Medicines Formulary
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Ophthalmic Adverse Effects of Nasal Decongestants on an Experimental
A RQUIVOS B RASILEIROS DE ORIGINAL ARTICLE Ophthalmic adverse effects of nasal decongestants on an experimental rat model Efeitos oftálmicos adversos de descongestionantes nasais em modelo experimental com ratos Ayse Ipek Akyuz Unsal1, Yesim Basal2, Serap Birincioglu3, Tolga Kocaturk1, Harun Cakmak1, Alparslan Unsal4, Gizem Cakiroz5, Nüket Eliyatkın6, Ozden Yukselen7, Buket Demirci5 1. Department of Ophthalmology, Medical Faculty, Adnan Menderes University, Aydin, Turkey. 2. Department of Otorhinolaringology, Medical Faculty, Adnan Menderes University, Aydin, Turkey. 3. Department of Pathology, Veterinary Faculty, Adnan Menderes University, Aydin, Turkey. 4. Department of Radiology, Medical Faculty, Adnan Menderes University, Aydin, Turkey. 5. Department of Medical Pharmacology, Medical Faculty, Adnan Menderes University, Aydin, Turkey. 6. Department of Medical Pathology, Medical Faculty, Adnan Menderes University, Aydin, Turkey. 7. Department of Pathology, Aydin State Hospital, Aydin, Turkey. ABSTRACT | Purpose: To investigate the potential effects of cause ophthalmic problems such as dry eyes, corneal edema, chronic exposure to a nasal decongestant and its excipients cataracts, retinal nerve fiber layer, and vascular damage in on ocular tissues using an experimental rat model. Methods: rats. Although these results were obtained from experimental Sixty adult male Wistar rats were randomized into six groups. animals, ophthalmologists should keep in mind the potential The first two groups were control (serum physiologic) and ophthalmic adverse effects of this medicine and/or its excipients Otrivine® groups. The remaining four groups received the and exercise caution with drugs containing xylometazoline, Otrivine excipients xylometazoline, benzalkonium chloride, ethylene diamine tetra acetic acid, benzalkonium chloride and sorbitol, and ethylene diamine tetra acetic acid. Medications sorbitol for patients with underlying ocular problems. -

(CD-P-PH/PHO) Report Classification/Justifica
COMMITTEE OF EXPERTS ON THE CLASSIFICATION OF MEDICINES AS REGARDS THEIR SUPPLY (CD-P-PH/PHO) Report classification/justification of medicines belonging to the ATC group R01 (Nasal preparations) Table of Contents Page INTRODUCTION 5 DISCLAIMER 7 GLOSSARY OF TERMS USED IN THIS DOCUMENT 8 ACTIVE SUBSTANCES Cyclopentamine (ATC: R01AA02) 10 Ephedrine (ATC: R01AA03) 11 Phenylephrine (ATC: R01AA04) 14 Oxymetazoline (ATC: R01AA05) 16 Tetryzoline (ATC: R01AA06) 19 Xylometazoline (ATC: R01AA07) 20 Naphazoline (ATC: R01AA08) 23 Tramazoline (ATC: R01AA09) 26 Metizoline (ATC: R01AA10) 29 Tuaminoheptane (ATC: R01AA11) 30 Fenoxazoline (ATC: R01AA12) 31 Tymazoline (ATC: R01AA13) 32 Epinephrine (ATC: R01AA14) 33 Indanazoline (ATC: R01AA15) 34 Phenylephrine (ATC: R01AB01) 35 Naphazoline (ATC: R01AB02) 37 Tetryzoline (ATC: R01AB03) 39 Ephedrine (ATC: R01AB05) 40 Xylometazoline (ATC: R01AB06) 41 Oxymetazoline (ATC: R01AB07) 45 Tuaminoheptane (ATC: R01AB08) 46 Cromoglicic Acid (ATC: R01AC01) 49 2 Levocabastine (ATC: R01AC02) 51 Azelastine (ATC: R01AC03) 53 Antazoline (ATC: R01AC04) 56 Spaglumic Acid (ATC: R01AC05) 57 Thonzylamine (ATC: R01AC06) 58 Nedocromil (ATC: R01AC07) 59 Olopatadine (ATC: R01AC08) 60 Cromoglicic Acid, Combinations (ATC: R01AC51) 61 Beclometasone (ATC: R01AD01) 62 Prednisolone (ATC: R01AD02) 66 Dexamethasone (ATC: R01AD03) 67 Flunisolide (ATC: R01AD04) 68 Budesonide (ATC: R01AD05) 69 Betamethasone (ATC: R01AD06) 72 Tixocortol (ATC: R01AD07) 73 Fluticasone (ATC: R01AD08) 74 Mometasone (ATC: R01AD09) 78 Triamcinolone (ATC: R01AD11) 82 -

