SI-Brochure-9-Concise-EN.Pdf
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
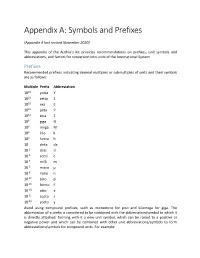
Appendix A: Symbols and Prefixes
Appendix A: Symbols and Prefixes (Appendix A last revised November 2020) This appendix of the Author's Kit provides recommendations on prefixes, unit symbols and abbreviations, and factors for conversion into units of the International System. Prefixes Recommended prefixes indicating decimal multiples or submultiples of units and their symbols are as follows: Multiple Prefix Abbreviation 1024 yotta Y 1021 zetta Z 1018 exa E 1015 peta P 1012 tera T 109 giga G 106 mega M 103 kilo k 102 hecto h 10 deka da 10-1 deci d 10-2 centi c 10-3 milli m 10-6 micro μ 10-9 nano n 10-12 pico p 10-15 femto f 10-18 atto a 10-21 zepto z 10-24 yocto y Avoid using compound prefixes, such as micromicro for pico and kilomega for giga. The abbreviation of a prefix is considered to be combined with the abbreviation/symbol to which it is directly attached, forming with it a new unit symbol, which can be raised to a positive or negative power and which can be combined with other unit abbreviations/symbols to form abbreviations/symbols for compound units. For example: 1 cm3 = (10-2 m)3 = 10-6 m3 1 μs-1 = (10-6 s)-1 = 106 s-1 1 mm2/s = (10-3 m)2/s = 10-6 m2/s Abbreviations and Symbols Whenever possible, avoid using abbreviations and symbols in paragraph text; however, when it is deemed necessary to use such, define all but the most common at first use. The following is a recommended list of abbreviations/symbols for some important units. -

New Realization of the Ohm and Farad Using the NBS Calculable Capacitor
IEEE TRANSACTIONS ON INSTRUMENTATION AND MEASUREMENT, VOL. 38, NO. 2, APRIL 1989 249 New Realization of the Ohm and Farad Using the NBS Calculable Capacitor JOHN Q. SHIELDS, RONALD F. DZIUBA, MEMBER, IEEE,AND HOWARD P. LAYER Abstrucl-Results of a new realization of the ohm and farad using part of the NBS effort. This paper describes our measure- the NBS calculable capacitor and associated apparatus are reported. ments and gives our latest results. The results show that both the NBS representation of the ohm and the NBS representation of the farad are changing with time, cNBSat the rate of -0.054 ppmlyear and FNssat the rate of 0.010 ppm/year. The 11. AC MEASUREMENTS realization of the ohm is of particular significance at this time because The measurement sequence used in the 1974 ohm and of its role in assigning an SI value to the quantized Hall resistance. The farad determinations [2] has been retained in the present estimated uncertainty of the ohm realization is 0.022 ppm (lo) while the estimated uncertainty of the farad realization is 0.014 ppm (la). NBS measurements. A 0.5-pF calculable cross-capacitor is used to measure a transportable 10-pF reference capac- itor which is carried to the laboratory containing the NBS I. INTRODUCTION bank of 10-pF fused silica reference capacitors. A 10: 1 HE NBS REPRESENTATION of the ohm is bridge is used in two stages to measure two 1000-pF ca- T based on the mean resistance of five Thomas-type pacitors which are in turn used as two arms of a special wire-wound resistors maintained in a 25°C oil bath at NBS frequency-dependent bridge for measuring two 100-kQ Gaithersburg, MD. -

