2005 List of Controlled Substances
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Controlled Substances Included. [M.S.A
1998 PUBLIC AND LOCAL ACTS [No. 319] (HB 4065) AN ACT to amend 1978 PA 368, entitled “An act to protect and promote the public health; to codify, revise, consolidate, classify, and add to the laws relating to public health; to provide for the prevention and control of diseases and disabilities; to provide for the classification, administration, regulation, financing, and maintenance of personal, environ- mental, and other health services and activities; to create or continue, and prescribe the powers and duties of, departments, boards, commissions, councils, committees, task forces, and other agencies; to prescribe the powers and duties of governmental entities and officials; to regulate occupations, facilities, and agencies affecting the public health; to regulate health maintenance organizations and certain third party administrators and insurers; to provide for the imposition of a regulatory fee; to promote the efficient and economical delivery of health care services, to provide for the appropriate utilization of health care facilities and services, and to provide for the closure of hospitals or consolidation of hospitals or services; to provide for the collection and use of data and information; to provide for the transfer of property; to provide certain immunity from liability; to regulate and prohibit the sale and offering for sale of drug paraphernalia under certain circumstances; to provide for penalties and remedies; to provide for sanctions for violations of this act and local ordinances; to repeal certain acts and parts of acts; to repeal certain parts of this act; and to repeal certain parts of this act on specific dates,” by amending sections 7218 and 7401 (MCL 333.7218 and 333.7401), section 7401 as amended by 1996 PA 249, and by adding section 7401a. -

124.210 Schedule IV — Substances Included. 1
1 CONTROLLED SUBSTANCES, §124.210 124.210 Schedule IV — substances included. 1. Schedule IV shall consist of the drugs and other substances, by whatever official name, common or usual name, chemical name, or brand name designated, listed in this section. 2. Narcotic drugs. Unless specifically excepted or unless listed in another schedule, any material, compound, mixture, or preparation containing any of the following narcotic drugs, or their salts calculated as the free anhydrous base or alkaloid, in limited quantities as set forth below: a. Not more than one milligram of difenoxin and not less than twenty-five micrograms of atropine sulfate per dosage unit. b. Dextropropoxyphene (alpha-(+)-4-dimethylamino-1,2-diphenyl-3-methyl-2- propionoxybutane). c. 2-[(dimethylamino)methyl]-1-(3-methoxyphenyl)cyclohexanol, its salts, optical and geometric isomers and salts of these isomers (including tramadol). 3. Depressants. Unless specifically excepted or unless listed in another schedule, any material, compound, mixture, or preparation which contains any quantity of the following substances, including its salts, isomers, and salts of isomers whenever the existence of such salts, isomers, and salts of isomers is possible within the specific chemical designation: a. Alprazolam. b. Barbital. c. Bromazepam. d. Camazepam. e. Carisoprodol. f. Chloral betaine. g. Chloral hydrate. h. Chlordiazepoxide. i. Clobazam. j. Clonazepam. k. Clorazepate. l. Clotiazepam. m. Cloxazolam. n. Delorazepam. o. Diazepam. p. Dichloralphenazone. q. Estazolam. r. Ethchlorvynol. s. Ethinamate. t. Ethyl Loflazepate. u. Fludiazepam. v. Flunitrazepam. w. Flurazepam. x. Halazepam. y. Haloxazolam. z. Ketazolam. aa. Loprazolam. ab. Lorazepam. ac. Lormetazepam. ad. Mebutamate. ae. Medazepam. af. Meprobamate. ag. Methohexital. ah. Methylphenobarbital (mephobarbital). -