(12) Patent Application Publication (10) Pub. No.: US 2006/0110428A1 De Juan Et Al
US 200601 10428A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2006/0110428A1 de Juan et al. (43) Pub. Date: May 25, 2006 (54) METHODS AND DEVICES FOR THE Publication Classification TREATMENT OF OCULAR CONDITIONS (51) Int. Cl. (76) Inventors: Eugene de Juan, LaCanada, CA (US); A6F 2/00 (2006.01) Signe E. Varner, Los Angeles, CA (52) U.S. Cl. .............................................................. 424/427 (US); Laurie R. Lawin, New Brighton, MN (US) (57) ABSTRACT Correspondence Address: Featured is a method for instilling one or more bioactive SCOTT PRIBNOW agents into ocular tissue within an eye of a patient for the Kagan Binder, PLLC treatment of an ocular condition, the method comprising Suite 200 concurrently using at least two of the following bioactive 221 Main Street North agent delivery methods (A)-(C): Stillwater, MN 55082 (US) (A) implanting a Sustained release delivery device com (21) Appl. No.: 11/175,850 prising one or more bioactive agents in a posterior region of the eye so that it delivers the one or more (22) Filed: Jul. 5, 2005 bioactive agents into the vitreous humor of the eye; (B) instilling (e.g., injecting or implanting) one or more Related U.S. Application Data bioactive agents Subretinally; and (60) Provisional application No. 60/585,236, filed on Jul. (C) instilling (e.g., injecting or delivering by ocular ion 2, 2004. Provisional application No. 60/669,701, filed tophoresis) one or more bioactive agents into the Vit on Apr. 8, 2005. reous humor of the eye. Patent Application Publication May 25, 2006 Sheet 1 of 22 US 2006/0110428A1 R 2 2 C.6 Fig. -

Review of the Existing Recommendations for Essential Medicines for Ear, Nose and Throat Conditions in Adults and Children and Suggested Modifications
REVIEW OF THE EXISTING RECOMMENDATIONS FOR ESSENTIAL MEDICINES FOR EAR, NOSE AND THROAT CONDITIONS IN ADULTS AND CHILDREN AND SUGGESTED MODIFICATIONS 2012 Shelly Chadha, Andnet Kebede Prevention of Blindness and Deafness, World Health Organization REVIEW OF THE EXISTING RECOMMENDATIONS FOR ESSENTIAL MEDICINES (Ear, Nose and Throat conditions) FOR USE IN ADULTS AND CHILDREN AND SUGGESTED MODIFICATIONS Context: The WHO Essential Medicines List includes a section for ENT conditions in children. The current section does not make any reference to medicines and dosages recommended for adults. As most of the conditions for which the listed medicines are indicated, are common in adults, the list needs to be appropriately reviewed in that context. Methodology: Each of the medicines listed in the EML for children was reviewed to consider its appropriateness for inclusion in the EML for adults. The recommended dosages for adults and children were also considered. Recommendations: The medicine list should include a list for adults as well as children. The list for adults should include Xylometazoline hydrochloride nasal spray 0.1%. It should be stated that the medicine (xylometazoline nasal spray) should not be used for prolonged periods of time, unless specifically advised and under medical supervision. The adult list should also include the other medicines mentioned in the list of children, i.e.: o Ciprofloxacin ear drops: 0.3%, as hydrochloride, for tropical use. o Aectic acid ear drops, 2% in alcohol, for topical use. Budesonide nasal spray listed in the EML for children needs greater, in depth review, which may be considered for the next EML update. 1 Xylometazoline Hydrochloride nasal spray Indications: Nasal congestion is obstruction of nasal passages, mainly caused by mucosal inflammation due to increased venous engorgement, nasal secretions and tissue swelling. -

