Chapter 2: Alkanes Alkanes from Carbon and Hydrogen
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Wo 2009/082437 A9
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) CORRECTED VERSION (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date 2 July 2009 (02.07.2009) WO 2009/082437 A9 (51) International Patent Classification: KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, ME, C07D 207/08 (2006.01) A61K 31/402 (2006.01) MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, C07D 207/09 (2006.01) A61P 5/26 (2006.01) NZ, OM, PG, PH, PL, PT, RO, RS, RU, SC, SD, SE, SG, C07D 498/04 (2006.01) SK, SL, SM, ST, SV, SY, TJ, TM, TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (21) International Application Number: PCT/US2008/013657 (84) Designated States (unless otherwise indicated, for every kind of regional protection available): ARIPO (BW, GH, (22) International Filing Date: G M M N S D S L s z τ z U G Z M 12 December 2008 (12.12.2008) z w Eurasian (A M B γ KG> M D RU> τ (25) Filing Language: English TM), European (AT, BE, BG, CH, CY, CZ, DE, DK, EE, ES, FI, FR, GB, GR, HR, HU, IE, IS, IT, LT, LU, LV, (26) Publication Language: English M C M T N L N O P L P T R O S E S I s T R OAPI (30) Priority Data: B F ' B J ' C F ' C G ' C I' C M ' G A ' G N ' 0 G W ' M L ' M R ' 61/008,731 2 1 December 2007 (21 .12.2007) US N E ' S N ' T D ' T G ) - (71) Applicant (for all designated States except US): LIG- Declarations under Rule 4.17: AND PHARMACEUTICALS INCORPORATED [US/ — as to applicant's entitlement to apply for and be granted US]; 11085 N. -

Transport of Dangerous Goods
ST/SG/AC.10/1/Rev.16 (Vol.I) Recommendations on the TRANSPORT OF DANGEROUS GOODS Model Regulations Volume I Sixteenth revised edition UNITED NATIONS New York and Geneva, 2009 NOTE The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of the Secretariat of the United Nations concerning the legal status of any country, territory, city or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries. ST/SG/AC.10/1/Rev.16 (Vol.I) Copyright © United Nations, 2009 All rights reserved. No part of this publication may, for sales purposes, be reproduced, stored in a retrieval system or transmitted in any form or by any means, electronic, electrostatic, magnetic tape, mechanical, photocopying or otherwise, without prior permission in writing from the United Nations. UNITED NATIONS Sales No. E.09.VIII.2 ISBN 978-92-1-139136-7 (complete set of two volumes) ISSN 1014-5753 Volumes I and II not to be sold separately FOREWORD The Recommendations on the Transport of Dangerous Goods are addressed to governments and to the international organizations concerned with safety in the transport of dangerous goods. The first version, prepared by the United Nations Economic and Social Council's Committee of Experts on the Transport of Dangerous Goods, was published in 1956 (ST/ECA/43-E/CN.2/170). In response to developments in technology and the changing needs of users, they have been regularly amended and updated at succeeding sessions of the Committee of Experts pursuant to Resolution 645 G (XXIII) of 26 April 1957 of the Economic and Social Council and subsequent resolutions. -