Standard Conversion Factors
Standard conversion factors 7 1 tonne of oil equivalent (toe) = 10 kilocalories = 396.83 therms = 41.868 GJ = 11,630 kWh 1 therm = 100,000 British thermal units (Btu) The following prefixes are used for multiples of joules, watts and watt hours: kilo (k) = 1,000 or 103 mega (M) = 1,000,000 or 106 giga (G) = 1,000,000,000 or 109 tera (T) = 1,000,000,000,000 or 1012 peta (P) = 1,000,000,000,000,000 or 1015 WEIGHT 1 kilogramme (kg) = 2.2046 pounds (lb) VOLUME 1 cubic metre (cu m) = 35.31 cu ft 1 pound (lb) = 0.4536 kg 1 cubic foot (cu ft) = 0.02832 cu m 1 tonne (t) = 1,000 kg 1 litre = 0.22 Imperial = 0.9842 long ton gallon (UK gal.) = 1.102 short ton (sh tn) 1 UK gallon = 8 UK pints 1 Statute or long ton = 2,240 lb = 1.201 U.S. gallons (US gal) = 1.016 t = 4.54609 litres = 1.120 sh tn 1 barrel = 159.0 litres = 34.97 UK gal = 42 US gal LENGTH 1 mile = 1.6093 kilometres 1 kilometre (km) = 0.62137 miles TEMPERATURE 1 scale degree Celsius (C) = 1.8 scale degrees Fahrenheit (F) For conversion of temperatures: °C = 5/9 (°F - 32); °F = 9/5 °C + 32 Average conversion factors for petroleum Imperial Litres Imperial Litres gallons per gallons per per tonne tonne per tonne tonne Crude oil: Gas/diesel oil: Indigenous Gas oil 257 1,167 Imported Marine diesel oil 253 1,150 Average of refining throughput Fuel oil: Ethane All grades 222 1,021 Propane Light fuel oil: Butane 1% or less sulphur 1,071 Naphtha (l.d.f.) >1% sulphur 232 1,071 Medium fuel oil: Aviation gasoline 1% or less sulphur 237 1,079 >1% sulphur 1,028 Motor -

Units and Conversions
Units and Conversions This unit of the Metrology Fundamentals series was developed by the Mitutoyo Institute of Metrology, the educational department within Mitutoyo America Corporation. The Mitutoyo Institute of Metrology provides educational courses and free on-demand resources across a wide variety of measurement related topics including basic inspection techniques, principles of dimensional metrology, calibration methods, and GD&T. For more information on the educational opportunities available from Mitutoyo America Corporation, visit us at www.mitutoyo.com/education. This technical bulletin addresses an important aspect of the language of measurement – the units used when reporting or discussing measured values. The dimensioning and tolerancing practices used on engineering drawings and related product specifications use either decimal inch (in) or millimeter (mm) units. Dimensional measurements are therefore usually reported in either of these units, but there are a number of variations and conversions that must be understood. Measurement accuracy, equipment specifications, measured deviations, and errors are typically very small numbers, and therefore a more practical spoken language of units has grown out of manufacturing and precision measurement practice. Metric System In the metric system (SI or International System of Units), the fundamental unit of length is the meter (m). Engineering drawings and measurement systems use the millimeter (mm), which is one thousandths of a meter (1 mm = 0.001 m). In general practice, however, the common spoken unit is the “micron”, which is slang for the micrometer (m), one millionth of a meter (1 m = 0.001 mm = 0.000001 m). In more rare cases, the nanometer (nm) is used, which is one billionth of a meter. -