Conceptual Model for Using Imidazoline Derivative Solutions in Pulpal Management
Journal of Clinical Medicine Review Conceptual Model for Using Imidazoline Derivative Solutions in Pulpal Management Robert S. Jones Division of Pediatric Dentistry, Department of Developmental & Surgical Sciences, School of Dentistry, University of Minnesota, Minneapolis, MN 55455, USA; [email protected] Abstract: Alpha-adrenergic agonists, such as the Imidazoline derivatives (ImDs) of oxymetazoline and xylometazoline, are highly effective hemostatic agents. ImDs have not been widely used in dentistry but their use in medicine, specifically in ophthalmology and otolaryngology, warrants consideration for pulpal hemostasis. This review presents dental healthcare professionals with an overview of ImDs in medicine. ImD solutions have the potential to be more effective and biocompatible than existing topical hemostatic compounds in pulpal management. Through a comprehensive analysis of the pharmacology of ImDs and the microphysiology of hemostasis regulation in oral tissues, a conceptual model of pulpal management by ImD solutions is presented. Keywords: hemostasis; alpha-adrenergic agonists; imidazoline; oxymetazoline; nasal; dental pulp; mucosa; apexogenesis; pulpotomy; direct pulp cap; dentistry 1. Overview Citation: Jones, R.S. Conceptual The purpose of this review is to formulate a conceptual model on the potential man- Model for Using Imidazoline agement of pulpal tissue by imidazoline derivatives (ImDs) based on a review of the Derivative Solutions in Pulpal literature that examines the hemostatic properties and mechanistic actions of these com- Management. J. Clin. Med. 2021, 10, 1212. https://doi.org/10.3390/ pounds in other human tissues. Commercial ImDs are formulated in solution with an- jcm10061212 timicrobial preservatives in order to act as ‘parenteral topical agents’ and used to manage ophthalmic inflammation, nasal congestion, and to control bleeding during otolaryngology Academic Editor: Rosalia surgery [1,2]. -

Bonviva, INN- Ibandronic Acid
European Medicines Agency London, 1 September 2005 Doc.Ref.: EMEA/278873/2005 Bonviva International Nonproprietary Name: Ibandronic acid Following the procedure EMEA/H/C/501/X/01 7 Westferry Circus, Canary Wharf, London E14 4HB, UK Tel. (44-20) 74 18 84 00 Fax (44-20) 74 18 86 68 E-mail: [email protected] http://www.emea.eu.int EMEA 2005 Reproduction and/or distribution of this document is authorised for non commercial purposes only provided the EMEA is acknowledged 1 SCIENTIFIC DISCUSSION 1.1 Introduction and rationale The MAH submitted an extension application under Annex II, point 2 iii to Commission Regulation (EC) No 1085/2003 to request the approval of a 150 mg tablet as a monthly dosing regimen of ibandronate only for the indication of “treatment of osteoporosis in postmenopausal women, in order to reduce the risk of vertebral fractures”. This claim is based on the results of a phase III study MOBILE (BM16549) comparing 100 and 150 mg once monthly to 2.5 mg once daily. Due to the inconveniences associated with intake of oral bisphosphonates (i.e. fasting conditions, frequent upper gastrointestinal intolerance) that may result in poor compliance, it was considered desirable to develop a more convenient drug formulation. Hence a 150 mg once monthly oral regimen is expected to offer greater convenience to postmenopausal women when compared to the currently approved 2.5mg once daily tablet. The development programme No new pre-clinical pharmacodynamic and pharmacokinetic studies have been performed in addition to those included in the previous submission for ibandronate 2.5 mg daily oral tablets. -

CCA Senior Care Options Formulary
Commonwealth Care Alliance Senior Care Option HMO SNP 2021 List of Covered Drugs Formulary 30 Winter Street • Boston, MA 02108 PLEASE READ: THIS DOCUMENT CONTAINS INFORMATION ABOUT THE DRUGS WE COVER IN THIS PLAN This formulary was updated on 08/01/2021. For more recent information or other questions, please contact Senior Care Options Program (HMO SNP) Member Services, at 1-866-610-2273 or, for TTY users, 711, 8 a.m. – 8 p.m., 7 days a week, or visit www.commonwealthcaresco.org. HPMS Approved Formulary File Submission ID 00021589, Version Number 13 Senior Care Options Program (HMO SNP) 2021 Formulary (List of Covered Drugs) PLEASE READ: THIS DO CUMENT CONTAINS INFORMATION ABOUT THE DRUGS WE COVER IN THIS PLAN HPMS Approved Formulary File Submission ID 00021589, Version Number 13 Note to existing members: This formulary has changed since last year. Please review this document to make sure that it still contains the drugs you take. When this drug list (formulary) refers to “we,” “us”, or “our,” it means Commonwealth Care Alliance. When it refers to “plan” or “our plan,” it means 2021 Senior Care Options Program. This document includes list of the drugs (formulary) for our plan which is current as of 08/01/2021. This formulary document applies to all SCO members. For an updated formulary, please contact us. Our contact information, along with the date we last updated the formulary, appears on the front and back cover pages. You must generally use network pharmacies to use your prescription drug benefit. Benefits, formulary, pharmacy n etwork, and/or copayments/coinsurance may change on January 1, 2022, and from time to time during the year. -