Outline for Controlled Substances Program
Environmental Health and Safety Controlled Substances Program Date of Issuance: Review Date: 10/1/2019 (no changes) 10/01/2018 Revision Number: Initial Prepared by: EH&S Table of Contents HEADINGS Introduction Applicability Responsibilities Registration Requirements Authorized Use Ordering/Purchasing Administering and Dispensing Inventory Procedures (Continuing Records) Security Disposal FORMS: Registering or renewing a DEA or state license (CMU) Controlled Substances Authorized users list (CMU) Employee questionnaire for those with access to controlled substances (CMU) Record of Form 222 use (Order form) (CMU) Records of Controlled Substance Purchases (CMU) Record of Controlled Substance Administering and dispensing (CMU) Controlled Substance Physical Inventory (CMU) DEA Registration of Persons doing research or analysis (Form 225) DEA Registration of Dispensers (Form 224) DEA Registration Instructional (Form 224 and 226 to renew) DEA Report of loss or theft (Form 106) DEA Report of drugs surrendered (From 41) DEA SCHEDULES: Schedule I Schedule II Schedule III Schedule IV Schedule V INTRODUCTION State and Federal regulations have been promulgated concerning the use and handling of US Department of Justice Drug Enforcement Administration (DEA) controlled substances. These regulations are in place to address materials which are or have the potential to be addictive or habit forming. These substances have been categorized into “schedules” that have been created by the DEA to reflect their level of concern. The “Carnegie Mellon University DEA Controlled Substances Program” is intended to ensure that Carnegie Mellon University is in compliance with our regulatory requirements. Required activities under the DEA include: 1. Registration of your work with the DEA and with Carnegie Mellon’s Department of Environmental Health and Safety (EH&S). -

EUROPEAN PHARMACOPOEIA 10.0 Index 1. General Notices
EUROPEAN PHARMACOPOEIA 10.0 Index 1. General notices......................................................................... 3 2.2.66. Detection and measurement of radioactivity........... 119 2.1. Apparatus ............................................................................. 15 2.2.7. Optical rotation................................................................ 26 2.1.1. Droppers ........................................................................... 15 2.2.8. Viscosity ............................................................................ 27 2.1.2. Comparative table of porosity of sintered-glass filters.. 15 2.2.9. Capillary viscometer method ......................................... 27 2.1.3. Ultraviolet ray lamps for analytical purposes............... 15 2.3. Identification...................................................................... 129 2.1.4. Sieves ................................................................................. 16 2.3.1. Identification reactions of ions and functional 2.1.5. Tubes for comparative tests ............................................ 17 groups ...................................................................................... 129 2.1.6. Gas detector tubes............................................................ 17 2.3.2. Identification of fatty oils by thin-layer 2.2. Physical and physico-chemical methods.......................... 21 chromatography...................................................................... 132 2.2.1. Clarity and degree of opalescence of -

A. Schedule IV Shall Consist Of
1 REVISOR 6800.4240 6800.4240 SCHEDULE IV CONTROLLED SUBSTANCES. The following items are listed in Schedule IV: A. Schedule IV shall consist of the drugs and other substances, by whatever official name, common or usual name, chemical name, or brand name designated, listed in this part. B. Narcotic drugs. Unless specifically excepted or unless listed in another schedule, any material, compound, mixture, or preparation containing any of the following narcotic drugs, or their salts calculated as the free anhydrous base or alkaloid, in limited quantities as follows: (1) Not more than one milligram of difenoxin and not less than 25 micrograms of atropine sulfate per dosage unit. (2) Dextropropoxyphene (alpha-(+)-4-dimethylamino-1,2-diphenyl-3-methyl-2- propionoxybutane), for example, Darvon, Darvocet. C. Depressants. Unless specifically excepted or unless listed in another schedule, any material, compound, mixture, or preparation which contains any quantity of the following substances, including its salts, isomers, and salts of isomers whenever the existence of such salts, isomers, and salts of isomers is possible within the specific chemical designation: Statutory Name Some examples of common names, trade names, or names of products which contain a controlled substance (1) Alprazolam Xanax (2) Barbital Barbitone (3) Bromazepam (4) Camazepam (5) Chloral betaine Beta-Chlor (6) Chloral hydrate Noctec, Somnos (7) Chlordiazepoxide Librium, Libritabs (8) Clobazam (9) Clonazepam Clonopin (10) Clorazepate Tranxene (11) Clotiazepam Copyright ©2013 -