3 Litre User's Manual
™ 3 Litre User’s Manual Please visit www.drewandcole.com for video instructions and cooking demonstrations. 1 IMPORTANT SAFETY INFORMATION IMPORTANT SAFETY INFORMATION BEFORE YOU GET STARTED, PLEASE READ THE FOLLOWING IMPORTANT SAFETY INFORMATION, ALONG WITH THE MANUAL ENCLOSED AND KEEP PRESSURE RELEASE METHODS BOTH FOR FUTURE REFERENCE. WARNING YOU ARE WORKING WITH • When the programme is finished and you wish to commence pressure release press the “Cancel” button to cancel HOT LIQUIDS. YOU MUST READ THIS BEFORE USE. the Keep Warm function. • When releasing the pressure valve, always use tongs and please wear oven gloves to turn the pressure valve to the open position. This will protect against hot steam. The valve will lift up slightly and steam will release. The lid won’t BEFORE COOKING open until the steam has vented and pressure has released. • When opening the lid food will be hot, please always wear oven gloves and an apron to protect against any • ALWAYS ensure the INNER POT is in place before cooking. splashing of the hot food. • Food with skins (e.g sausages, chicken and fruit) MUST be pierced before cooking. Not piercing QUICK RELEASE SLOW RELEASE the skin may result in the food expanding and may cause splashing of hot food after the lid is Recommended for: Recommended for: released. - Quick cooking recipes and steaming, including - Food with skins (e.g sausages, chicken and fruit) and vegetables and seafood. foods with large liquid volume or high starch content • Do not overfill the inner pot. (such as porridge, soup, pasta, rice, fruit and grains, and When the Keep Warm function has been also delicate foods such as meats and potato) can trap air cancelled, move the pressure release valve to and cause the food to foam and expand which may cause To Max the open position and only attempt to open lid MAX MAX splashing of hot food after the lid is removed. -

Consultative Committee for Amount of Substance; Metrology in Chemistry and Biology CCQM Working Group on Isotope Ratios (IRWG) S
Consultative Committee for Amount of Substance; Metrology in Chemistry and Biology CCQM Working Group on Isotope Ratios (IRWG) Strategy for Rolling Programme Development (2021-2030) 1. EXECUTIVE SUMMARY In April 2017, the Consultative Committee for Amount of Substance; Metrology in Chemistry and Biology (CCQM) established a task group to study the metrological state of isotope ratio measurements and to formulate recommendations to the Consultative Committee (CC) regarding potential engagement in this field. In April 2018 the Isotope Ratio Working Group (IRWG) was established by the CCQM based on the recommendation of the task group. The main focus of the IRWG is on the stable isotope ratio measurement science activities needed to improve measurement comparability to advance the science of isotope ratio measurement among National Metrology Institutes (NMIs) and Designated Institutes (DIs) focused on serving stake holder isotope ratio measurement needs. During the current five-year mandate, it is expected that the IRWG will make significant advances in: i. delta scale definition, ii. measurement comparability of relative isotope ratio measurements, iii. comparable measurement capabilities for C and N isotope ratio measurement; and iv. the understanding of calibration modalities used in metal isotope ratio characterization. 2. SCIENTIFIC, ECONOMIC AND SOCIAL CHALLENGES Isotopes have long been recognized as markers for a wide variety of molecular processes. Indeed, applications where isotope ratios are used provide scientific, economic, and social value. Early applications of isotope measurements were recognized with the 1943 Nobel Prize for Chemistry which included the determination of the water content in the human body, determination of solubility of various low-solubility salts, and development of the isotope dilution method which has since become the cornerstone of analytical chemistry. -

Binary Operator
Table of Contents Teaching and Learning The Metric System Unit 1 1 - Suggested Teaching Sequence 1 - Objectives 1 - Rules of Notation 1 - Metric Units, Symbols, and Referents 2 - Metric Prefixes 2 - Linear Measurement Activities 3 - Area Measurement Activities 5 - Volume Measurement Activities 7 - Mass (Weight) Measurement Activities 9 - Temperature Measurement Activities 11 Unit 2 12 - Objectives 12 - Suggested Teaching Sequence 12 - Metrics in this Occupation 12 - Metric Units For Binary Operation 13 - Trying Out Metric Units 14 - Binding With Metrics 15 Unit 3 16 - Objective 16 - Suggested Teaching Sequence 16 - Metric-Metric Equivalents 16 - Changing Units at Work 18 Unit 4 19 - Objective 19 - Suggested Teaching Sequence 19 - Selecting and Using Metric Instruments, Tools and Devices 19 - Which Tools for the Job? 20 - Measuring Up in Binary Operations 20 Unit 5 21 - Objective 21 - Suggested Teaching Sequence 21 - Metric-Customary Equivalents 21 - Conversion Tables 22 - Any Way You Want It 23 Testing Metric Abilities 24 Answers to Exercises and Test 25 Tools and Devices List References TEACHING AND LEARNING THE METRIC SYSTEM Thi.s metric instructional package was designed to meet job-related Unit 2 provides the metric terms which are used in this occupation metric measurement needs of students. To use this package students and gives experience with occupational measurement tasks. should already know the occupational terminology, measurement terms, and tools currently in use. These materials were prepared with Unit 3 focuses on job-related metric equivalents and their relation the help of experienced vocational teachers, reviewed by experts, tested ships. in classrooms in different parts of the United States, and revised before distribution. -