Package Leaflet
English Master Package Leaflet, PACKAGE LEAFLET 1 PACKAGE LEAFLET: INFORMATION FOR THE USER Xylometazoline preservative free TAKEDA, 0,5 mg/ml and 1 mg/ml nasal spray, solution [To be completed nationally] Xylometazoline hydrochloride Read all of this leaflet carefully because it contains important information for you. This medicine is available without prescription. However, you still need to use Xylometazoline preservative free TAKEDA carefully to get the best results from it. - Keep this leaflet. You may need to read it again. - Ask your pharmacist if you need more information or advice. - You must contact a doctor if your symptoms worsen or do not improve after 5 days (0,5 mg/ml) or 10 days (1 mg/ml). - If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist. In this leaflet: 1. What Xylometazoline preservative free TAKEDA is and what it is used for 2. Before you use Xylometazoline preservative free TAKEDA 3. How to use Xylometazoline preservative free TAKEDA 4. Possible side effects 5. How to store Xylometazoline preservative free TAKEDA 6. Further information 1. WHAT XYLOMETAZOLINE PRESERVATIVE FREE TAKEDA IS AND WHAT IT IS USED FOR Xylometazoline preservative free TAKEDA is used for symptomatic relief of nasal congestion caused by rhinitis or sinusitis. Xylometazoline preservative free TAKEDA contains xylometazoline hydrochloride, which makes the blood vessels in the nose contract, thus enables you to breathe easier through the nose. 2. BEFORE YOU USE XYLOMETAZOLINE PRESERVATIVE FREE TAKEDA Do not use Xylometazoline preservative free TAKEDA - if you are allergic (hypersensitive) to xylometazoline hydrochloride or any of the other ingredients of Xylometazoline preservative free TAKEDA. -

Bisphosphonate Use in Patients with Breast Cancer
Bisphosphonate use in patients with breast cancer Information for patients, relatives and carers For more information, please contact: Medicines Information Email: [email protected] York Hospital Medicines Information Tel: 01904 725960 Page 2 Contents Page Why have I been given this leaflet? ............................... 4 How is the treatment given? .......................................... 4 How long will I need to take the treatment for? .............. 6 Can I take other medicines at the same time? ............... 6 Who will prescribe the medication? ............................... 7 What if I am already on bisphosphonate treatment? ...... 7 Bisphosphonates are not licensed for reducing the risk of breast cancer recurrence. What does this mean? ...... 8 Are there any side effects? ............................................ 9 What is osteonecrosis? ............................................... 10 Are there any signs and symptoms I should look out for during treatment? ........................................................ 11 How can I decrease the risk of developing osteonecrosis of the jaw? ................................................................... 12 What do I need to do before starting treatment? .......... 13 Will I require any monitoring, or blood tests? ............... 14 Should I stop taking the bisphosphonate? ................... 14 Tell us what you think of this leaflet ............................. 15 Teaching, training and research ................................... 15 Patient Advice and Liaison Service (PALS) .................. 15 Page 3 Why have I been given this leaflet? You have been given this leaflet because you are going to be treated with a bisphosphonate medicine (zoledronic acid and/or ibandronic acid). In post-menopausal women, with early breast cancer, these drugs have been shown to reduce the risk of the disease recurring and increase length of life. How is the treatment given? The treatment will usually be provided as an oral tablet of ibandronic acid (one 50mg tablet taken once daily). -
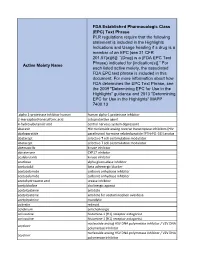
Active Moiety Name FDA Established Pharmacologic Class (EPC) Text
FDA Established Pharmacologic Class (EPC) Text Phrase PLR regulations require that the following statement is included in the Highlights Indications and Usage heading if a drug is a member of an EPC [see 21 CFR 201.57(a)(6)]: “(Drug) is a (FDA EPC Text Phrase) indicated for [indication(s)].” For Active Moiety Name each listed active moiety, the associated FDA EPC text phrase is included in this document. For more information about how FDA determines the EPC Text Phrase, see the 2009 "Determining EPC for Use in the Highlights" guidance and 2013 "Determining EPC for Use in the Highlights" MAPP 7400.13. .alpha. -

