124.210 Schedule IV — Substances Included. 1
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Management of Status Epilepticus
Published online: 2019-11-21 THIEME Review Article 267 Management of Status Epilepticus Ritesh Lamsal1 Navindra R. Bista1 1Department of Anaesthesiology, Tribhuvan University Teaching Address for correspondence Ritesh Lamsal, MD, DM, Department Hospital, Institute of Medicine, Tribhuvan University, Nepal of Anaesthesiology, Tribhuvan University Teaching Hospital, Institute of Medicine, Tribhuvan University,Kathmandu, Nepal (e-mail: [email protected]). J Neuroanaesthesiol Crit Care 2019;6:267–274 Abstract Status epilepticus (SE) is a life-threatening neurologic condition that requires imme- diate assessment and intervention. Over the past few decades, the duration of seizure Keywords required to define status epilepticus has shortened, reflecting the need to start thera- ► convulsive status py without the slightest delay. The focus of this review is on the management of con- epilepticus vulsive and nonconvulsive status epilepticus in critically ill patients. Initial treatment ► neurocritical care of both forms of status epilepticus includes immediate assessment and stabilization, ► nonconvulsive status and administration of rapidly acting benzodiazepine therapy followed by nonbenzodi- epilepticus azepine antiepileptic drug. Refractory and super-refractory status epilepticus (RSE and ► refractory status SRSE) pose a lot of therapeutic problems, necessitating the administration of contin- epilepticus uous infusion of high doses of anesthetic agents, and carry a high risk of debilitating ► status epilepticus morbidity as well as mortality. ► super-refractory sta- tus epilepticus Introduction occur after 30 minutes of seizure activity. However, this working definition did not indicate the need to immediately Status epilepticus (SE) is a medical and neurologic emergency commence treatment and that permanent neuronal injury that requires immediate evaluation and treatment. It is associat- could occur by the time a clinical diagnosis of SE was made. -

The National Drugs List
^ ^ ^ ^ ^[ ^ The National Drugs List Of Syrian Arab Republic Sexth Edition 2006 ! " # "$ % &'() " # * +$, -. / & 0 /+12 3 4" 5 "$ . "$ 67"5,) 0 " /! !2 4? @ % 88 9 3: " # "$ ;+<=2 – G# H H2 I) – 6( – 65 : A B C "5 : , D )* . J!* HK"3 H"$ T ) 4 B K<) +$ LMA N O 3 4P<B &Q / RS ) H< C4VH /430 / 1988 V W* < C A GQ ") 4V / 1000 / C4VH /820 / 2001 V XX K<# C ,V /500 / 1992 V "!X V /946 / 2004 V Z < C V /914 / 2003 V ) < ] +$, [2 / ,) @# @ S%Q2 J"= [ &<\ @ +$ LMA 1 O \ . S X '( ^ & M_ `AB @ &' 3 4" + @ V= 4 )\ " : N " # "$ 6 ) G" 3Q + a C G /<"B d3: C K7 e , fM 4 Q b"$ " < $\ c"7: 5) G . HHH3Q J # Hg ' V"h 6< G* H5 !" # $%" & $' ,* ( )* + 2 ا اوا ادو +% 5 j 2 i1 6 B J' 6<X " 6"[ i2 "$ "< * i3 10 6 i4 11 6! ^ i5 13 6<X "!# * i6 15 7 G!, 6 - k 24"$d dl ?K V *4V h 63[46 ' i8 19 Adl 20 "( 2 i9 20 G Q) 6 i10 20 a 6 m[, 6 i11 21 ?K V $n i12 21 "% * i13 23 b+ 6 i14 23 oe C * i15 24 !, 2 6\ i16 25 C V pq * i17 26 ( S 6) 1, ++ &"r i19 3 +% 27 G 6 ""% i19 28 ^ Ks 2 i20 31 % Ks 2 i21 32 s * i22 35 " " * i23 37 "$ * i24 38 6" i25 39 V t h Gu* v!* 2 i26 39 ( 2 i27 40 B w< Ks 2 i28 40 d C &"r i29 42 "' 6 i30 42 " * i31 42 ":< * i32 5 ./ 0" -33 4 : ANAESTHETICS $ 1 2 -1 :GENERAL ANAESTHETICS AND OXYGEN 4 $1 2 2- ATRACURIUM BESYLATE DROPERIDOL ETHER FENTANYL HALOTHANE ISOFLURANE KETAMINE HCL NITROUS OXIDE OXYGEN PROPOFOL REMIFENTANIL SEVOFLURANE SUFENTANIL THIOPENTAL :LOCAL ANAESTHETICS !67$1 2 -5 AMYLEINE HCL=AMYLOCAINE ARTICAINE BENZOCAINE BUPIVACAINE CINCHOCAINE LIDOCAINE MEPIVACAINE OXETHAZAINE PRAMOXINE PRILOCAINE PREOPERATIVE MEDICATION & SEDATION FOR 9*: ;< " 2 -8 : : SHORT -TERM PROCEDURES ATROPINE DIAZEPAM INJ. -