Stembook 2018.Pdf
The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances FORMER DOCUMENT NUMBER: WHO/PHARM S/NOM 15 WHO/EMP/RHT/TSN/2018.1 © World Health Organization 2018 Some rights reserved. This work is available under the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 IGO licence (CC BY-NC-SA 3.0 IGO; https://creativecommons.org/licenses/by-nc-sa/3.0/igo). Under the terms of this licence, you may copy, redistribute and adapt the work for non-commercial purposes, provided the work is appropriately cited, as indicated below. In any use of this work, there should be no suggestion that WHO endorses any specific organization, products or services. The use of the WHO logo is not permitted. If you adapt the work, then you must license your work under the same or equivalent Creative Commons licence. If you create a translation of this work, you should add the following disclaimer along with the suggested citation: “This translation was not created by the World Health Organization (WHO). WHO is not responsible for the content or accuracy of this translation. The original English edition shall be the binding and authentic edition”. Any mediation relating to disputes arising under the licence shall be conducted in accordance with the mediation rules of the World Intellectual Property Organization. Suggested citation. The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances. Geneva: World Health Organization; 2018 (WHO/EMP/RHT/TSN/2018.1). Licence: CC BY-NC-SA 3.0 IGO. Cataloguing-in-Publication (CIP) data. -

Drug/Substance Trade Name(S)
A B C D E F G H I J K 1 Drug/Substance Trade Name(s) Drug Class Existing Penalty Class Special Notation T1:Doping/Endangerment Level T2: Mismanagement Level Comments Methylenedioxypyrovalerone is a stimulant of the cathinone class which acts as a 3,4-methylenedioxypyprovaleroneMDPV, “bath salts” norepinephrine-dopamine reuptake inhibitor. It was first developed in the 1960s by a team at 1 A Yes A A 2 Boehringer Ingelheim. No 3 Alfentanil Alfenta Narcotic used to control pain and keep patients asleep during surgery. 1 A Yes A No A Aminoxafen, Aminorex is a weight loss stimulant drug. It was withdrawn from the market after it was found Aminorex Aminoxaphen, Apiquel, to cause pulmonary hypertension. 1 A Yes A A 4 McN-742, Menocil No Amphetamine is a potent central nervous system stimulant that is used in the treatment of Amphetamine Speed, Upper 1 A Yes A A 5 attention deficit hyperactivity disorder, narcolepsy, and obesity. No Anileridine is a synthetic analgesic drug and is a member of the piperidine class of analgesic Anileridine Leritine 1 A Yes A A 6 agents developed by Merck & Co. in the 1950s. No Dopamine promoter used to treat loss of muscle movement control caused by Parkinson's Apomorphine Apokyn, Ixense 1 A Yes A A 7 disease. No Recreational drug with euphoriant and stimulant properties. The effects produced by BZP are comparable to those produced by amphetamine. It is often claimed that BZP was originally Benzylpiperazine BZP 1 A Yes A A synthesized as a potential antihelminthic (anti-parasitic) agent for use in farm animals. -
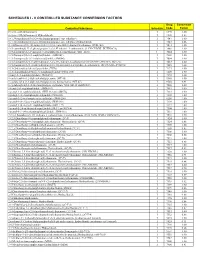
Controlled Substance Conversion Factors
SCHEDULES I - V CONTROLLED SUBSTANCE CONVERSION FACTORS Drug Conversion Controlled Substance Schedule Code Factor (+/-)cis -4-Methylaminorex I 1590 1.00 (+/-)cis -4-Methylaminorex Hydrochloride I 1590 0.83 1-(1,3-benzodioxol-5-yl)-2-(ethylamino)propan-1-one (ethylone) I 7547 1.00 1-(1,3-benzodioxol-5-yl)-2-(ethylamino)propan-1-one (ethylone) hydrochloride I 7547 0.86 (1-(4-fluorobenzyl)-1H -indol-3-yl)(2,2,3,3-tetramethylcyclopropyl)methanone (FUB-144) I 7014 1.00 1-(4-cyanobutyl)-N -(2-phenylpropan-2-yl)-1H -indazole-3-carboxamide (4-CN-CUMYL-BUTINACA) I 7089 1.00 1-(5-fluoropentyl)-1H -indazol-3-yl](naphthalen-1-yl)methanone (THJ–2201) 1 7024 1.00 1-(5-fluoropentyl)-3-(1-naphthoyl)indole (AM2201) I 7201 1.00 1-(5-fluoropentyl)-3-(2-iodobenzoyl)indole (AM694) I 7694 1.00 1-(5-fluoropentyl)-N -(2-phenylpropan-2-yl)-1H -indazole-3-carboxamide (5F-CUMYL-PINACA; SGT-25) I 7083 1.00 1-(5-fluoropentyl)-N -(2-phenylpropan-2-yl)-1H -pyrrolo[2,3-b]pyridine-3-carboxamide (5F-CUMYL-P7AICA) I 7085 1.00 1-[1-(2-thienyl)cyclohexyl]pyrrolidine (TCPy) I 7473 1.00 1-[2-(4-morpholinyl)ethyl]-3-(1-naphthoyl)indole (JWH- 200) I 7200 1.00 1-butyl-3-(1-naphthoyl)indole (JWH-073) I 7173 1.00 1-cyclohexyl-4-(1,2-diphenylethyl)piperazine (MT-45) I 9560 1.00 1-cyclohexyl-4-(1,2-diphenylethyl)piperazine hydrochloride (MT-45) I 9560 0.91 1-cyclohexylethyl-3-(2-methoxyphenylacetyl)indole 7008 (SR-18 and RCS-8) I 7008 1.00 1-hexyl-3-(1-naphthoyl)indole (JWH-019) I 7019 1.00 1-pentyl-3-(1-naphthoyl)indole (JWH-018 and AM678) I 7118 1.00 1-pentyl-3-(2-chlorophenylacetyl)indole -