Having Regard to the Opinion of the European Chapter 1 of the Annex Binding Within Five Years of Parliament1 ; the Date of Entry Into Force of This Directive
878 Official Journal of the European Communities 29.10.71 Official Journal of the European Communities No L 243/29 COUNCIL DIRECTIVE of 18 October 1971 on the approximation of the laws of the Member States relating to units of measurement (71/354/EEC ) THE COUNCIL OF THE EUROPEAN COMMUNITIES, particular their names, symbols and use are not identical in the Member countries ; Having regard to the Treaty establishing the European Economic Community, and in particular HAS ADOPTED THIS DIRECTIVE : Article 100 thereof; Article 1 Having regard to the proposal from the Commission ; 1 . Member States shall make the provisions of Having regard to the Opinion of the European Chapter 1 of the Annex binding within five years of Parliament1 ; the date of entry into force of this Directive. 2 . Member States shall, with effect from 31 Having regard to the Opinion of the Economic and December 1977 at the latest, prohibit the use of the Social Committee2; units of measurement listed in Chapter III of the Annex. Whereas - the laws which regulate the use of units of measurement in the Member States differ from one 3 . The units of measurement temporarily" retained Member State to another and therefore hinder trade ; in accordance with the provisions of Chapter II or whereas application of the rules relating to measuring Chapter III of the Annex may not be brought into instruments is closely linked to the use of units of compulsory use by the Member States where they ' are measurement in the metrological system ; whereas, in not authorised at the date when this Directive enters into force . -

About SI Units
Units SI units: International system of units (French: “SI” means “Système Internationale”) The seven SI base units are: Mass: kg Defined by the prototype “standard kilogram” located in Paris. The standard kilogram is made of Pt metal. Length: m Originally defined by the prototype “standard meter” located in Paris. Then defined as 1,650,763.73 wavelengths of the orange-red radiation of Krypton86 under certain specified conditions. (Official definition: The distance traveled by light in vacuum during a time interval of 1 / 299 792 458 of a second) Time: s The second is defined as the duration of a certain number of oscillations of radiation coming from Cs atoms. (Official definition: The second is the duration of 9,192,631,770 periods of the radiation of the hyperfine- level transition of the ground state of the Cs133 atom) Current: A Defined as the current that causes a certain force between two parallel wires. (Official definition: The ampere is that constant current which, if maintained in two straight parallel conductors of infinite length, of negligible circular cross-section, and placed 1 meter apart in vacuum, would produce between these conductors a force equal to 2 × 10-7 Newton per meter of length. Temperature: K One percent of the temperature difference between boiling point and freezing point of water. (Official definition: The Kelvin, unit of thermodynamic temperature, is the fraction 1 / 273.16 of the thermodynamic temperature of the triple point of water. Amount of substance: mol The amount of a substance that contains Avogadro’s number NAvo = 6.0221 × 1023 of atoms or molecules. -