Federal Register/Vol. 79, No. 39/Thursday, February 27, 2014/Rules and Regulations
Federal Register / Vol. 79, No. 39 / Thursday, February 27, 2014 / Rules and Regulations 10985 (2) Turkeys— Combination in Zoalene in grams/ton grams per ton Indications for use Limitations Sponsor (i) 113.5 to 170.3 ..... ................................. Growing turkeys: For prevention and con- Feed continuously as sole ration. For tur- 054771 trol of coccidiosis. keys grown for meat purposes only. Do not feed to laying birds. (ii) 113.5 to 170.3 .... Bacitracin methylene Growing turkeys: For prevention and con- Feed continuously as sole ration until 14 054771 disalicylate 4 to 50. trol of coccidiosis; and for increased to 16 weeks of age. For turkeys grown rate of weight gain and improved feed for meat purposes only. Do not feed to efficiency. laying birds. Dated: February 3, 2014. Legal Authority initiated by the Attorney General (1) on Bernadette Dunham, The DEA implements and enforces his own motion; (2) at the request of the Director, Center for Veterinary Medicine. titles II and III of the Comprehensive Secretary of the Department of Health 1 [FR Doc. 2014–02617 Filed 2–26–14; 8:45 am] Drug Abuse Prevention and Control Act and Human Services (HHS), or (3) on the petition of any interested party. 21 BILLING CODE 4160–01–P of 1970, as amended. Titles II and III are referred to as the ‘‘Controlled U.S.C. 811(a). This action is based on a Substances Act’’ and the ‘‘Controlled recommendation from the Assistant DEPARTMENT OF JUSTICE Substances Import and Export Act,’’ Secretary of the HHS and on an respectively, and are collectively evaluation of all other relevant data by Drug Enforcement Administration referred to as the ‘‘Controlled the DEA. -

Drug and Alcohol Withdrawal Clinical Practice Guidelines - NSW
Guideline Drug and Alcohol Withdrawal Clinical Practice Guidelines - NSW Summary To provide the most up-to-date knowledge and current level of best practice for the treatment of withdrawal from alcohol and other drugs such as heroin, and other opioids, benzodiazepines, cannabis and psychostimulants. Document type Guideline Document number GL2008_011 Publication date 04 July 2008 Author branch Centre for Alcohol and Other Drugs Branch contact (02) 9424 5938 Review date 18 April 2018 Policy manual Not applicable File number 04/2766 Previous reference N/A Status Active Functional group Clinical/Patient Services - Pharmaceutical, Medical Treatment Population Health - Pharmaceutical Applies to Area Health Services/Chief Executive Governed Statutory Health Corporation, Board Governed Statutory Health Corporations, Affiliated Health Organisations, Affiliated Health Organisations - Declared Distributed to Public Health System, Ministry of Health, Public Hospitals Audience All groups of health care workers;particularly prescribers of opioid treatments Secretary, NSW Health Guideline Ministry of Health, NSW 73 Miller Street North Sydney NSW 2060 Locked Mail Bag 961 North Sydney NSW 2059 Telephone (02) 9391 9000 Fax (02) 9391 9101 http://www.health.nsw.gov.au/policies/ space space Drug and Alcohol Withdrawal Clinical Practice Guidelines - NSW space Document Number GL2008_011 Publication date 04-Jul-2008 Functional Sub group Clinical/ Patient Services - Pharmaceutical Clinical/ Patient Services - Medical Treatment Population Health - Pharmaceutical -