Chapter 961 Uniform Controlled Substances Act
1 Updated 99−00 Wis. Stats. Database UNIFORM CONTROLLED SUBSTANCES ACT 961.01 CHAPTER 961 UNIFORM CONTROLLED SUBSTANCES ACT 961.001 Declaration of intent. 961.437 Possession and disposal of waste from manufacture of methamphetamine. SUBCHAPTER I 961.438 Minimum sentence. DEFINITIONS 961.44 Penalties under other laws. 961.01 Definitions. 961.45 Bar to prosecution. SUBCHAPTER II 961.455 Using a child for illegal drug distribution or manufacturing purposes. 961.46 Distribution to persons under age 18. STANDARDS AND SCHEDULES 961.465 Distribution to prisoners. 961.11 Authority to control. 961.47 Conditional discharge for possession or attempted possession as first 961.115 Native American Church exemption. offense. 961.12 Nomenclature. 961.472 Assessment; certain possession or attempted possession offenses. 961.13 Schedule I tests. 961.475 Treatment option. 961.14 Schedule I. 961.48 Second or subsequent offenses. 961.15 Schedule II tests. 961.49 Distribution of or possession with intent to deliver a controlled substance 961.16 Schedule II. on or near certain places. 961.17 Schedule III tests. 961.492 Distribution or possession with intent to deliver certain controlled sub- 961.18 Schedule III. stances on public transit vehicles. 961.19 Schedule IV tests. 961.495 Possession or attempted possession of a controlled substance on or near 961.20 Schedule IV. certain places. 961.21 Schedule V tests. 961.50 Suspension or revocation of operating privilege. 961.22 Schedule V. SUBCHAPTER V 961.23 Dispensing of schedule V substances. ENFORCEMENT AND ADMINISTRATIVE PROVISIONS 961.24 Publishing of updated schedules. 961.51 Powers of enforcement personnel. 961.25 Controlled substance analog treated as a schedule I substance. -