United States Patent (19) 11) Patent Number: 5,072,065 Sun Et Al
United States Patent (19) 11) Patent Number: 5,072,065 Sun et al. 45 Date of Patent: Dec. 10, 1991 54 NHIBITING POPCORN POLYMER FORMATION WITH ALKYL HALDES FOREIGN PATENT DOCUMENTS 63-223003 9/1988 Japan. (75) inventors: Hsiang-ning Sun, Houston, Tex.; Steven J. Potlock, Baton Rouge, La. OTHER PUBLICATIONS Liu, Plugging-Up of Equipment by Self-Polymeriza (73) Assignee: Exxon Chemical Patents Inc., tion Butadiene Production and its Prevention, China Linden, N.J. Synthetic Rubber Industry, 11(5) 356-360 (1988) & English abstract. 21 Appl. No.: 647,358 Liu et al., Determination of Traces of Diethylhydrox ylamine Inhibitor in c5 Fraction by Gas Chromatogra 22 Fied: Jan. 29, 1991 phy, China Synthetic Rubber Industry, 12(6), 408-410 (1989) & English abstract. (51 Int, C. ........................... C07C 7/20; B08B 9/00 (52) U.S. C. ..................................... 585/2; 134/22.19; Primary Examiner-Curtis R. Davis 585/3,585/950 Attorney, Agent, or Firm-Linda K. Russell (58 Field of Search ............................... 585/3, 2,950; 57 ABSTRACT 34/22.19 Inhibition of popcorn polymer growth by treatment with an alkyl halide. This compound can be admixed (56) References Cited with organic material from which popcorn polymer is U.S. PATENT DOCUMENTS formed, or added to a system for the recovery, use or 4,404,413 9/1983 Heskell ................................ 585/950 storage of such organic material. 4,941,926 7/1990 Nakajima ................................ 585/5 4,956,020 9/1990 Nakajima ................................ 585/5 16 Claims, No Drawings 5,072,065 1. 2 once present in a system. Some of the seeds become NHIBITING POPCORN POLYMER FORMATION attached to the processing and handling equipment, and WITH ALKYL HALDES cannot be readily removed by mechanical means; more over, being insoluble in most common solvents, they are CONCURRENTLY FILED APPLICATIONS virtually impossible to wash out by use of such solvents. -

Aldrich Vapor
Aldrich Vapor Library Listing – 6,611 spectra This library is an ideal tool for investigator using FT-IR to analyze gas phase materials. It contains gas phase spectra collected by Aldrich using a GC-IR interface to ensure chromatographically pure samples. The Aldrich FT-IR Vapor Phase Library contains 6,611 gas phase FT-IR spectra collected by Aldrich Chemical Company using a GC interface. The library includes compound name, molecular formula, CAS (Chemical Abstract Service) registry number, Aldrich catalog number, and page number in the Aldrich Library of FT-IR Spectra, Edition 1, Volume 3, Vapor-Phase. Aldrich Vapor Index Compound Name Index Compound Name 6417 ((1- 3495 (1,2-Dibromoethyl)benzene; Styrene Ethoxycyclopropyl)oxy)trimethylsilane dibromide 2081 (+)-3-(Heptafluorobutyryl)camphor 3494 (1-Bromoethyl)benzene; 1-Phenylethyl 2080 (+)-3-(Trifluoroacetyl)camphor bromide 262 (+)-Camphene; 2,2-Dimethyl-3- 6410 (1-Hydroxyallyl)trimethylsilane methylenebicyclo[2.2.1]heptane 6605 (1-Methyl-2,4-cyclopentadien-1- 2828 (+)-Diisopropyl L-tartrate yl)manganese tricarbonyl 947 (+)-Isomenthol; [1S-(1a,2b,5b)]-2- 6250 (1-Propynyl)benzene; 1-Phenylpropyne Isopropyl-5-methylcyclohexano 2079 (1R)-(+)-3-Bromocamphor, endo- 1230 (+)-Limonene oxide, cis + trans; (+)-1,2- 2077 (1R)-(+)-Camphor; (1R)-(+)-1,7,7- Epoxy-4-isopropenyl-1- Trimethylbicyclo[2.2.1]heptan- 317 (+)-Longifolene; (1S)-8-Methylene- 976 (1R)-(+)-Fenchyl alcohol, endo- 3,3,7-trimethyltricyclo[5.4.0 2074 (1R)-(+)-Nopinone; (1R)-(+)-6,6- 949 (+)-Menthol; [1S-(1a,2b,5a)]-(+)-2- Dimethylbicyclo[3.1.1]heptan-2- -