The Stimulants and Hallucinogens Under Consideration: a Brief Overview of Their Chemistry and Pharmacology
Drug and Alcohol Dependence, 17 (1986) 107-118 107 Elsevier Scientific Publishers Ireland Ltd. THE STIMULANTS AND HALLUCINOGENS UNDER CONSIDERATION: A BRIEF OVERVIEW OF THEIR CHEMISTRY AND PHARMACOLOGY LOUIS S. HARRIS Dcparlmcnl of Pharmacology, Medical College of Virginia, Virginia Commonwealth Unwersity, Richmond, VA 23298 (U.S.A.) SUMMARY The substances under review are a heterogenous set of compounds from a pharmacological point of view, though many have a common phenylethyl- amine structure. Variations in structure lead to marked changes in potency and characteristic action. The introductory material presented here is meant to provide a set of chemical and pharmacological highlights of the 28 substances under con- sideration. The most commonly used names or INN names, Chemical Abstract (CA) names and numbers, and elemental formulae are provided in the accompanying figures. This provides both some basic information on the substances and a starting point for the more detailed information that follows in the individual papers by contributors to the symposium. Key words: Stimulants, their chemistry and pharmacology - Hallucinogens, their chemistry and pharmacology INTRODUCTION Cathine (Fig. 1) is one of the active principles of khat (Catha edulis). The structure has two asymmetric centers and exists as two geometric isomers, each of which has been resolved into its optical isomers. In the plant it exists as d-nor-pseudoephedrine. It is a typical sympathomimetic amine with a strong component of amphetamine-like activity. The racemic mixture is known generically in this country and others as phenylpropanolamine (dl- norephedrine). It is widely available as an over-the-counter (OTC) anti- appetite agent and nasal decongestant. -

22-244 Biopharmaceutics Review(S)
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 22-244 CLINICAL PHARMACOLOGY AND BIOPHARMACEUTICS REVIEW(S) DEP\RT\IE.'\T OF IIEALTH .\ '\ D Clinical Pharmacology & Biopharmaceutics IH\L\!\ SER\ICES PI HUC IIE\LTH SERVICE (HFD 870) FOOD\'\D DRIG .-\D\""ISTR\TION Tracl"ingl Action Sheet foi' Foi'malllnformai Consults . Iniiii: \rikdllih C. '\dll.lIi_ I'h.I), To: DOCl'\'IENT ROOi\l (LOG-IN and LOC-OlT) Please log-in this consult and review action for the specified IND/NDA submission 1); I I.: II I X::I)I)X ~;\~) :\u.: ND.\ :\u. :::: - ::-1-1 I),\TI 01 DOel "II.NT IIJ' 1:1/::()IJX Sni.11 :\u.: IV-IJIJIJ Class I Resuhiiission '\.\\11.01 imi(, l'I~IORln (()'\ \11)1 R\II ()'\ J).lIC ui' illl,)lli.il.;lmlldl I IJ I Y::()IJX I LlCEDR.\ (Fospropofol) Standanl (ulisLlli: Injection I 1111 :\/\\11 01 \I'O'\\OR: I \1(;1 Pharma I TYPE OF SL InIiSSIO,\ CLI'\I( .\L PIIARMACOI,O(;YIBIOPIL\R\L\CEITlCS RELATED ISSUE o I'I~I.-I:\I) 0 1)IS\OI.11 IO:\I:\-\III~O 1~1I.1 ..\SI. 0 IIN/\1. I'RI1\III) 1..\131 1.1:\( , 0\:\1\ 1\111' III \1\:\ \(\1 IN(i o 1311h \1\11./\131111 Y \ 1(1)11\ ~ L\l31I1NCi RI. VISIO'\ i:\-\iiim \111 ,\HI)I 0 1\\1 0 I "i - \ I \ 0 \\\ I \ I J( i~ Ii)l lSi o (ORRI\I)ONI)IMî o I'R()lI)(.I)1 o Si '1)\( I~II\TII) 0 IWI.(j /\I)VIRTI\I:\( j 01'11\\1. -

Understanding Benzodiazephine Use, Abuse, and Detection