Illness Code A
Long Term Illness (LTI) Scheme: Reference Guide as to what is covered in the Health Service Executive for each of the 16 scheduled LTI illnesses. (January 2021) Illness Code C: Cerebral Palsy Antibiotics 1. Doxycycline J01AA02 2. Lymecycline J01AA04 3. Minocycline J01AA08 4. Ampicillin J01CA01 5. Amoxicillin J01CA04 6. Azlocillin J01CA09 7. Benzylpenicillin J01CE01 8. Phenoxymethylpenicillin J01CE02 9. Flucloxacillin J01CF05 10. Amoxicillin & Enzyme Inhibitor J01CR02 11. Cefalexin J01DB01 12. Cefuroxime J01DC02 13. Cefaclor J01DC04 14. Ceftazidime J01DD02 15. Ceftriaxone J01DD04 16. Cefixime J01DD08 17. Cefpodoxime J01DD13 18. Trimethoprim J01EA01 19. Sulfamethoxazole & Trimethoprim J01EE01 20. Erythromycin J01FA01 21. Clarithromycin J01FA09 22. Azithromycin J01FA10 23. Clindamycin J01FF01 24. Gentamicin J01GB03 25. Amikacin J01GB06 26. Ofloxacin J01MA01 27. Ciprofloxacin J01MA02 28. Levofloxacin J01MA12 29. Moxifloxacin J01MA14 30. Vancomycin J01XA01 31. Teicoplanin J01XA02 32. Colistin J01XB01 33. Fusidic Acid J01XC01 34. Nitrofurantoin J01XE01 Muscle relaxants 1. Baclofen M03BX01 2. Tizanidine M03BX02 3. Dantrolene M03CA01 Anticonvulsants (including Benzodiazepines used for this purpose) 1. Phenobarbital N03AA02 2. Primidone N03AA03 page 1 3. Phenytoin N03AB02 4. Ethosuximide N03AD01 5. Clonazepam N03AE01 6. Carbamazepine N03AF01 7. Oxcarbazepine N03AF02 8. Rufinamide N03AF03 9. Eslicarbazepine N03AF04 10. Valproic Acid N03AG01 11. Vigabatrin N03AG04 12. Tiagabine N03AG06 13. Lamotrigine N03AX09 14. Topiramate N03AX11 15. Gabapentin N03AX12 16. Levetiracetam N03AX14 17. Zonisamide N03AX15 18. Pregabalin N03AX16 19. Lacosamide N03AX18 20. Retigabine N03AX21 21. Perampanel N03AX22 22. Diazepam (Rectal) N05BA01 23. Clobazam N05BA09 24. Midazolam N05CD08 25. Acetazolamide S01EC01 Anxiolytics 1. Diazepam N05BA01 2. Chlordiazepoxide N05BA02 3. Lorazepam N05BA06 4. Bromazepam N05BA08 5. Clobazam N05BA09 6. Prazepam N05BA11 7. Alprazolam N05BA12 Hypnotics 8. -

World Health Organization Model List of Essential Medicines, 21St List, 2019
World Health Organizatio n Model List of Essential Medicines 21st List 2019 World Health Organizatio n Model List of Essential Medicines 21st List 2019 WHO/MVP/EMP/IAU/2019.06 © World Health Organization 2019 Some rights reserved. This work is available under the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 IGO licence (CC BY-NC-SA 3.0 IGO; https://creativecommons.org/licenses/by-nc-sa/3.0/igo). Under the terms of this licence, you may copy, redistribute and adapt the work for non-commercial purposes, provided the work is appropriately cited, as indicated below. In any use of this work, there should be no suggestion that WHO endorses any specific organization, products or services. The use of the WHO logo is not permitted. If you adapt the work, then you must license your work under the same or equivalent Creative Commons licence. If you create a translation of this work, you should add the following disclaimer along with the suggested citation: “This translation was not created by the World Health Organization (WHO). WHO is not responsible for the content or accuracy of this translation. The original English edition shall be the binding and authentic edition”. Any mediation relating to disputes arising under the licence shall be conducted in accordance with the mediation rules of the World Intellectual Property Organization. Suggested citation. World Health Organization Model List of Essential Medicines, 21st List, 2019. Geneva: World Health Organization; 2019. Licence: CC BY-NC-SA 3.0 IGO. Cataloguing-in-Publication (CIP) data. CIP data are available at http://apps.who.int/iris.