Paul Hertz Astrophysics Subcommittee
Paul Hertz Astrophysics Subcommittee February 23, 2012 Agenda • Science Highlights • Organizational Changes • Program Update • President’s FY13 Budget Request • Addressing Decadal Survey Priorities 2 Chandra Finds Milky Way’s Black Hole Grazing on Asteroids • The giant black hole at the center of the Milky Way may be vaporizing and devouring asteroids, which could explain the frequent flares observed, according to astronomers using data from NASA's Chandra X-ray Observatory. For several years Chandra has detected X-ray flares about once a day from the supermassive black hole known as Sagittarius A*, or "Sgr A*" for short. The flares last a few hours with brightness ranging from a few times to nearly one hundred times that of the black hole's regular output. The flares also have been seen in infrared data from ESO's Very Large Telescope in Chile. • Scientists suggest there is a cloud around Sgr A* containing trillions of asteroids and comets, stripped from their parent stars. Asteroids passing within about 100 million miles of the black hole, roughly the distance between the Earth and the sun, would be torn into pieces by the tidal forces from the black hole. • These fragments then would be vaporized by friction as they pass through the hot, thin gas flowing onto Sgr A*, similar to a meteor heating up and glowing as it falls through Earth's atmosphere. A flare is produced and the remains of the asteroid are eventually swallowed by the black hole. • The authors estimate that it would take asteroids larger than about six miles in radius to generate the flares observed by Chandra. -

Guide for the Use of the International System of Units (SI)
Guide for the Use of the International System of Units (SI) m kg s cd SI mol K A NIST Special Publication 811 2008 Edition Ambler Thompson and Barry N. Taylor NIST Special Publication 811 2008 Edition Guide for the Use of the International System of Units (SI) Ambler Thompson Technology Services and Barry N. Taylor Physics Laboratory National Institute of Standards and Technology Gaithersburg, MD 20899 (Supersedes NIST Special Publication 811, 1995 Edition, April 1995) March 2008 U.S. Department of Commerce Carlos M. Gutierrez, Secretary National Institute of Standards and Technology James M. Turner, Acting Director National Institute of Standards and Technology Special Publication 811, 2008 Edition (Supersedes NIST Special Publication 811, April 1995 Edition) Natl. Inst. Stand. Technol. Spec. Publ. 811, 2008 Ed., 85 pages (March 2008; 2nd printing November 2008) CODEN: NSPUE3 Note on 2nd printing: This 2nd printing dated November 2008 of NIST SP811 corrects a number of minor typographical errors present in the 1st printing dated March 2008. Guide for the Use of the International System of Units (SI) Preface The International System of Units, universally abbreviated SI (from the French Le Système International d’Unités), is the modern metric system of measurement. Long the dominant measurement system used in science, the SI is becoming the dominant measurement system used in international commerce. The Omnibus Trade and Competitiveness Act of August 1988 [Public Law (PL) 100-418] changed the name of the National Bureau of Standards (NBS) to the National Institute of Standards and Technology (NIST) and gave to NIST the added task of helping U.S. -

Role of Metrology in Conformity Assessment
Role of Metrology in Conformity Assessment Andy Henson Director of International Liaison and Communications of the BIPM Metrology, the science of measurement… and its applications “When you can measure what you are speaking about, and express it in numbers, you know something about it; but when you cannot express it in numbers, your knowledge is of a meagre and unsatisfactory kind” William Thompson (Lord Kelvin): Lecture on "Electrical Units of Measurement" (3 May 1883) Conformity assessment Overall umbrella of measures taken by : ‐manufacturers ‐customers ‐regulatory authorities ‐independent third parties To assess that a product/service meets standards or technical regulations 2 Metrology is a part of our lives from birth Best regulation Safe food Technical evidence Safe treatment Safe baby food Manufacturing Safe traveling Globalization weighing of baby Healthcare Innovation Climate change Without metrology, you can’t discover, design, build, test, manufacture, maintain, prove, buy or operate anything safely and reliably. From filling your car with petrol to having an X‐ray at a hospital, your life is surrounded by measurements. In industry, from the thread of a nut and bolt and the precision machined parts on engines down to tiny structures on micro and nano components, all require an accurate measurement that is recognized around the world. Good measurement allows country to remain competitive, trade throughout the world and improve quality of life. www.bipm.org 3 Metrology, the science of measurement As we have seen, measurement science is not purely the preserve of scientists. It is something of vital importance to us all. The intricate but invisible network of services, suppliers and communications upon which we are all dependent rely on metrology for their efficient and reliable operation.