Siemens Healthcare Diagnostics, the leading clinical diagnostics company, is committed to providing clinicians with the vital information they need for the accurate diagnosis, treatment and monitoring of patients. Our comprehensive portfolio of performance-driven systems, unmatched menu offering and IT solutions, in conjunction with highly responsive service, is designed to streamline workflow, enhance operational efficiency and support improved patient care. Syva, EMIT, EMIT II, EMIT d.a.u., and all associated marks are trademarks of General Siemens Healthcare Diagnostics Inc. All Drugs other trademarks and brands are the Global Division property of their respective owners. of Abuse Siemens Healthcare Product availability may vary from Diagnostics Inc. country to country and is subject 1717 Deerfield Road to varying regulatory requirements. Deerfield, IL 60015-0778 Please contact your local USA representative for availability. www.siemens.com/diagnostics Siemens Global Headquarters Global Siemens Healthcare Headquarters Siemens AG Understanding Wittelsbacherplatz 2 Siemens AG 80333 Muenchen Healthcare Sector Germany Henkestrasse 127 Benzodiazephine Use, 91052 Erlangen Germany Abuse, and Detection Telephone: +49 9131 84 - 0 www.siemens.com/healthcare www.usa.siemens.com/diagnostics Answers for life. Order No. A91DX-0701526-UC1-4A00 | Printed in USA | © 2009 Siemens Healthcare Diagnostics Inc. Syva has been R1 R2 a leading developer N and manufacturer of AB R3 X N drugs-of-abuse tests R4 for more than 30 years. R2 C Now part of Siemens Healthcare ® Diagnostics, Syva boasts a long and Benzodiazepines have as their basic chemical structure successful track record in drugs-of-abuse a benzene ring fused to a seven-membered diazepine ring. testing, and leads the industry in the All important benzodiazepines contain a 5-aryl substituent ring (ring C) and a 1,4–diazepine ring. -

Risk Based Requirements for Medicines Handling
Risk based requirements for medicines handling Including requirements for Schedule 4 Restricted medicines Contents 1. Introduction 2 2. Summary of roles and responsibilities 3 3. Schedule 4 Restricted medicines 4 4. Medicines acquisition 4 5. Storage of medicines, including control of access to storage 4 5.1. Staff access to medicines storage areas 5 5.2. Storage of S4R medicines 5 5.3. Storage of S4R medicines for medical emergencies 6 5.4. Access to storage for S4R and S8 medicines 6 5.5. Pharmacy Department access, including after hours 7 5.6. After-hours access to S8 medicines in the Pharmacy Department 7 5.7. Storage of nitrous oxide 8 5.8. Management of patients’ own medicines 8 6. Distribution of medicines 9 6.1. Distribution outside Pharmacy Department operating hours 10 6.2. Distribution of S4R and S8 medicines 10 7. Administration of medicines to patients 11 7.1. Self-administration of scheduled medicines by patients 11 7.2. Administration of S8 medicines 11 8. Supply of medicines to patients 12 8.1. Supply of scheduled medicines to patients by health professionals other than pharmacists 12 9. Record keeping 13 9.1. General record keeping requirements for S4R medicines 13 9.2. Management of the distribution and archiving of S8 registers 14 9.3. Inventories of S4R medicines 14 9.4. Inventories of S8 medicines 15 10. Destruction and discards of S4R and S8 medicines 15 11. Management of oral liquid S4R and S8 medicines 16 12. Cannabis based products 17 13. Management of opioid pharmacotherapy 18 14. -