A Low Temperature Structure of Nonane-1,9-Diaminium Chloride Chloroacetate: Hydroxyacetic Acid (1:1)
J Chem Crystallogr (2011) 41:703–707 DOI 10.1007/s10870-010-9957-6 ORIGINAL PAPER A Low Temperature Structure of Nonane-1,9-Diaminium Chloride Chloroacetate: Hydroxyacetic Acid (1:1) Agnieszka Paul • Maciej Kubicki Received: 24 February 2010 / Accepted: 31 December 2010 / Published online: 14 January 2011 Ó The Author(s) 2011. This article is published with open access at Springerlink.com Abstract The crystal structure of nonane-1,9-diaminium protons and to form salts with both, organic and inorganic chloride chloroacetate–hydroxyacetic acid (1:1) was deter- acids, together with a significant flexibility of the chain mined by X-ray diffraction at 100(1)K. The asymmetric fragment and tendency towards creation of the highly unit is composed of diaminium dication, chloroacetate and symmetric networks, can be used in so-called crystal chloride anions, and neutral hydroxyacetic acid molecule. engineering [2], i.e. in designing new materials with The aliphatic chain of 1,9-diamine is fully extended and it expected features. Such a predictable motifs in the crystal deviates only slightly from the perfect all-trans confor- structures, giving rise to a layer structure, were already mation. The two acidic residues are also nearly planar. The investigated in the series of hexane-1,6-diamine and layer structure is obtained as a consequence of hydrogen butane-1,4-diamine salts [3, 4]. bond interactions of a different lengths, with N–H and O–H Only five structures deposited in the Cambridge Struc- groups playing the role of donors and oxygen atoms and tural Database [5] (no multiple determinations, herein and chloride cations as acceptors. -

SAFETY DATA SHEET 1,9-Dibromo-Nonane-2,8-Dione According to Regulation (EC) No 1907/2006, Annex II, As Amended
Revision date: 03/01/2020 Revision: 1 SAFETY DATA SHEET 1,9-Dibromo-nonane-2,8-dione According to Regulation (EC) No 1907/2006, Annex II, as amended. Commission Regulation (EU) No 2015/830 of 28 May 2015. SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product name 1,9-Dibromo-nonane-2,8-dione Product number FD173289 CAS number 91492-76-1 1.2. Relevant identified uses of the substance or mixture and uses advised against Identified uses Laboratory reagent. Manufacture of substances. Research and development. 1.3. Details of the supplier of the safety data sheet Supplier Carbosynth Ltd 8&9 Old Station Business Park Compton Berkshire RG20 6NE UK +44 1635 578444 +44 1635 579444 [email protected] 1.4. Emergency telephone number Emergency telephone +44 7887 998634 SECTION 2: Hazards identification 2.1. Classification of the substance or mixture Classification (EC 1272/2008) Physical hazards Not Classified Health hazards Not Classified Environmental hazards Not Classified Additional information Caution. Not fully tested. 2.2. Label elements Hazard statements NC Not Classified 2.3. Other hazards No data available. SECTION 3: Composition/information on ingredients 3.1. Substances Product name 1,9-Dibromo-nonane-2,8-dione 1/8 Revision date: 03/01/2020 Revision: 1 1,9-Dibromo-nonane-2,8-dione CAS number 91492-76-1 Chemical formula C₉H₁₄Br₂O₂ SECTION 4: First aid measures 4.1. Description of first aid measures General information Get medical advice/attention if you feel unwell. Inhalation Remove person to fresh air and keep comfortable for breathing. -

Oxidation of Ethane to Ethylene and Acetic Acid by Movnbo Catalysts Martial Roussel, Michel Bouchard, Elisabeth Bordes-Richard, Khalid Karim, Saleh Al-Sayari
Oxidation of ethane to ethylene and acetic acid by MoVNbO catalysts Martial Roussel, Michel Bouchard, Elisabeth Bordes-Richard, Khalid Karim, Saleh Al-Sayari To cite this version: Martial Roussel, Michel Bouchard, Elisabeth Bordes-Richard, Khalid Karim, Saleh Al-Sayari. Oxida- tion of ethane to ethylene and acetic acid by MoVNbO catalysts. Catalysis Today, Elsevier, 2005, 99, pp.77-87. 10.1016/j.cattod.2004.09.026. hal-00103604 HAL Id: hal-00103604 https://hal.archives-ouvertes.fr/hal-00103604 Submitted on 4 Oct 2006 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Oxidation of ethane to ethylene and acetic acid by MoVNbO catalysts M. Roussel 1, M. Bouchard 1, E. Bordes-Richard 1*, K. Karim 2, S. Al-Sayari 2 1: Laboratoire de Catalyse de Lille, UMR CNRS 8010, USTL-ENSCL, 59655 Villeneuve d'Ascq Cedex, France 2: SABIC R&T, P.O. Box 42503, Riyadh, Saoudi Arabia ABSTRACT The influence of niobium on the physicochemical properties of the Mo-V-O system and on its catalytic properties in the oxidation of ethane to ethylene and acetic acid is examined. Solids based on MoV 0.4 Ox and MoV 0.4 Nb 0.12 Oy composition and calcined at 350 or 400°C were studied by X-ray diffraction, and by laser Raman and X-ray photoelectron spectroscopies. -