Terminology and Information on Drugs Third Edition
Terminology and Information on Drugs Third edition Terminology and Information on Drugs Third edition UNITED NATIONS NEW YORK, 2016 UNITED NATIONS PUBLICATION Sales No. E.16.XI.8 ISBN: 978-92-1-148287-4 eISBN: 978-92-1-057914-8 Original language: English © United Nations, March 2016. All rights reserved, worldwide The designations employed and the presentation of material in this publication do not imply the expression of any opinion whatsoever on the part of the Secretariat of the United Nations concerning the legal status of any country, territory, city or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries. This publication has not been formally edited. Publishing production: English, Publishing and Library Section, United Nations Office at Vienna. Photographs: UNODC and Health Sciences Authority, Singapore. ACKNOWLEDGEMENTS UNODC’s Laboratory and Scientific Section (LSS), headed by Dr. Justice Tettey, wishes to express its appreciation and thanks to the following experts who participated in an expert group meeting and/or contributed to the development and review of this revision of Terminology and Information on Drugs: Dr. Heesun Chung Graduate School of Analytical Science and Technology (GRAST), South Korea Prof. Les Iversen Advisory Council on the Misuse of Drugs (ACMD), United Kingdom Dr. Wendy Lim Jong Lee Health Sciences Authority, Singapore Dr. Franco Tagliaro University of Verona, Italy Mr. Eleuterio Umpiérrez Faculty of Chemistry, Universidad de la República, Uruguay Mr. Scott Oulton DEA Office of Forensic Sciences, United States The preparation of this manual was coordinated by Dr. Conor Crean, Dr. Iphigenia Naidis and Ms. Susan Ifeagwu, staff of LSS. -

A Summary of California's Alcohol and Drug Abuse Laws
If you have issues viewing or accessing this file contact us at NCJRS.gov. A SUMMARY OF CALIFORNIA'S ALCOHOL AND DRUG ABUSE LAWS Prepared pursuant to SB 2599 (Seymour), (Chapter 983, Statutes of 1988) Prepared by: SENATE OFFICE OF RESEARCH Elisabeth Kersten, Director June 3D, 1989 462-8 127464 U.S. Department of Justice National Institute of Justice This document has been reproduced exactly as received from the person or organization originating It. Points of view or opinions stated In this document are those of the authors and do not necessarily represent the official position or policies of the National Institute of Justice. Permission to reproduce this copyrighted material has been granted bl:: California Legislature to the National Criminal Justice Reference Service (NCJRS). Further reproduction outside of the NCJRS system requires permis sion of the copyright owner. A SUMMARY OF CALIFORNIA'S ALCOHOL AND DRUG ABUSE LAWS Prepared pursuant to SB 2599 (Seymour), (Chapter 983, Statutes of 1988) June 30, 1989 Prepared by: SENATE OFFICE OF RESEARCH, Ken Hurdle, Project Director, Kim Connor, Dan Englund, Dolores Sanchez, Lenore Tate Edited by: Judith A. Ryder, Thomas F. Wilspn ~;:- ~~~: ~M8ERS: ~ILLIAM A. CRAVEN to DAVIS CALIFORNIA LEGISLATURE VADIE DEDDEH TERRI DELGADILLO ~'ECIL GREEN , COMMITTEE CONSULTANT ;'ARY HART !ILL" LOCKYER STATE CAPITOL. ROOM 3074 !'E8ECCA MORGAN SACRAMENTO. CA 95814 ilCHOLAS PETRIS (916) 445-4264 to ROYCE ~ANEWATSON ~ SENATE SELECT COMMITTEE ~ ON ~ ~:, SUBSTANCE ABUSE r~, ~ t; JOHN SEYMOUR ~: !i CHAIRMAN 1 ~~ tl December 19, 1989 r1 \i ~ ~, As Chairman of the Senate Select Committee on Substance l\ Abuse, I am pleased to participate with the Senate Office of I' Research in the release of A Summary of California's Alcohol ~ and Drug Abuse Laws. -

Uniform Classification Guidelines for Foreign Substances, Version 7.00
Association of Racing Commissioners International, Inc. Drug Testing Standards and Practices Program Model Rules Guidelines Uniform Classification Guidelines for Foreign Substances and Recommended Penalties and Model Rule Version 7.00 Revised January 2014 Association of Racing Commissioners International, Inc. Uniform Classification Guidelines for Foreign Substances Table of Contents Preamble to the Uniform Classification Guidelines of Foreign Substances ..................................................................................... ii Notes Regarding Classification Guidelines ...................................................................................................................................... ii Classification Criteria ....................................................................................................................................................................... iii Classification Definitions ................................................................................................................................................................. iv Drug Classification Scheme ............................................................................................................................................................. vi Alphabetical Substance List ............................................................................................................................................................... 1 Listing by Classification ..................................................................................................................................................................