Retention Behaviour of Some Benzodiazepines in Solid-Phase Extraction Using Modified Silica Adsorbents Having Various Hydrophobicities
ACADEMIA ROMÂNĂ Rev. Roum. Chim., Revue Roumaine de Chimie 2015, 60(9), 891-898 http://web.icf.ro/rrch/ RETENTION BEHAVIOUR OF SOME BENZODIAZEPINES IN SOLID-PHASE EXTRACTION USING MODIFIED SILICA ADSORBENTS HAVING VARIOUS HYDROPHOBICITIES Elena BACALUM,a Mihaela CHEREGIb,* and Victor DAVIDb,* a Research Institute from University of Bucharest – ICUB, 36-46 M. Kogalniceanu Blvd., Bucharest, 050107, Roumania b University of Bucharest, Faculty of Chemistry, Department of Analytical Chemistry, 90 Panduri Ave, Bucharest – 050663, Roumania Received April 6, 2015 The retention properties of six benzodiazepines (alprazolam, bromazepam, diazepam, flunitrazepam, medazepam, and nitrazepam) on four different solid phase extraction silica 1.0 adsorbents with various hydrophobicities (octadecylsilica, octylsilica, phenylsilica, and cyanopropylsilica) were 0.8 H O investigated. The breakthrough curves showed a significant N retention of these compounds on octadecylsilica, octylsilica, 0.6 Br N phenylsilica, excepting alprazolam that has a poor retention on 0 C/C N octadecylsilica. These results can be explained by the 0.4 PHENYL CN hydrophobic character of studied benzodiazepines (octanol- C18 0.2 C8 Bromazepam water partition constant, log Kow, being situated within the interval 1.90-4.45). A poor retention on cyanopropylsilica was 0.0 observed for all studied compounds indicating that π-π and 0 102030405060708090100 Volume (mL) polar intermolecular interactions have a less significant role in their retention on this adsorbent. Generally, the breakthrough -

The 2014 Prohibited List International Standard
The World Anti-Doping Code THE 2014 PROHIBITED LIST INTERNATIONAL STANDARD Version 2.0 (revised 2014 version) The official text of the Prohibited List shall be maintained by WADA and shall be published in English and French. In the event of any conflict between the English and French versions, the English version shall prevail. This List shall come into effect on 1 September 2014 The revised 2014 Prohibited List 17 May 2014 THE 2014 PROHIBITED LIST WORLD ANTI-DOPING CODE Valid 1 September 2014 In accordance with Article 4.2.2 of the World Anti-Doping Code, all Prohibited Substances shall be considered as “Specified Substances” except Substances in classes S1, S2, S4.4, S4.5, S6.a, and Prohibited Methods M1, M2 and M3. SUBSTANCES AND METHODS PROHIBITED AT ALL TIMES (IN- AND OUT-OF-COMPETITION) PROHIBITED SUBSTANCES S0. NON-APPROVED SUBSTANCES Any pharmacological substance which is not addressed by any of the subsequent sections of the List and with no current approval by any governmental regulatory health authority for human therapeutic use (e.g drugs under pre-clinical or clinical development or discontinued, designer drugs, substances approved only for veterinary use) is prohibited at all times. S1. ANABOLIC AGENTS Anabolic agents are prohibited. 1. Anabolic Androgenic Steroids (AAS) a. Exogenous* AAS, including: 1-androstenediol (5α-androst-1-ene-3β,17β-diol ); 1-androstenedione (5α- androst-1-ene-3,17-dione); bolandiol (estr-4-ene-3β,17β-diol ); bolasterone; boldenone; boldione (androsta-1,4-diene-3,17-dione); calusterone; clostebol; -

Benzodiazepines