Enabling Ethane As a Primary Gas Turbine Fuel: an Economic Benefit from the Growth of Shale Gas
GE Power GEA32198 November 2015 Enabling ethane as a primary gas turbine fuel: an economic benefit from the growth of shale gas Dr. Jeffrey Goldmeer GE Power, Gas Power Systems Schenectady, NY, USA John Bartle GE Power, Power Services Atlanta, GA, USA Scott Peever GE Power Toronto, ON, CAN © 2015 General Electric Company. All Rights Reserved. This material may not be copied or distributed in whole or in part, without prior permission of the copyright owner. GEA32198 Enabling ethane as a primary gas turbine fuel: an economic benefit from the growth of shale gas Contents Abstract ............................................................................3 Introduction ........................................................................3 Fuel Supply Dynamics ...............................................................3 Hydrocarbon Sources and Characteristics ...........................................5 Combustion System Considerations .................................................6 Evaluating Fuels .....................................................................6 GE’s Gas Turbine Combustion Systems ..............................................8 Non-Methane Hydrocarbon Capability ...............................................9 Economic Value of Alternative Fuels .................................................9 Conclusions ........................................................................10 References ........................................................................11 2 © 2015 General Electric Company. All Rights -
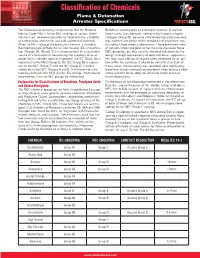
Classification of Chemicals
Classification of Chemicals Flame & Detonation Arrester Specifications PROTECTOSEAL ® The Protectoseal Company recommends that the National Butadiene would qualify as a Group D material. In each of Electric Code (NEC) Article 500, rankings of various chemi - these cases, the chemicals were primarly listed in a higher cals be used, whenever possible, to determine the suitability category (Group B), because of relatively high pressure read - of a detonation arrester for use with a particular chemical. ings noted in one phase of the standard test procedure con - When no NEC rating of the particular chemical is available, ducted by Underwriters Laboratories. These pressures were the International Electrotechnical Commission (IEC) classifica - of concern when categorizing the chemicals because these tion (Groups IIA, IIB and IIC) is recommended as a secondary NEC groupings are also used as standard indicators for the source of information for determining the suitability of an ar - design strength requirements of electrical boxes, apparatus, rester for its intended service. In general, the IEC Group IIA is etc. that must withstand the pressures generated by an igni - equivalent to the NEC Group D; the IEC Group IIB is equiva - tion within the container. It should be noted that, in each of lent to the NEC Group C; and the IEC Group IIC includes these cases, the test pressures recorded were significantly chemicals in the NEC Groups A and B. In the event of a dis - lower than those commonly encountered when testing a deto - crepancy between the NEC and the IEC ratings, Protectoseal nation arrester for its ability to withstand stable and over - recommends that the NEC groups be referenced. -

