Business Approval No Address Type of products Name number FROZEN CHUM SALMON DRESSED (Oncorhynchus keta) FROZEN DOLPHINFISH DRESSED (Coryphaena hippurus) FROZEN JAPANESE SARDINE ROUND (Sardinops melanostictus) FROZEN ALASKA POLLACK DRESSED (Theragra chalcogramma) 420, Misaki-cho, FROZEN ALASKA POLLACK ROUND Kaneshin Rausu-cho, (Theragra chalcogramma) 1. Tsuyama CO., VN01870001 Menashi-gun, FROZEN PACIFIC COD DRESSED LTD Hokkaido, Japan (Gadus macrocephalus) FROZEN PACIFIC COD ROUND (Gadus macrocephalus) FROZEN DOLPHIN FISH ROUND (Coryphaena hippurus) FROZEN ARABESQUE GREENLING ROUND (Pleurogrammus azonus) FROZEN PINK SALMON DRESSED (Oncorhynchus gorbuscha) - Fresh fish (excluding fish by-product) Maekawa Hokkaido Nemuro - Fresh bivalve mollusk. 2. Shouten Co., VN01860002 City Nishihamacho - Frozen fish (excluding fish by-product) Ltd 10-177 - Frozen processed bivalve mollusk Frozen Chum Salmon (round, dressed, semi- dressed,fillet,head,bone,skin) Frozen Alaska Pollack(round,dressed,semi- TAIYO 1-35-1 dressed,fillet) SANGYO CO., SHOWACHUO, Frozen Pacific Cod(round,dressed,semi- 3. LTD. VN01840003 KUSHIRO-CITY, dressed,fillet) KUSHIRO HOKKAIDO, Frozen Pacific Saury(round,dressed,semi- FACTORY JAPAN dressed) Frozen Chub Mackerel(round,fillet) Frozen Blue Mackerel(round,fillet) Frozen Salted Pollack Roe TAIYO 3-9 KOMABA- SANGYO CO., CHO, NEMURO- - Frozen fish 4. LTD. VN01860004 CITY, - Frozen processed fish NEMURO HOKKAIDO, (excluding by-product) FACTORY JAPAN -

Toxic Responses of Swimming Crab (Portunus Trituberculatus) Larvae Exposed to Environmentally Realistic Concentrations of Oxytetracycline
Chemosphere 173 (2017) 563e571 Contents lists available at ScienceDirect Chemosphere journal homepage: www.elsevier.com/locate/chemosphere Toxic responses of swimming crab (Portunus trituberculatus) larvae exposed to environmentally realistic concentrations of oxytetracycline * Xianyun Ren a, b, Zhuqing Wang a, b, c, Baoquan Gao a, b, Ping Liu a, b, Jian Li a, b, a Key Laboratory for Sustainable Utilization of Marine Fisheries Resources, Ministry of Agriculture, Yellow Sea Fisheries Research Institute, Chinese Academy of Fishery Sciences, Qingdao, PR China b Function Laboratory for Marine Fisheries Science and Food Production Processes, Qingdao National Laboratory for Marine Science and Technology, Qingdao, PR China c College of Fisheries and Life Science, Shanghai Ocean University, Shanghai, PR China highlights We investigated the effects of OTC on Portunus trituberculatus larvae. Exposure to OTC suppressed the antioxidant system of P. trituberculatus larvae. OTC affected genes and enzymes related to detoxification. Exposure to OTC induced biomolecule damage in P. trituberculatus larvae. article info abstract Article history: Oxytetracycline (OTC) is the most commonly used antibiotics for bacterial treatment in crustacean Received 31 August 2016 farming in China, and because of their intensive use, the potential harmful effects on aquatic organisms Received in revised form are of great concern. The aim of this study was to investigate the effects of oxytetracycline (OTC) on the 12 January 2017 antioxidant system, detoxification progress, and biomolecule damage in Portunus trituberculatus larvae. Accepted 13 January 2017 In this study, larvae that belonged to four zoeal stages were exposed to four different concentrations of OTC (0, 0.3, 3, and 30 mg/L) for 3 days. -

Chinese Red Swimming Crab (Portunus Haanii) Fishery Improvement Project (FIP) in Dongshan, China (August-December 2018)
Chinese Red Swimming Crab (Portunus haanii) Fishery Improvement Project (FIP) in Dongshan, China (August-December 2018) Prepared by Min Liu & Bai-an Lin Fish Biology Laboratory College of Ocean and Earth Sciences, Xiamen University March 2019 Contents 1. Introduction........................................................................................................ 5 2. Materials and Methods ...................................................................................... 6 2.1. Study site and survey frequency .................................................................... 6 2.2. Sample collection .......................................................................................... 7 2.3. Species identification................................................................................... 10 2.4. Sample measurement ................................................................................... 11 2.5. Interviews.................................................................................................... 13 2.6. Estimation of annual capture volume of Portunus haanii ............................. 15 3. Results .............................................................................................................. 15 3.1. Species diversity.......................................................................................... 15 3.1.1. Species composition .............................................................................. 15 3.1.2. ETP species ......................................................................................... -
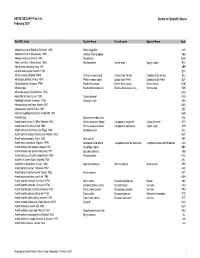
ASFIS ISSCAAP Fish List February 2007 Sorted on Scientific Name
ASFIS ISSCAAP Fish List Sorted on Scientific Name February 2007 Scientific name English Name French name Spanish Name Code Abalistes stellaris (Bloch & Schneider 1801) Starry triggerfish AJS Abbottina rivularis (Basilewsky 1855) Chinese false gudgeon ABB Ablabys binotatus (Peters 1855) Redskinfish ABW Ablennes hians (Valenciennes 1846) Flat needlefish Orphie plate Agujón sable BAF Aborichthys elongatus Hora 1921 ABE Abralia andamanika Goodrich 1898 BLK Abralia veranyi (Rüppell 1844) Verany's enope squid Encornet de Verany Enoploluria de Verany BLJ Abraliopsis pfefferi (Verany 1837) Pfeffer's enope squid Encornet de Pfeffer Enoploluria de Pfeffer BJF Abramis brama (Linnaeus 1758) Freshwater bream Brème d'eau douce Brema común FBM Abramis spp Freshwater breams nei Brèmes d'eau douce nca Bremas nep FBR Abramites eques (Steindachner 1878) ABQ Abudefduf luridus (Cuvier 1830) Canary damsel AUU Abudefduf saxatilis (Linnaeus 1758) Sergeant-major ABU Abyssobrotula galatheae Nielsen 1977 OAG Abyssocottus elochini Taliev 1955 AEZ Abythites lepidogenys (Smith & Radcliffe 1913) AHD Acanella spp Branched bamboo coral KQL Acanthacaris caeca (A. Milne Edwards 1881) Atlantic deep-sea lobster Langoustine arganelle Cigala de fondo NTK Acanthacaris tenuimana Bate 1888 Prickly deep-sea lobster Langoustine spinuleuse Cigala raspa NHI Acanthalburnus microlepis (De Filippi 1861) Blackbrow bleak AHL Acanthaphritis barbata (Okamura & Kishida 1963) NHT Acantharchus pomotis (Baird 1855) Mud sunfish AKP Acanthaxius caespitosa (Squires 1979) Deepwater mud lobster Langouste