Benzodiazepines Using benzodiazepines in Children and Adolescents Overview Benzodiazepines are group of medications used to treat several different conditions. Some examples of these medications include: lorazepam (Ativan®); clonazepam (Rivotril®); alprazolam (Xanax®) and oxazepam (Serax®). Other benzodiazepine medications are available, but are less commonly used in children and adolescents. What are benzodiazepines used for? Benzodiazepines may be used for the following conditions: • anxiety disorders: generalized anxiety disorder; social anxiety disorder; post-traumatic stress disorder (PTSD); panic attacks/disorder; excessive anxiety prior to surgery • sleep disorders: trouble sleeping (insomnia); waking up suddenly with great fear (night terrors); sleepwalking • seizure disorders (epilepsy) • alcohol withdrawal • treatment of periods of extreme slowing or excessive purposeless motor activity (catatonia) Your doctor may be using this medication for another reason. If you are unclear why this medication is being prescribed, please ask your doctor. How do benzodiazepines work? Benzodiazepines works by affecting the activity of the brain chemical (neurotransmitter) called GABA. By enhancing the action of GABA, benzodiazepines have a calming effect on parts of the brain that are too excitable. This in turn helps to manage anxiety, insomnia, and seizure disorders. How well do benzodiazepines work in children and adolescents? When used to treat anxiety disorders, benzodiazepines decrease symptoms such as nervousness, fear, and excessive worrying. Benzodiazepines may also help with the physical symptoms of anxiety, including fast or strong heart beat, trouble breathing, dizziness, shakiness, sweating, and restlessness. Typically, benzodiazepines are prescribed to manage anxiety symptoms that are uncomfortable, frightening or interfere with daily activities for a short period of time before conventional anti-anxiety treatments like cognitive-behavioural therapy or anti-anxiety takes effect. -
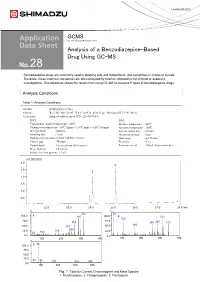
Analysis of a Benzodiazepine-Based Drug Using GC-MS 28
LAAN-E-MS-E028 GCMS Gas Chromatograph Mass Spectrometer Analysis of a Benzodiazepine-Based Drug Using GC-MS 28 Benzodiazepine drugs are commonly used in sleeping aids and tranquilizers, and sometimes in crimes or suicide. Therefore, these chemical substances are often analyzed by forensic laboratories for criminal or academic investigations. This datasheet shows the results from using GC-MS to measure 9 types of benzodiazepine drugs. Analysis Conditions Table 1: Analysis Conditions GC-MS :GCMS-QP2010 Ultra Column : Rxi®-5Sil MS (30 mL. X 0.25 mmI.D., df=0.25 µm, Shimadzu GLC P/N:13623) Glass insert :Silanized splitless insert (P/N: 221-48876-03) [GC] [MS] Vaporization chamber temperature : 260℃ Interface temperature : 280℃ Column oven temperature : 60℃ (2min) -> (10℃/min) -> 320℃ (10min) Ion source temperature : 200℃ Injection mode : Splitless Solvent elution time : 2.0 min Sampling time : 1 min Measurement mode : Scan High pressure injection method: 250 kPa (1.5 min) Mass range : m/z 35-600 Carrier gas : Helium Event time : 0.3 sec Control mode :Linear velocity (45.6 cm/sec) Emission current : 150 µA (high sensitivity) Purge flow rate :3.0 ml/min Sample injection quantity :1.0 µL (x1,000,000) 3.0 3 2.5 2 2.0 1.5 1.0 1 0.5 22.0 23.0 24.0 25.0 26.0 27.0 28.0 min % % 312 55 100.0 1 285 100.0 2 313 109 75.0 75.0 266 259287 342 166 50.0 238 50.0 248 25.0 183 25.0 63 109 0.0 0.0 100 200 300 400 100 200 300 400 % 86 100.0 3 75.0 50.0 25.0 58 99 183 315 387 0.0 100 200 300 400 Fig.