Chlorine and Hydrogen Chloride
This report contains the collective views of an nternational group of experts and does not xcessarily represent the decisions or the stated 1 icy of the United Nations Environment Pro- '€mme, the International Labour Organisation, or the World Health Organization. Environmental Health Criteria 21 CHLORINE AND HYDROGEN CHLORIDE 'ublished under the joint sponsorship of Ic United Nations Environment Programme. the International Labour Organisation, and the World Health Organization / \r4 ( o 4 UI o 1 o 'T F- World Health Organization kz Geneva, 1982 The International Programme on Chemical Safety (IPCS) is a joint ven- ture of the United Nations Environment Programme. the International Labour Organisation, and the World Health Organization. The main objective of the IPCS is to carry out and disseminate evaluations of the environment. Supporting activities include the development of epidemiological, experi- mental laboratory, and risk assessment methods that could produce interna- tionally comparable results, and the development of manpower in the field of toxicology. Other relevant activities carried out by the IPCS include the development of know-how for coping with chemical accidents, coordination of laboratory testing and epidemiological studies, and promotion of research on the mechanisms of the biological action of chemicals. ISBN 92 4 154081 8 World Health Organization 1982 Publications of the World Health Organization enjoy copyright protec- tion in accordance with the provisions of Protocol 2 of the Universal Copy- right Convention. For rights of reproduction or translation of WHO publica- tions, in part or in loto, application should be made to the Office of Publica- tions, World Health Organization, Geneva. Switzerland. The World Health Organization welcomes such applications. -

Organic Chemistry Name Formula Isomers Methane CH 1 Ethane C H
Organic Chemistry Organic chemistry is the chemistry of carbon. The simplest carbon molecules are compounds of just carbon and hydrogen, hydrocarbons. We name the compounds based on the length of the longest carbon chain. We then add prefixes and suffixes to describe the types of bonds and any add-ons the molecule has. When the molecule has just single bonds we use the -ane suffix. Name Formula Isomers Methane CH4 1 Ethane C2H6 1 Propane C3H8 1 Butane C4H10 2 Pentane C5H12 3 Hexane C6H14 5 Heptane C7H16 9 Octane C8H18 18 Nonane C9H20 35 Decane C10H22 75 Isomers are compounds that have the same formula but different bonding. isobutane n-butane 1 Naming Alkanes Hydrocarbons are always named based on the longest carbon chain. When an alkane is a substituent group they are named using the -yl ending instead of the -ane ending. So, -CH3 would be a methyl group. The substituent groups are named by numbering the carbons on the longest chain so that the first branching gets the lowest number possible. The substituents are listed alphabetically with out regard to the number prefixes that might be used. 3-methylhexane 1 2 3 4 5 6 6 5 4 3 2 1 Alkenes and Alkynes When a hydrocarbon has a double bond we replace the -ane ending with -ene. When the hydrocarbon has more than three carbon the position of the double bond must be specified with a number. 1-butene 2-butene Hydrocarbons with triple bonds are named basically the same, we replace the -ane ending with -yne. -

Cycloalkanes, Cycloalkenes, and Cycloalkynes
CYCLOALKANES, CYCLOALKENES, AND CYCLOALKYNES any important hydrocarbons, known as cycloalkanes, contain rings of carbon atoms linked together by single bonds. The simple cycloalkanes of formula (CH,), make up a particularly important homologous series in which the chemical properties change in a much more dramatic way with increasing n than do those of the acyclic hydrocarbons CH,(CH,),,-,H. The cyclo- alkanes with small rings (n = 3-6) are of special interest in exhibiting chemical properties intermediate between those of alkanes and alkenes. In this chapter we will show how this behavior can be explained in terms of angle strain and steric hindrance, concepts that have been introduced previously and will be used with increasing frequency as we proceed further. We also discuss the conformations of cycloalkanes, especially cyclo- hexane, in detail because of their importance to the chemistry of many kinds of naturally occurring organic compounds. Some attention also will be paid to polycyclic compounds, substances with more than one ring, and to cyclo- alkenes and cycloalkynes. 12-1 NOMENCLATURE AND PHYSICAL PROPERTIES OF CYCLOALKANES The IUPAC system for naming cycloalkanes and cycloalkenes was presented in some detail in Sections 3-2 and 3-3, and you may wish to review that ma- terial before proceeding further. Additional procedures are required for naming 446 12 Cycloalkanes, Cycloalkenes, and Cycloalkynes Table 12-1 Physical Properties of Alkanes and Cycloalkanes Density, Compounds Bp, "C Mp, "C diO,g ml-' propane cyclopropane butane cyclobutane pentane cyclopentane hexane cyclohexane heptane cycloheptane octane cyclooctane nonane cyclononane "At -40". bUnder pressure. polycyclic compounds, which have rings with common carbons, and these will be discussed later in this chapter.